PRELUDE n Most abundant class of organic compounds


PRELUDE n Most abundant class of organic compounds found in living organism. nn CO 2 + n H 2 O + energy Cn. H 2 n. On + n O 2 They fill numerous roles in livings, such as storage , transport of energy & stuctural components, immune system, fertilisation. . n Classification n monosaccharides ex- glucose, galactose, frutose. n simple disaccharides ex- sucrose, lactose, maltose.
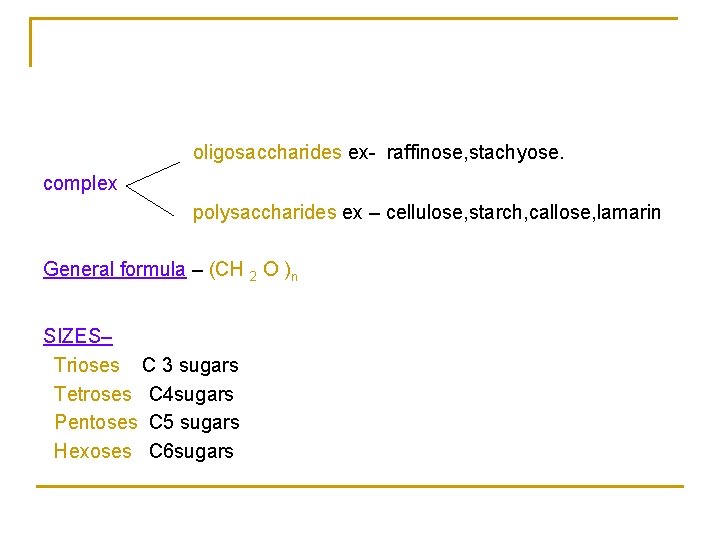
oligosaccharides ex- raffinose, stachyose. complex polysaccharides ex – cellulose, starch, callose, lamarin General formula – (CH 2 O )n SIZES– Trioses C 3 sugars Tetroses C 4 sugars Pentoses C 5 sugars Hexoses C 6 sugars
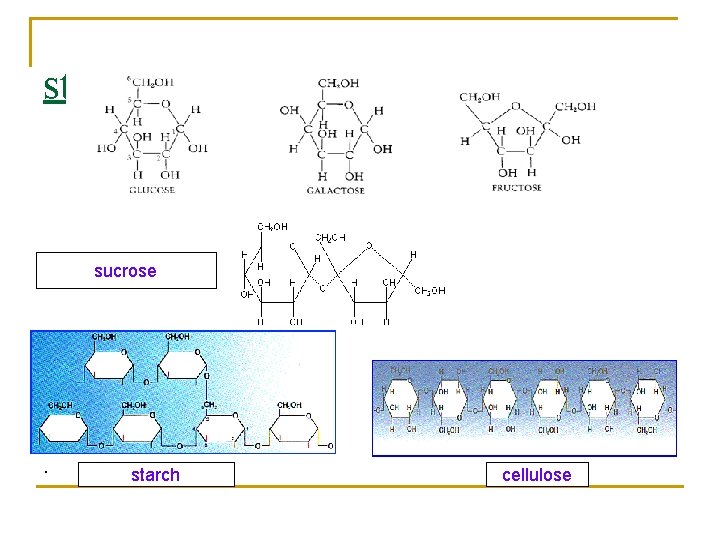
stuctures sucrose . starch cellulose

Evaluation methods n Chemical methods n Titrimetrical method n Gravimetrical method n Enzymatic method n Physical methods n Spectrophotometry methods n Chromatography methods n miscellaneous

Chemical methods n Fehling’s test n Benedict’s test n Barfoed’s test n Molisch’s test n Bial’s test n Seliwanoff’s test n Iodine test

