Prepared by Dental Materials Department Yenepoya Dental College

Prepared by: Dental Materials Department Yenepoya Dental College Yenepoya University, Mangalore.

Dental Applications n Gypsum products are used mainly for making positive reproductions or replicas of oral structures. – Casts, dies, or models – Other uses: impression, mounting, part of investment 2

n Casts – Study cast – Working cast Die materials n Binder for silica investments n 3

4
5
Desirable properties (1) Accuracy (2) Dimensional stability (3) Ability to reproduce fine detail (4) Strength and resistance to abrasion (5) Compatibility with the impression material (6) Color (7) Biological safety (8) Ease of use 6

Manufacture Chemical and Physical properties

n Mining n For dental purposes in form of “Calcium sulfate dihydrate” (Ca. SO 4. 2 H 2 O) Gypsum is a common name of calcium sulfate dihydrate. 8

Dental gypsum products Primary constituent = Calcium sulfate hemihydrate, Ca. SO 4·½H 2 O n From a process called “Calcination” n n Calcination: heating process performed by the manufacturer to create the hemihydrate powder form of the product. (Plaster or Stone) (Gypsum) Ca. SO 4· 2 H 2 O 110 -130 o. C Ca. SO 4·½H 2 O + H 2 O “Calcination” Dehydration 9
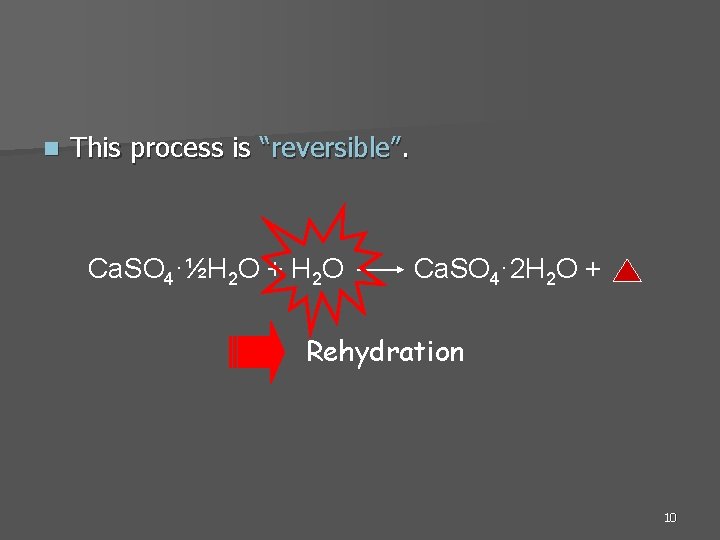
n This process is “reversible”. Ca. SO 4·½H 2 O + H 2 O Ca. SO 4· 2 H 2 O + Rehydration 10

Types of Dental Gypsum n Plaster n Stone n High-Strength or Improved Stone n Other types of gypsum: impression plaster, mounting plaster, . . etc 11

Plaster n b-hemihydrate n n n Manufactured by grinding the gypsum rock to a fine powder and then heating the powder in an open container eliminate water & shatter the crystal Porous, irregular powder particles Sometimes is referred to as bhemihydrate or Type II stone. Weakest and least expensive Usually white in color. Used mainly where strength is not a critical requirement e. g. a study model, model base Plaster + chemicals impression plaster (Type I). 12

Stone Made from gypsum by carefully controlled calcination under steam pressure in a closed container. n Powder particle is more regular, more uniform in shape, and less porous n Is often referred to as ahemihydrate, Type III stone, or Hydrocal. n Various colors. Used mainly for making casts for diagnostic purposes and casts used for complete and partial denture construction n a-hemihydrate 13
High-Strength or Improved Stone n n n Made from gypsum by calcining the gypsum in a calcium chloride solution. Very dense powder particle in cuboidal shape with a reduced surface area Strongest and most expensive Often referred to as Type IV stone, die stone, densite, and modified ahemihydrate Used mainly for making casts or dies for inlay and crown fabrication A newly developed high-strength stone with a higher compressive strength and higher setting expansion Type V stone. 14

Forms of hemihydrate n b-hemihydrate (plaster of Paris) – Sponginess, porous, irregular n a-hemihydrate (dental stone) – Well-formed crystals, regular, more dense 15

