Prepared by Timothy John D Matoy First described

Prepared by: Timothy John D. Matoy

§First described by ancient Greeks §Today, they are known as electromagnetic Energy §No mass, no identifiable form §They do have electric and magnetic fields that are continuously changing in sinusoidal fashion.

§A photon is the smallest quantity of any type of electromagnetic energy. §A photon may be pictured as a small bundle of energy, sometimes called quantum.

§Late in 19 th century. §Showed that visible light has both electric and magnetic properties, hence the term electromagnetic energy.

§It can travel through empty space. §It travels with the speed of light. §It travels in a wave-like fashion.

§The velocity of all electromagnetic 8 radiation is 3 x 10 m/s or 186, 000 miles per second.

§The distance from one crest to another, from one valley to another, or from any point on the sine wave to the next corresponding point.
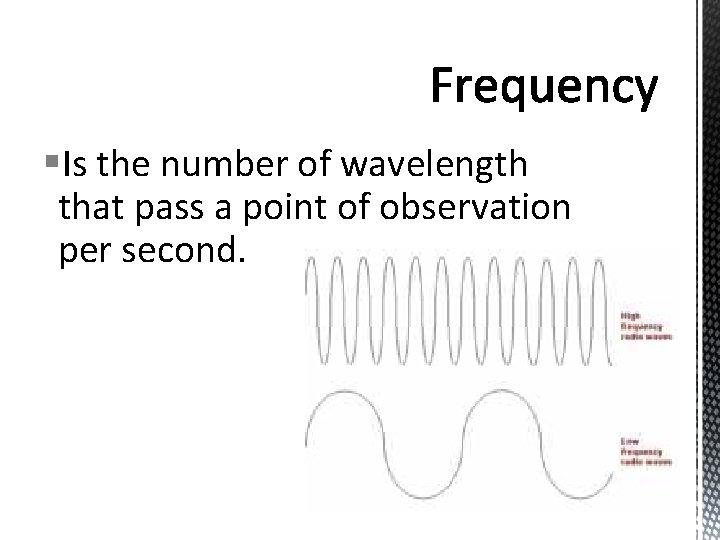
§Is the number of wavelength that pass a point of observation per second.

§V = λf §Velocity = wavelength x frequency

§C = λf §Speed of light = wavelength x frequency

§The highest energy x-ray produced at 120 k. Vp has a 19 frequency of 2. 50 x 10 Hz. What is the wavelength?

§What is the frequency of a certain electromagnetic wave with a wavelength of 300 cm?

§The speed of sound in air is approximately 340 m/s. The highest treble tone that the person can hear is about 20 k. Hz. What is the wavelength of this sound?

