Presence of HIV1 C broadly neutralizing antibodies in

Presence of HIV-1 C broadly neutralizing antibodies in pregnancy and at delivery Takafira Mduluza * Biochemistry Department, University of Zimbabwe * School of Laboratory Medicine and Medical Sciences, UKZN; * Botswana Harvard AIDS Institute Partnership; tmduluza@yahoo. com or mduluza@medic. uz. ac. zw #AIDS 2016 | @AIDS_conference

Main Aim Investigating HIV transmission blocking Immunity in pregnant mothers. Specific Objectives To identify plasma with broadly HIV-1 neutralizing antibody activity. To compare HIV-1 neutralizing antibody activity in plasma samples from transmitters and non-transmitters. To compare gp 120 (V 3) and gp 41 (MPER) Ig. G subclass (Ig. G 1, Ig. G 2, Ig. G 3), specific responses of plasma from transmitting versus non-transmitting mothers. #AIDS 2016 | @AIDS_conference
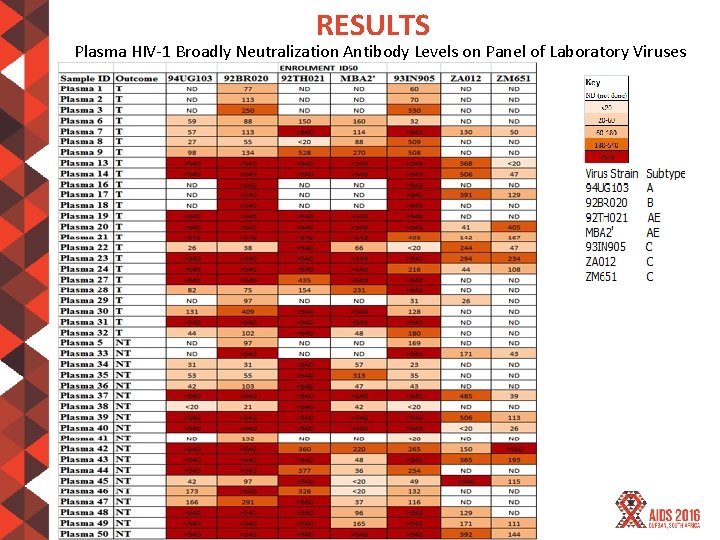
RESULTS Plasma HIV-1 Broadly Neutralization Antibody Levels on Panel of Laboratory Viruses #AIDS 2016 | @AIDS_conference
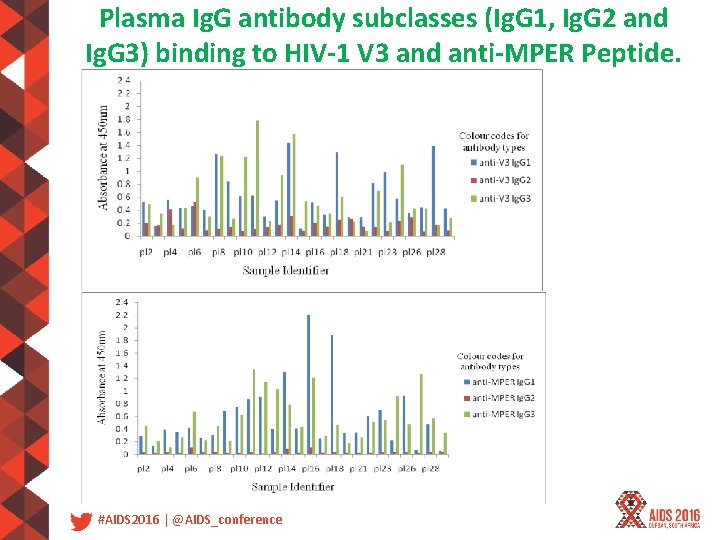
Plasma Ig. G antibody subclasses (Ig. G 1, Ig. G 2 and Ig. G 3) binding to HIV-1 V 3 and anti-MPER Peptide. #AIDS 2016 | @AIDS_conference
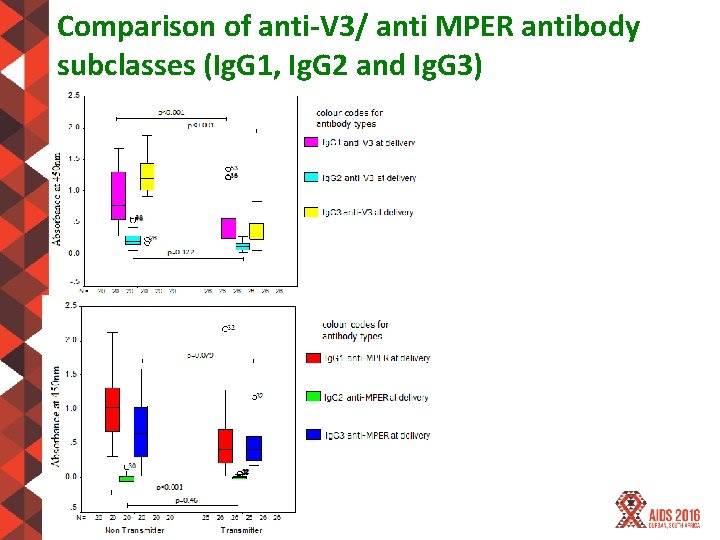
Comparison of anti-V 3/ anti MPER antibody subclasses (Ig. G 1, Ig. G 2 and Ig. G 3) #AIDS 2016 | @AIDS_conference

CONCLUSION HIV-1 broadly neutralizing antibodies are present during pregnancy and there is potential of their role in reducing mother to child transmission of HIV-1 at delivery HIV-1 broadly neutralizing antibody activity identified in samples from transmitters and non -transmitters. Anti HIV-1 C gp 120 (V 3) and gp 41 (MPER) Ig. G subclass (Ig. G 1 & Ig. G 3) found elevated in nontransmitting versus transmitting mothers. #AIDS 2016 | @AIDS_conference

ACKNOWLEDGEMENTS Sheron Dzoro Keabetswe Bedi Sununguko W. Mpoloka Participants in the MASHI study Botswana Harvard Partnership - BHP 004 Research Reagents Source: NIH AIDS Research and Reference Reagent Program. Funding: EDCTP Grant # TA. 2009. 40200. 005 #AIDS 2016 | @AIDS_conference
- Slides: 7