Particles pack better. 16

Setting Reaction

Something to remember! n All gypsum products set by the same reaction. (Ca. SO 4)2·½H 2 O + 3 H 2 O 2 Ca. SO 4· 2 H 2 O + Heat After calcination process, some calcium sulfate dihydrate remain in the powder. n Both hemi- and dihydrate are water soluble but the hemihydrate is four times more soluble. n 18

Hemihydrate + water suspension (fluid and workable) 1 2 3

Dihydrate (supersaturated) Dihydrate precipitate out in form of needle-like crystals. (some calcium sulfate dihydrate remaining in the powder are nuclei for precipitation. ) Soln no longer sat. with hemihydrate • ↑dissolution & precipitation or • ↑crystal growth

Hemihydrate + water suspension (fluid and workable) ↓ Dihydrate (supersaturated) ↓ Dihydrate precipitate out in form of needle-like crystals. ↓ Soln no longer sat. with hemihydrate • • ↑dissolution & precipitation or ↑crystal growth proceeds until all hemihydrate is consumed or other factors interfere or insufficient water is present.

n Formation of needle like crystals Mass thickens 22

Water/Powder ratio

n Theoretically, if 100 g of any hemihydrate were combined with 19 ml of water, all the hemihydrate would be converted to the dihydrate. (19 ml = sufficient water for 100 g hemihydrate to react) n However in practice, this amount of water will not produce a mass that can be manipulated and poured into an impression. (too dry and crumbly mix) Therefore, in practice, excess water must be added when one is mixing to produce a workable mass. 24

n Various gypsum products require different amount of water to mix. – a-hemihydrate requires less water than b-hemihydrate. § b-hemihydrate => Very porous, irregular, do not pack together very well 25

n Depends on the physical characteristics of the powder particles – Plaster (Type II) 45 -50 ml/100 g (0. 45 -0. 50) – Stone (Type III) 28 -30 ml/100 g (0. 28 -0. 30) – Improved stone (Type IV) 19 -24 ml/100 g (0. 19 -0. 24) different consistencies of a workable mix 26

** The excess water does not react but is simply trapped in the mass when it sets. porosities or voids in the mass n Set plaster has the lowest density (most porous) because it had the most excess water and the most voids in the mass. n Set high-strength stone has the highest density. Excess water Porosity 27

The water/powder ratio has a direct effect on the properties of each gypsum product and must be controlled for optimum results. n Higher W: P ratio (Thinner mix) n § § Longer setting time Weaker product (more free water more porosities when set) Lower hardness Reduce expansion (in the same type of gypsum) § § Broader crystals stronger and more solid mass Shorter setting time Stronger product Too low W: P ratio may be difficult to pour and may create some voids since the mass can not flow well. Lower W: P ratio (Thicker mix) 28

Increasing the water-powder ratio results in a more “dilute” structure, with greater spaces between crystals. 29
Setting Time Types of setting time Factors to control setting time

n Working or Initial Setting Time – Represents the available time for manipulating – Indicates partial progress of the setting reaction – “Loss of gloss” – Vicat needle does not completely penetrat. – 8 to 16 minutes Start mixing n Final Setting Time – Represents the time until the setting mass becomes rigid and can be separated from the mold – Indicates the major completion of the hydration reaction – Failure of penetration of a fingernail or knife. Dissipation of the heat of reaction – 30 to 45 minutes Material starts to set. Material sets. Mixing time Working time /Initial setting time (8 -16 min) Final setting time 31

Control of the Setting Time n Controlled by the manufacturer’s particular formulation n 3 methods – Control solubility of the hemihydrate – Control number of nuclei for crystallization – Control rate of crystal growth 32

n n n Gypsum particles remain after calcination nuclei for dihydrate to precipitate shorter setting time Finer particle size of the hemihydrate shorter setting time (particles: plaster<stone<improved stone) Increase W: P ratio prolong setting Mixing: Longer and more rapidly spatulate shorter setting time Temperature: erratic (25°C-37°C may accelerate. , > 50°C - retardation, 100°C - no reaction) Retarders and Accelerators 33