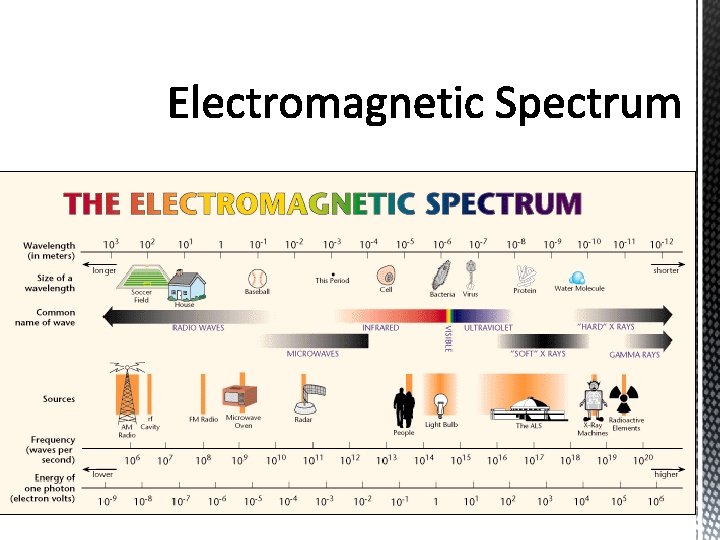
§The frequency range of electromagnetic energy extends from approximately 102 to 1024 Hz. §The wavelength are approximately 107 to 10 -16 m.
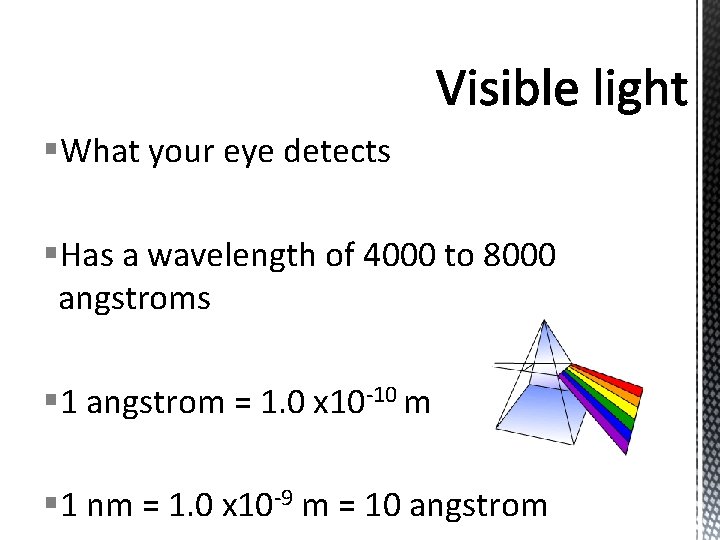
§What your eye detects §Has a wavelength of 4000 to 8000 angstroms § 1 angstrom = 1. 0 x 10 -10 m § 1 nm = 1. 0 x 10 -9 m = 10 angstrom

§Relatively low energy and relatively very long wavelength

§Gamma ray §X-ray

§Photons interact with matter most easily when matter is approximately the same size as the photon wavelength. §All radiation with wavelength longer than those of x-radiation interacts primarily as a wave phenomenon.

§“X-rays are created with the speed of light (c), and they exist with the velocity (c) or they do not exist at all” – Max Planck

§E = hf §Where: §E = photon energy §h = Planck’s constant §f = photon frequency

§Also known as Planck’s constant (h), has a numeric value of 4. 15 x 10 -15 e. Vs or 6. 63 x 10 -34 Js.

§What is the frequency of a 70 ke. V x-ray?

§What is the energy of a 960 k. Hz electromagnetic wave?

§What is the frequency of a 86 ke. V x-ray?

§Law of conservation of matter §Law of conservation of energy
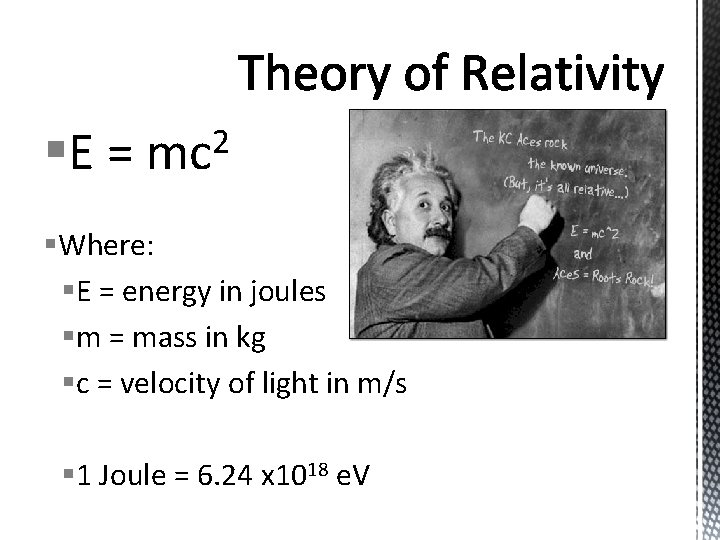
§E = 2 mc §Where: §E = energy in joules §m = mass in kg §c = velocity of light in m/s § 1 Joule = 6. 24 x 1018 e. V

§What is the energy equivalence of an electron -31 (mass = 9. 109 x 10 kg), as measured in joules and electron volts?
§What is the mass equivalence of a 90 ke. V x-ray?

§What is the mass equivalence of a 340 nm photon of UV light?
- Slides: 30