Preparation of MidiScale Plasmid DNA from E coli








- Slides: 15

Preparation of Midi-Scale Plasmid DNA from E. coli (midi-scale plasmid DNA) 생화학 실험 (2) 1주차 담당교수 : 송재환 교수님 담당조교 : 오대석, 이민식

Contents 1. Plasmid DNA for gene cloning 2. Components of plasmid DNA 3. Plasmid purification methods 4. Preparation of Plasmid DNA 5. Identification of purified plasmid DNA

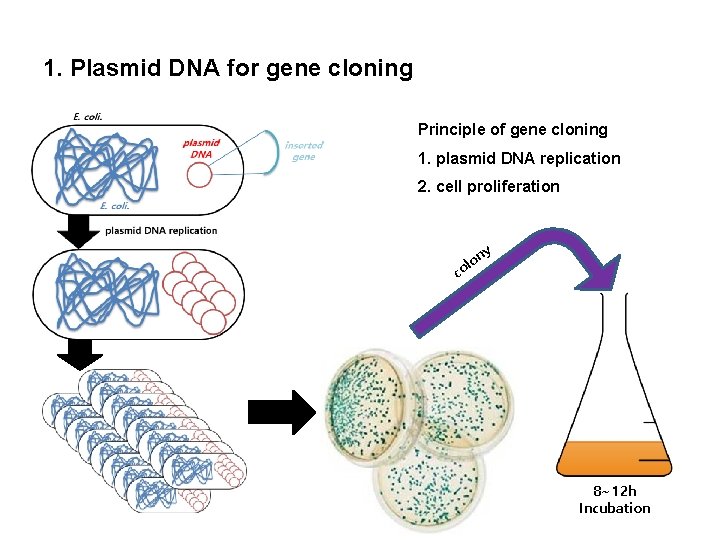
1. Plasmid DNA for gene cloning Principle of gene cloning 1. plasmid DNA replication 2. cell proliferation ny c o ol 8~12 h Incubation

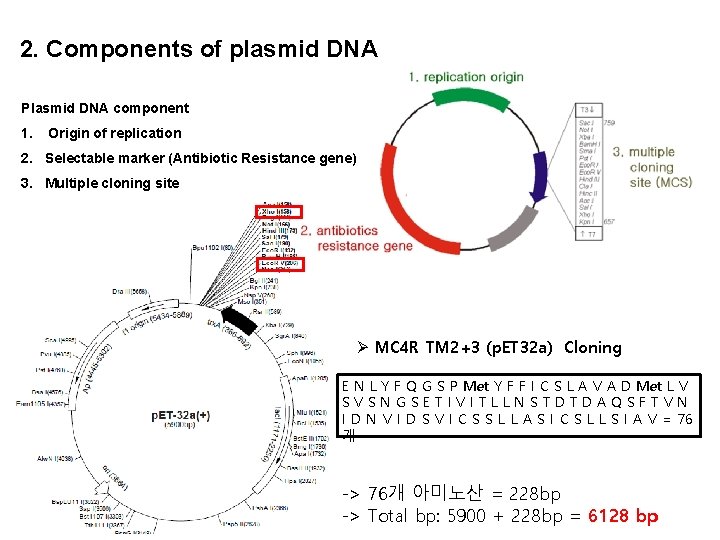
2. Components of plasmid DNA Plasmid DNA component 1. Origin of replication 2. Selectable marker (Antibiotic Resistance gene) 3. Multiple cloning site Ø MC 4 R TM 2+3 (p. ET 32 a) Cloning E N L Y F Q G S P Met Y F F I C S L A V A D Met L V SVSNGSETIVITLLNSTDTDAQSFTVN I D N V I D S V I C S S L L A S I C S L L S I A V = 76 개 -> 76개 아미노산 = 228 bp -> Total bp: 5900 + 228 bp = 6128 bp

3. Plasmid purification methods 1. Alkaline lysis Method - Cell lysis 2. Anion-Exchange Chromatography Method - DNA separation Types of preperation Mini Midi Maxi Mega Giga DNA yield(ug) 100 ug> 100 -350 500 -850 1500 -2500 7500 -10000 purification scale