n Increase setting time – Decreased mixing – Higher W/P ratio (thinner mix) – Addition of retarder n Decrease setting time – Increased mixing – Lower W/P ratio (thicker mix) – Addition of accelerator § Borax 34
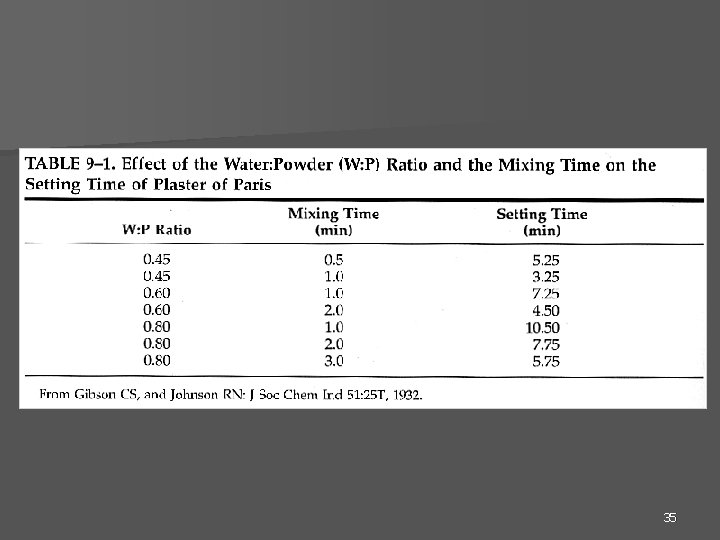
35

W: P ratio mixing time setting time 36

Accelerators and Retarders n The addition of an accelerator produces a set that makes it possible to use the plaster within 30 min. but the working time has been seriously reduced. n The addition of retarder makes the mix retain a reasonable plastic state that permits handling or working it into a useful shape. 37

38

Accelerators n Increase the solubility of the hemihydrate without increasing the solubility of the dihydrate inorganic salts – Sodium Chloride up to 2% conc. , Potassium sulfate > 2% conc. , Sodium sulfate up to 3. 4% conc. n Powdered gypsum (Ca. SO 4. H 2 O) or slurry of ground gypsum casts – Increase nuclei of crystallization n Increase spatulation (time and speed) – Increase the formation of more nuclei 39

Retarders n Certain chemicals form a coating on the hemihydrate particles and prevent them from going into solution in the normal manner. – Citrates, acetates, borates n Colloid particles such as blood, saliva, or unset alginate will retard the setting reaction. (they bind to the hemihydrate and interfere with the addition of water to form the dihydrate) soft, easily abraded surface Rinse impression thoroughly before pouring 40

Moisture contamination during storage ? 41

Setting Expansion

n All products expand upon setting. – Plaster 0. 2 -0. 3% > Stone 0. 08 -0. 10% > Improved stone 0. 05 -0. 07% HOW? The growing crystals of the gypsum cause an outward crystal thrust an external expansion + internal porosity in the set mass. 43

