Preparation of Chemical Compound in Laboratory Scale SMK

Preparation of Chemical Compound in Laboratory Scale SMK Negeri 13 Bandung

Objectives After this session, the students are expected to be able to : - classify the apparatus of separation - Conduct the separation and isolation - Explain the steps of separation - arrange the planning of preparation of chemical compound according to the job sheet - explain the objectives of every step - explain the function of reagents - conduct the test of physical and chemical properties of the chemical compound - do the calculation in the determination of nickel - arrange the report of the preparation of chemical compound

Separation v Distillation - Simple distillation - Vacuum distillation - Fractional distillation - Steam distillation v Extraction - Soxhlet - separation funnel v Crystallization Teknologi dan Rekayasa

Simple distillation apparatus 1: 2: 3: 4: Heat source Still pot Still head Thermometer/Boiling point temperature 5: Condenser 6: Cooling water in 7: Cooling water out 8: Distillate/receiving flask 9: Vacuum/gas inlet 10: Still receiver 11: Heat control 12: Stirrer speed control 13: Stirrer/heat plate 14: Heating (Oil/sand) bath 15: Stirrer bar/anti-bumping granules 16: Cooling bath. http//1. bp. blogspot. com

Vacuum Distillation Apparatus chem-is-try. org

Extraction Apparatus http: //www. aquaculture. ugent. be/
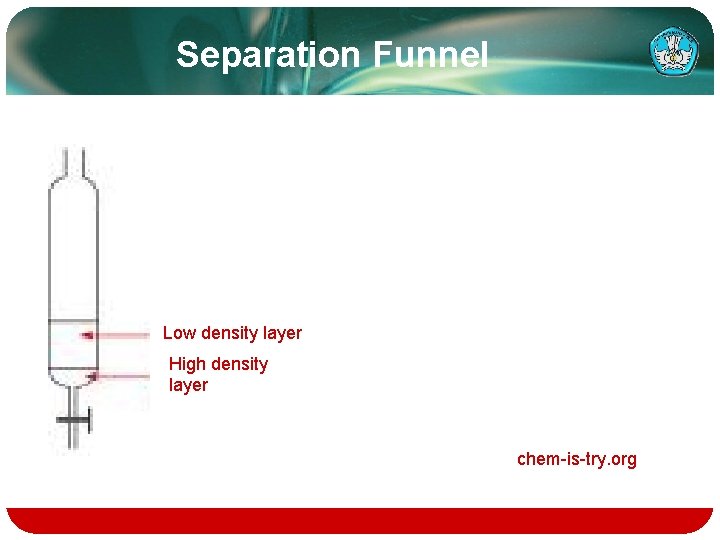
Separation Funnel Low density layer High density layer chem-is-try. org

Preparation of Cyclohexene v Weigh out 20 m. L of cyclohexanol in to a round bottom flask of 200 m. L v Add 2 m. L of concentrated sulfuric acid and 2 or 3 boiling stones v Put a fractional colum and a reflux condenser on the top of the flask v Heat the flask in a heating mantle or oil bath. Set the temperature not more than 1300 C in wise the distillation conduct at 950 C

Procedure…. . v Continue the distillation process until the residue produces white steam v Move the distillate in to a separation funnel and let the two layers separated v Remove the water layer v Wash the organic layer with 10 m. L of water, then with 10 m. L of 10% Na 2 CO 3 and then with 10 m. L of water

procedure v Pour the hydrocarbon layer through the top of the funnel in to a clean and dry Erlenmeyer flask v Add 3 -4 gram of dry anhidrous Ca. Cl 2 shake for 2 -3 minute. Let it for 15 minute with sporadic shaking v Pour the dried hydrocarbon in to a round flask of 50 m. L and put some boiling stone

procedure v Conduct the distillation at 80 – 900 C v Collect the fractional part of 80 -850 C

Testing Reaction v Prepare 3 test tube and fill each with - 2 m. L of % brom in CCl 4 - 2 m. L of 1% KMn. O 4 - 2 m. L of concentrated H 2 SO 4 v Add to each test tube 1 -2 drops of distillate v Watch the reaction and write

Physical Properties Test v Purpose To identify the purity of product, it must be carried out the physical properties test and refer to handbook v Phase of matter - Gas - Liquid - Solid

Physical Properties of Solid v Melting point v Crystal shape Teknologi dan Rekayasa

