Prequalification of Quality Control Laboratories WHO Quality Monitoring

Prequalification of Quality Control Laboratories WHO Quality Monitoring Jitka Sabartova WHO Prequalification of Medicines Programme WHO/UNICEF Technical Briefing Seminar 1 Nov 2012, Geneva

QCLs Prequalification Procedure • Established in 2004 in cooperation with UN agencies – Initially limited to QC laboratories in Africa • 3 rd EOI published in September 2007, without regional limits – http: //www. who. int/prequal/info_applicants/eoi/EOI-QCLabs. V 3. pdf • Participation of a QC laboratory is voluntary – Any laboratory (private or governmental) can participate – Currently free of charge • Scope - chemical and microbiological testing (including LAL test) of medicines (vaccines, biologicals not included) • Priority in the assessment is given to – National QC laboratories and laboratories providing testing services to the governments – QC laboratories in areas where UN agencies identify the need for quality testing WHO/UNICEF TBS, Geneva, 1 Nov 2012 2
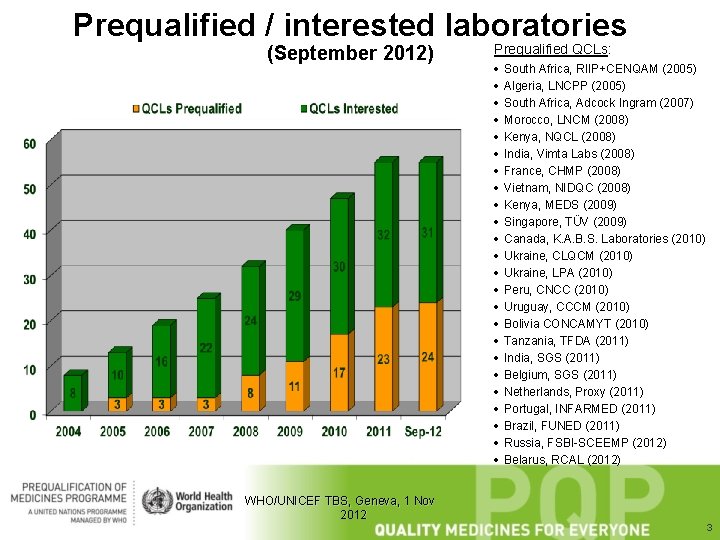
Prequalified / interested laboratories (September 2012) WHO/UNICEF TBS, Geneva, 1 Nov 2012 Prequalified QCLs: · · · · · · South Africa, RIIP+CENQAM (2005) Algeria, LNCPP (2005) South Africa, Adcock Ingram (2007) Morocco, LNCM (2008) Kenya, NQCL (2008) India, Vimta Labs (2008) France, CHMP (2008) Vietnam, NIDQC (2008) Kenya, MEDS (2009) Singapore, TÜV (2009) Canada, K. A. B. S. Laboratories (2010) Ukraine, CLQCM (2010) Ukraine, LPA (2010) Peru, CNCC (2010) Uruguay, CCCM (2010) Bolivia CONCAMYT (2010) Tanzania, TFDA (2011) India, SGS (2011) Belgium, SGS (2011) Netherlands, Proxy (2011) Portugal, INFARMED (2011) Brazil, FUNED (2011) Russia, FSBI-SCEEMP (2012) Belarus, RCAL (2012) 3
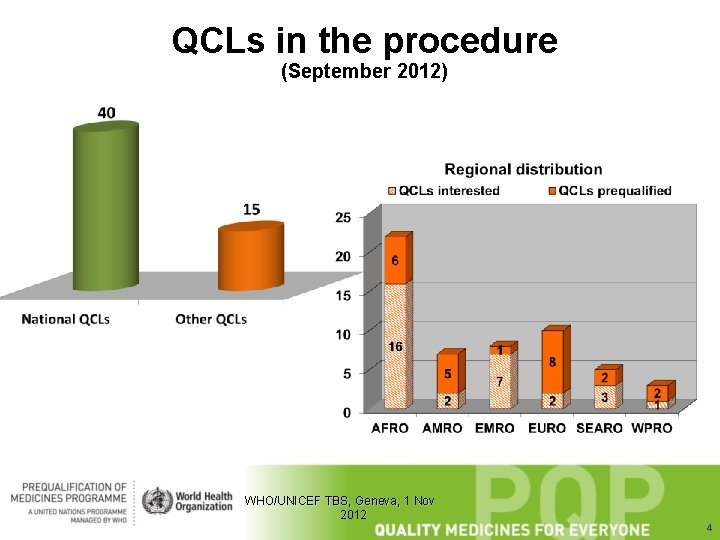
QCLs in the procedure (September 2012) WHO/UNICEF TBS, Geneva, 1 Nov 2012 4
WHO/UNICEF TBS, Geneva, 1 Nov 2012 5
WHO/UNICEF TBS, Geneva, 1 Nov 2012 6