Control of Setting Expansion can be advantage or disadvantage. In general, minimal setting expansion is desirable. accurate dimensional reproduction n Less W: P ratio and/or Longer mixing time (within practical limit) ↑expansion n Higher W: P ratio fewer nuclei/unit volume ↑space between nuclei less growth interaction of the dihydrate crystals and less outward thrust – (in the same type of stone) n The most effective method addition of chemical (controlled by manufacturer) 44
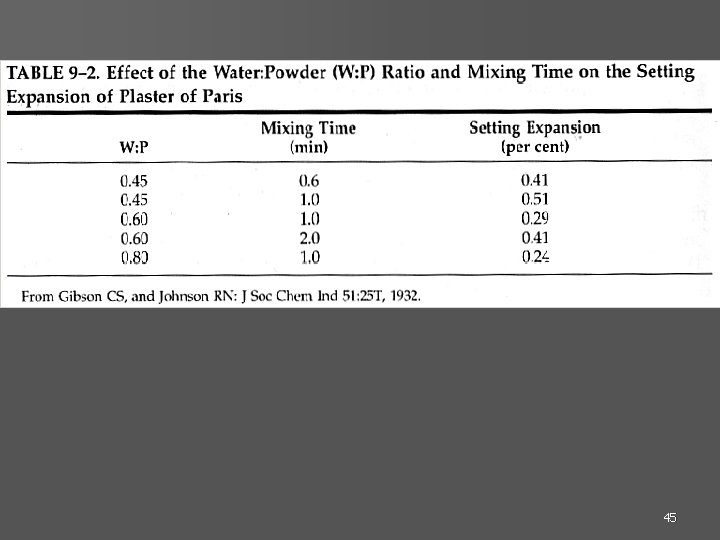
45
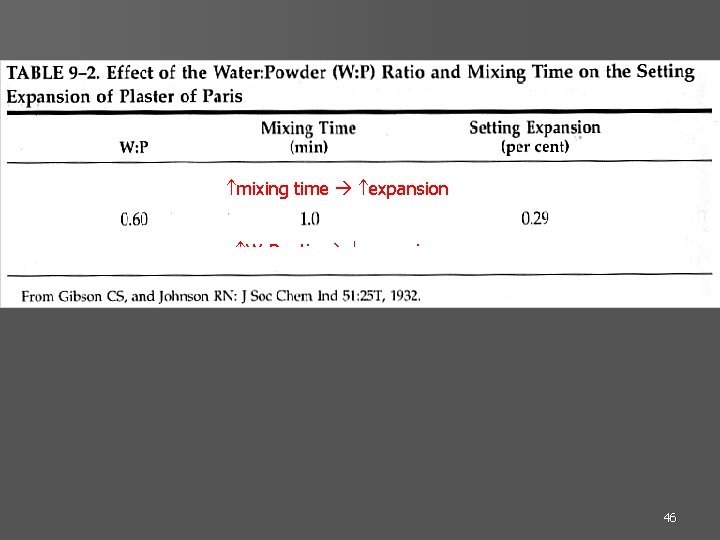
mixing time expansion W: P ratio expansion 46

The expansion characteristics should not be altered by manipulation. 47

What will happen if the setting process occurs under water? ↑↑Expansion because additional crystal growth is permitted. Hygroscopic Setting Expansion

Hygroscopic Setting Expansion n n Stage I: initial mix Stage II: crystals of dihydrate are formed – Lt: water around the particles is reduced by the hydration and the particles are drawn more closely together by the surface tension action of the water – Rt: hydrated water is replaced n Stage III: crystals grow n Stage IV and V – Lt: water is decreased, particles are drawn together – Rt: water is replaced, crystals grow freely – Lt: water is used up, crystals stop growing – Rt: water is replaced. 50
Reproduction of Detail

n Reproduction of detail of an impression depends on the compatibility and interactions between the model material and the impression material. n The ability of the material to flow into and register finer detail and finer lines is used to rate its detail of reproduction. n Factors: Compatibility + Wettability between gypsum and impression materials 52
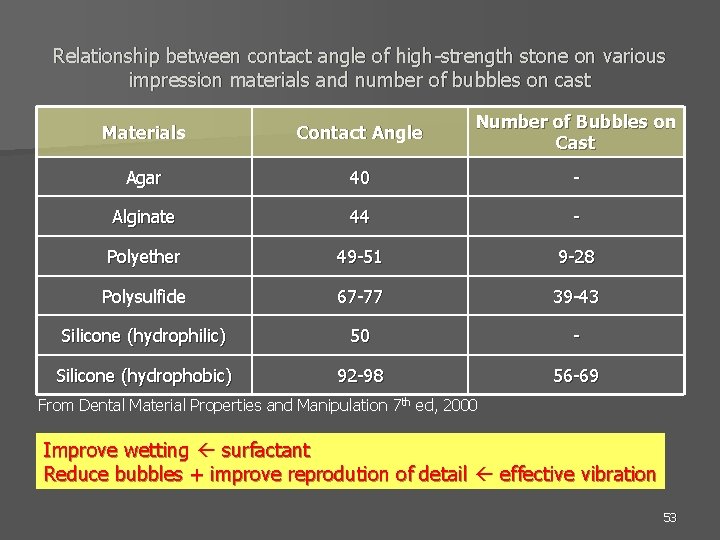
Relationship between contact angle of high-strength stone on various impression materials and number of bubbles on cast Materials Contact Angle Number of Bubbles on Cast Agar 40 - Alginate 44 - Polyether 49 -51 9 -28 Polysulfide 67 -77 39 -43 Silicone (hydrophilic) 50 - Silicone (hydrophobic) 92 -98 56 -69 From Dental Material Properties and Manipulation 7 th ed, 2000 Improve wetting surfactant Reduce bubbles + improve reprodution of detail effective vibration 53