Physical Properties of Solid Crystal Shape NH 4 Cl and Na. Cl

Melting Point Apparatus http: //laborimpex. com/images/Barlo%20 me lting%20 point%20 apa%20 smp 11. JPG Teknologi dan Rekayasa

Melting Point Apparatus http: //www. chemistry. mcmaster. ca/~chem 2 o 6/labmanual/expt 1/exp 1 -f 7. gif Teknologi dan Rekayasa

Melting Point Determination With your spatula, grind the sample into fine powder

Poke the inverted capillary into the powder, trapping some sample in the open end. v Repeat this step, if necessary to obtain enough sample to see.

v Pack the sample by bouncing it down your condenser several times. v. . . or you can hold the capillary with your thumb and forefinger, and RAP it sharply on the bench top; - but this could hurt!!

Insert the capillary into one of the 3 slots, and select a desired setting, -- say about 5. v View the sample through the observation window and watch as it melts.

It should melt something like THIS: only MUCH, MUCH SLOWER!!

v. Record BOTH temperature readings (a melting point RANGE) RANGE v. Where it STARTS (when you see the first liquid run). v. Where it ENDS (when the last bit of solid becomes liquid).

Physical Properties of Liquid Boiling Point Freezing Point Refractive index Density

Refractometer / http: //suninstruments. com Teknologi dan Rekayasa

Densitometer http: //media-2. web. britannica. com/ebmedia/84/124884 -004 -530100 D 8. jpg http: //www. glassthermometers. com/productsimages/densito meter_147655. jpg Teknologi dan Rekayasa

Densitometer http: //www. watthoursystems. com/gifs/densi http: //chestofbooks. com/crafts/scientificamerican/sup 6/images/Devicetometer 1. jpg Illustration. png Teknologi dan Rekayasa

Picnometer Hole Cap Measuring flask http: //2. bp. blogspot. com/ http: //anm. co. id/images/ Teknologi dan Rekayasa

Refractometer Portable Refractometer www. arssales. com/e Digital Handled Refractometer img. alibaba. com/ Teknologi dan Rekayasa
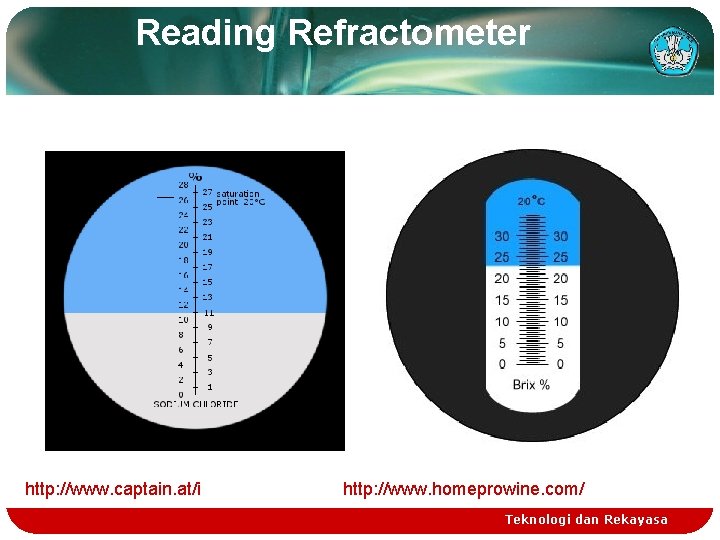
Reading Refractometer http: //www. captain. at/i http: //www. homeprowine. com/ Teknologi dan Rekayasa

Freezing Point Apparatus http: //intro. chem. okstate. edu/ http: //usm. maine. edu/ Teknologi dan Rekayasa

Freezing Point Apparatus http: //www. koehlerinstrument. com/images/ kicproductsbig/K 29750. jpg http: //www. petroleumlogistics. com/images/ ph_11. jpg Teknologi dan Rekayasa

Boiling Point Apparatus www. sciencemuseum. org. uk http: //www. pharmech. com/ Teknologi dan Rekayasa

Apparatus for Physical Properties Determination v Density - Picnometer - Areometer v Boiling Point - Distillation Apparatus - Siwoloboff Apparatus - BP meter v Melting Point - Melting Block - Thiele Apparatus

Apparatus for Physical Properties Determination v Refractive Index - Refractometer v Freezing Point - FP meter
- Slides: 35