Capacity building • Technical assistance provided to national medicines QCLs in developing countries – 18 since 2006 (6 in 2011, 4 in 2012) – Focus on implementation of quality system, microbiology testing • Training – Training in dissolution and water determination (organized with AFSSAPS and EDQM in September 2011, Senegal) – Training for microbiological laboratories (November 2011, Jordan) – Workshops on laboratory quality control of reproductive health products (organized with UNFPA in November-December 2011 Tanzania and Namibia; February 2012 – Ghana; November 2012 – Thailand) • External Quality Assessment Scheme for National Drug Quality Control Laboratories WHO/UNICEF TBS, Geneva, 1 Nov 2012 7

Potential benefits of PQ for QCLs • Possibility to provide testing services to UN agencies and other organizations - financial profit • Recognition as being WHO listed laboratory • Facilitated discussions with manufacturers/customers in case of non-compliant results • Learning process improving the standards of laboratory work • In case on a national QCLs in a developing country, possibility to be assisted by WHO expert consultants and participate in WHO organized trainings WHO/UNICEF TBS, Geneva, 1 Nov 2012 8

WHO/UNICEF TBS, Geneva, 1 Nov 2012 9
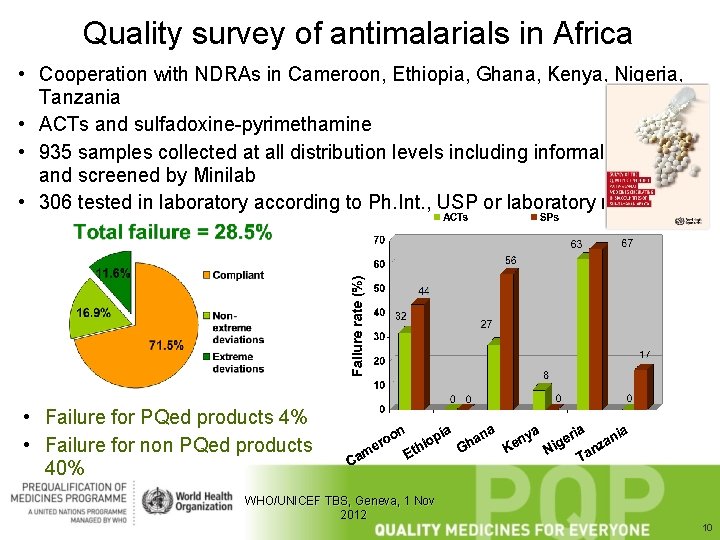
Quality survey of antimalarials in Africa • Cooperation with NDRAs in Cameroon, Ethiopia, Ghana, Kenya, Nigeria, Tanzania • ACTs and sulfadoxine-pyrimethamine • 935 samples collected at all distribution levels including informal market and screened by Minilab • 306 tested in laboratory according to Ph. Int. , USP or laboratory method • Failure for PQed products 4% • Failure for non PQed products 40% WHO/UNICEF TBS, Geneva, 1 Nov 2012 10

Quality survey of anti-TB medicines in NIS • Cooperation with NDRAs in Armenia, Azerbaijan, Belarus, Kazakhstan, Ukraine, Uzbekistan • Rifampicin, Isoniazid, Rifampicin/Isoniazid, Ofloxacin, Kanamycin • 291 samples collected at hospitals, dispensaries, pharmacies and tested according to Ph. Int. or USP • None of 38 samples of WHO-prequalified products failed WHO/UNICEF TBS, Geneva, 1 Nov 2012 11

What should be kept in mind when organizing testing of procured medicines • Considerations on necessity/frequency of testing based on risk evaluation – GMP compliance of the manufacturer, product prequalified or registered by SRA • Specifications for testing should be agreed with the supplier in advance – Manufacturer's specifications (with method transfer by the lab) or a pharmacopoeia monograph • Testing performed by a reliable laboratory – Out-of-specification results investigated by the lab – Discussion with the manufacturer – PQP should be informed about problems with PQed products WHO/UNICEF TBS, Geneva, 1 Nov 2012 12

Thanks for your attention sabartovaj@who. int prequallaboratories@who. int www. who. int/prequal WHO/UNICEF TBS, Geneva, 1 Nov 2012 13
- Slides: 13