Strength and Surface Hardness

n Strength and hardness develop rapidly during the first 30 to 45 minutes as the hydration is completed. n Hardening solution used in place of water reduces the amount of excess water needed to wet the hemihydrate particles while gives a workable consistency to the mix. – Hardening solution: water+colloidal silica+other chemical modifiers 55
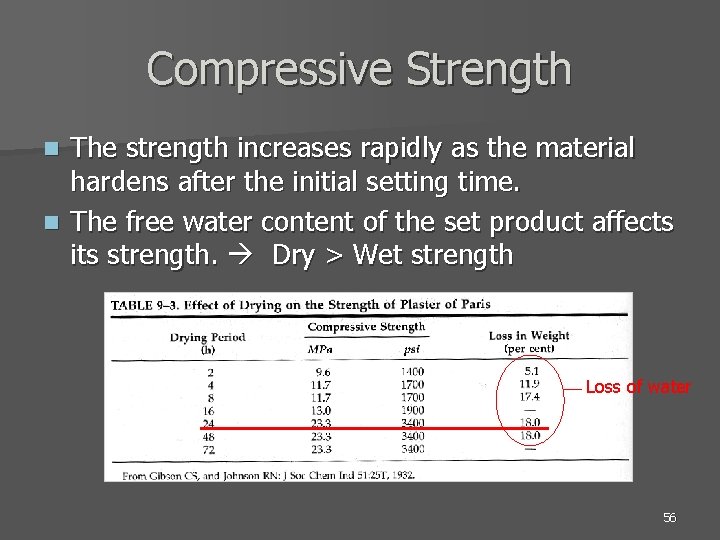
Compressive Strength The strength increases rapidly as the material hardens after the initial setting time. n The free water content of the set product affects its strength. Dry > Wet strength n Loss of water 56

n n The set plaster or stone is porous in nature. The greater W: P ratio greater porosity less dry strength ↑Mixing time ↑strength Accelerators&retarders ↓strength Mixing time strength W: P ratio strength 57

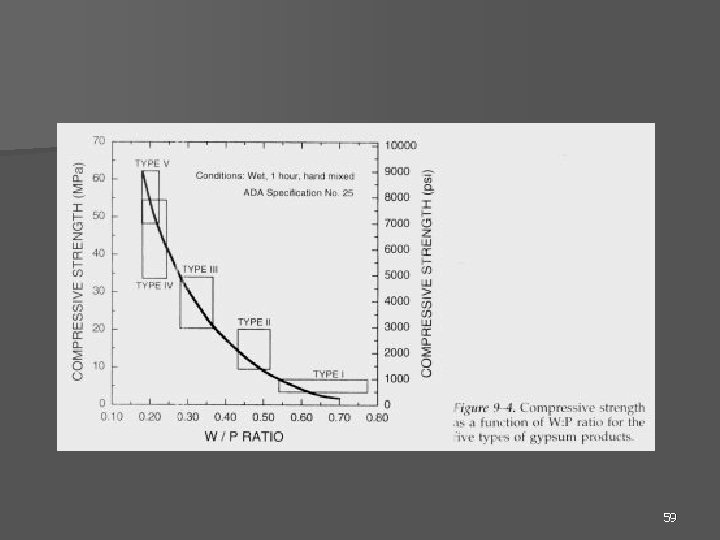
n n The set plaster or stone is porous in nature. The greater W: P ratio greater porosity less dry strength ↑Mixing time ↑strength ***** Accelerators&retarders ↓strength 58
59