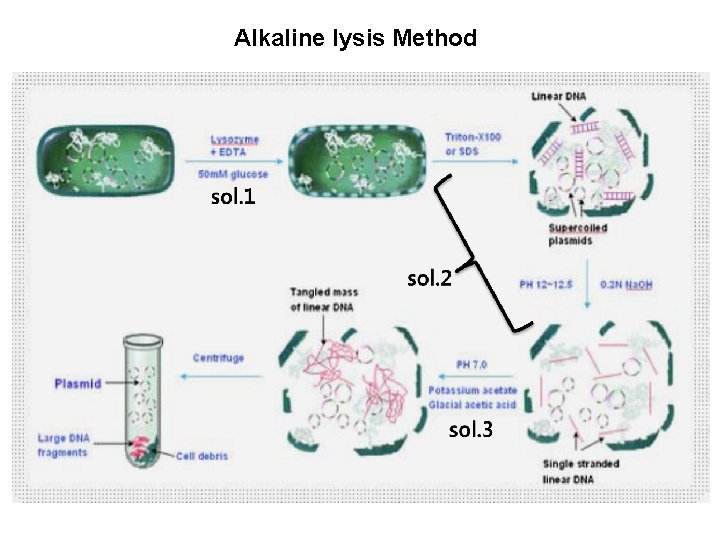
Alkaline lysis Method

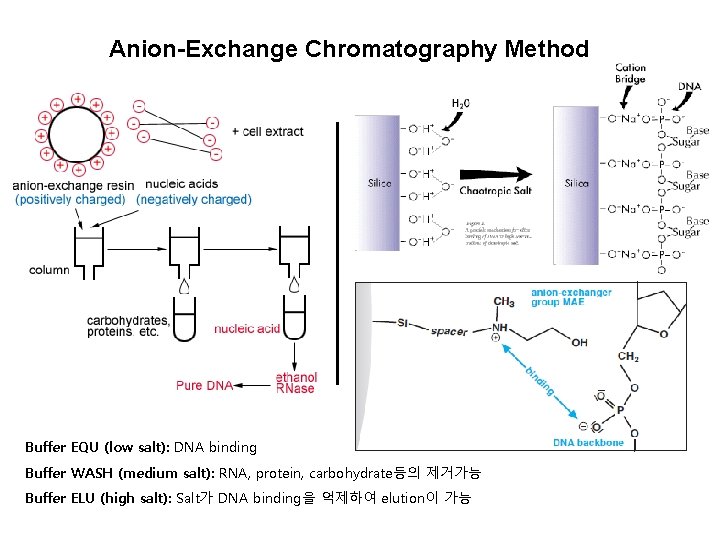
Anion-Exchange Chromatography Method Buffer EQU (low salt): DNA binding Buffer WASH (medium salt): RNA, protein, carbohydrate등의 제거가능 Buffer ELU (high salt): Salt가 DNA binding을 억제하여 elution이 가능

4. Preparation of Plasmid DNA

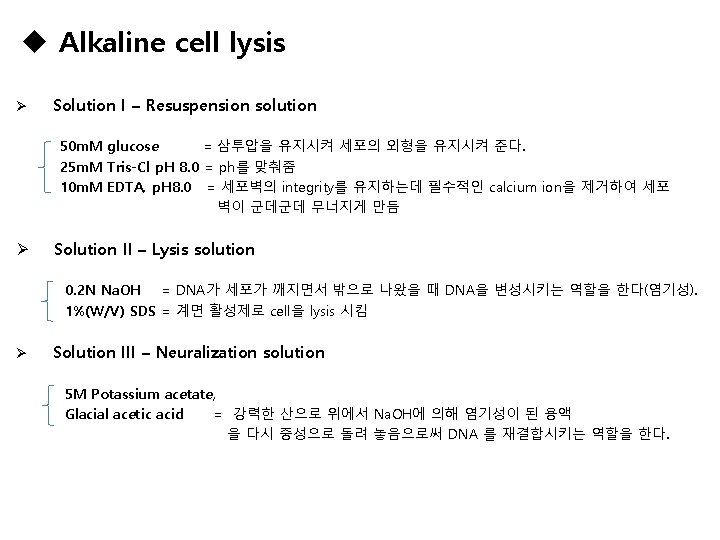
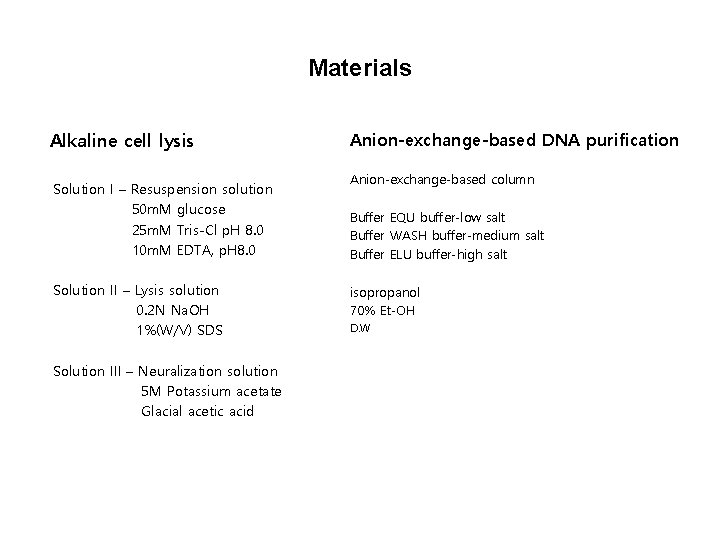
Materials Alkaline cell lysis Solution I – Resuspension solution 50 m. M glucose 25 m. M Tris-Cl p. H 8. 0 10 m. M EDTA, p. H 8. 0 Solution II – Lysis solution 0. 2 N Na. OH 1%(W/V) SDS Solution III – Neuralization solution 5 M Potassium acetate Glacial acetic acid Anion-exchange-based DNA purification Anion-exchange-based column Buffer EQU buffer-low salt Buffer WASH buffer-medium salt Buffer ELU buffer-high salt isopropanol 70% Et-OH D. W

Alkaline cell lysis method • Procedures 1) Resuspension the pellet in 3 ml of Alkaline lysis solution I (Buffer RES) “ Vol. [ m. L ] = Culture Volume [ m. L ] x OD 600 / 50” - Culture volume: 100 ml , O. D 600: 1. 5 => 3 ml 2) Add 3 ml solution II (Buffer LYS) - Mix the contents thoroghly by inverting the tube several times(5 times). - Incubate the mixture at room temperature (18– 25 °C) for 5 min. 3) Column equilibration (Buffer EQU) 12 ml 4) Add 3 ml solution III (Buffer NEU) - inverting the tube several times(10 ~15 times) 5) Inverting the tube 3 times directly before applying the lysate to the equilibrate Nucleo. Bond® Xtra Column Filter 6) Wash column filter and column (Buffer EQU) 5 ml 7) Discard column filter

Anion-exchange chromatography method 7) Add 8 ml of Buffer WASH into column and allow the column to empty by gravity flow 8) Elution - Add 3 ml (Buffer ELU) 9) Add 3 ml isopropanol 10) Centrifugate for 30 min at 13000 rpm at 4˚C in a microcentrifuge 11) Carefully decant the supernatant. 12) Wash with 2 ml of 70% Et-OH 13) Centrifugate for 5 min at 13000 rpm, RT in a microcentrifuge 14) Carefully decant the 70% Et-OH 15) Air dry 5 min and redissolve the DNA in 400 ul D. W.

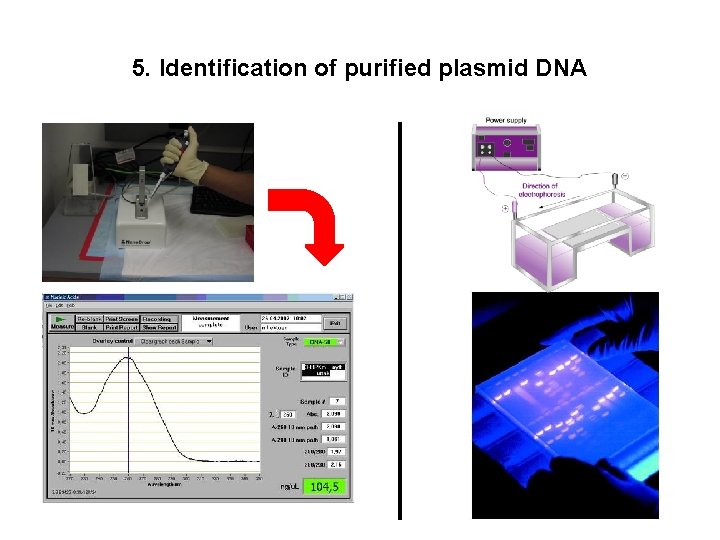
5. Identification of purified plasmid DNA