Tensile Strength n Important when separating dies from an impression. n From diametral tensile tests, in general: – 1 hr strength is 1/2 the dry strength. – Plaster’s tensile strength is 1/2 that of die stone. – Tensile strengths are usually 1/5 the compressive strength. 60
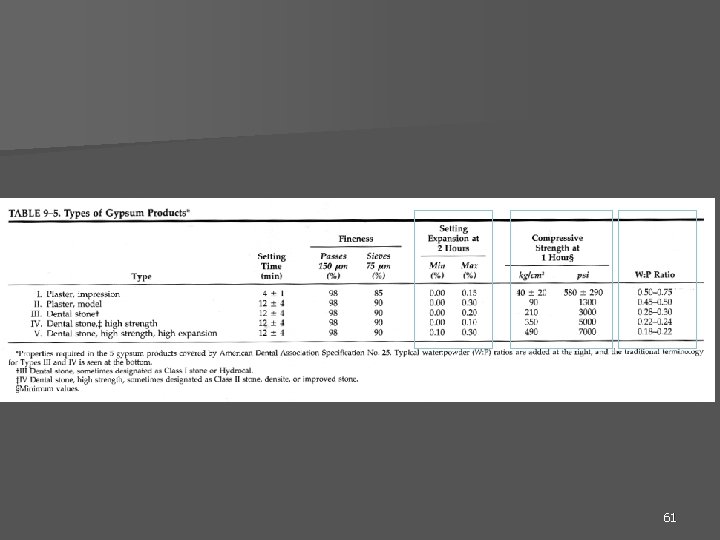
61

Proportioning, Mixing, Pouring and Caring
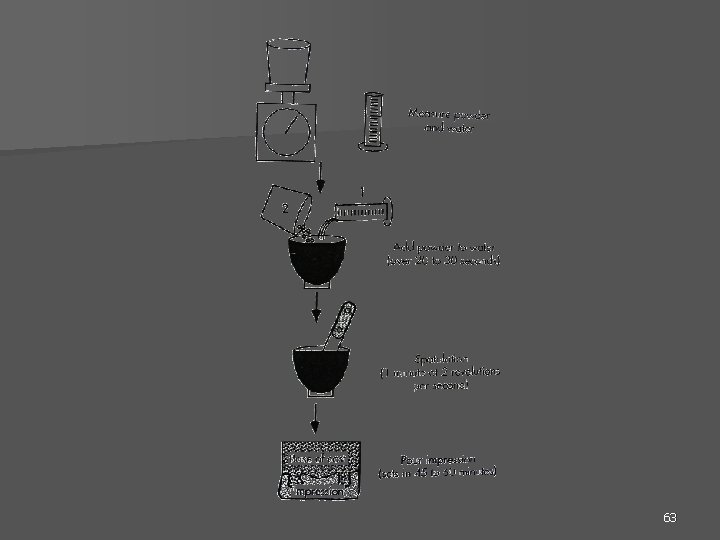
63

Proportioning n Use the manufacturer’s suggested W: P as a guide n The water and powder should be measured. 64

Mixing The preferred method of mixing is to add the measured water first, followed by gradual addition of the preweighed powder. n The use of an automatic vibrator, of high frequency and of low amplitude, is helpful. n The guesswork of repeatedly adding water and powder to achieve the proper consistency must be avoided. n 65

Pouring the Model The mixed gypsum is placed into the impression in increments, with use of vibration to enhance the flow of the material into the impression. n Allow the material to flow across the impression, with avoidance of entrapment of air. n • Alternatively, the teeth and soft tissues may be poured in stone or die stone and allowed to set, and the base can be made from plaster later. • The impression should not be removed from the gypsum for 45 to 60 minutes to allow the final set to occur. 66

Disinfection n Models, casts and dies may be disinfected with a spray of iodophor or by immersion in 1: 10 dilution of a 5% sodium hypochlorite solution. n ** preferable to disinfect the impression rather than the model** 67

Caring for the Cast Avoid abrasion of teeth and related structures in the cast. n If the stone cast is immersed in running water, its linear dimension may decrease approximately 0. 1% for every 20 minutes of such immersion. n The safest method for soaking the cast is to place it in a water bath made for the purpose, in which plaster debris is allowed to remain constantly on the bottom of the container to provide a saturated solution of calcium sulfate. n 68

Special Products n Mounting stones or plasters – Fast setting and have low setting expansion – Low strength n Modified stone – Add a small amount of plastic or resin to reduces the brittleness and improves the resistance to scratching 69

Summary n n n n n Desirable Properties Types of gypsum: Plaster, Stone, Improved stone Setting Reaction W: P Ratio Setting Time Setting Expansion, Hygroscopic Expansion Reproduction of Detail Strength and Hardness Factors to control setting time, expansion, strength Manipulation and Caring 70
- Slides: 69