Fehling’s test

Barfoed’s test
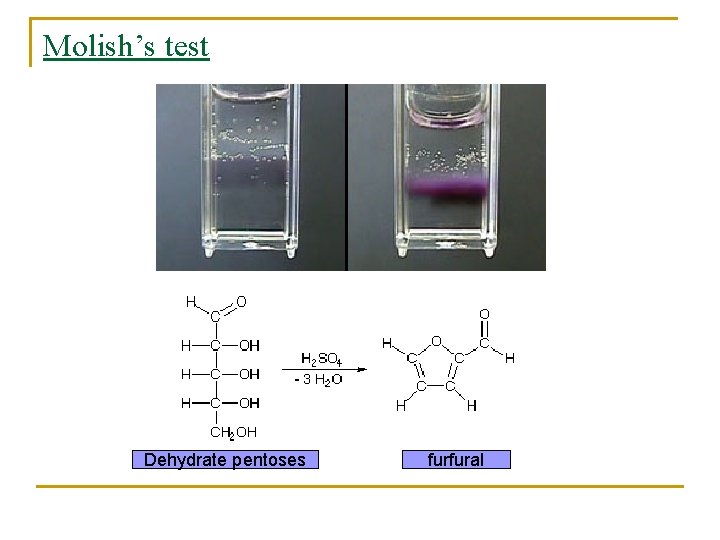
Molish’s test Dehydrate pentoses furfural
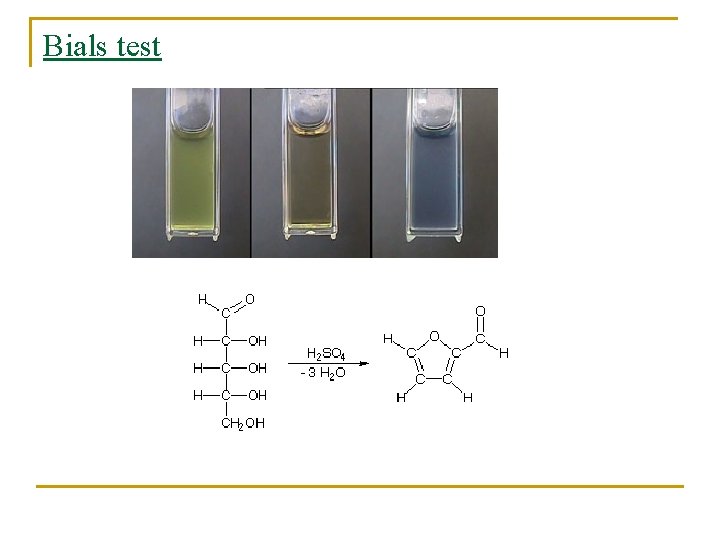
Bials test
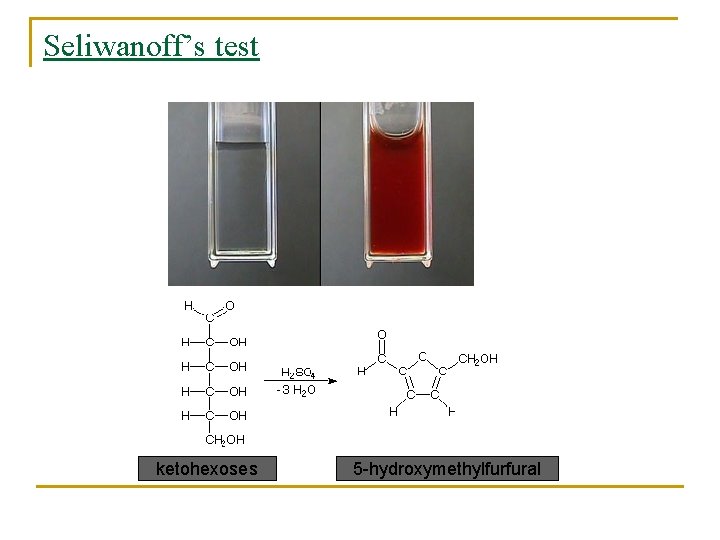
Seliwanoff’s test ketohexoses 5 -hydroxymethylfurfural

Benedict’s test

Iodine test
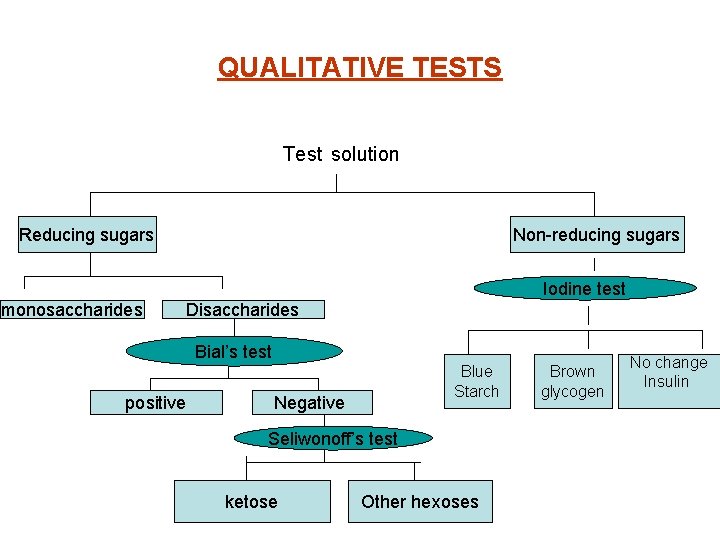
QUALITATIVE TESTS Test solution Reducing sugars Non-reducing sugars Iodine test monosaccharides Disaccharides Bial’s test positive Blue Starch Negative Seliwonoff’s test ketose Other hexoses Brown glycogen No change Insulin

TITRIMETRY METHOD n EDTA has been used in place of fehling’s & benedict’s reagents to prevent precipitation of cupric ions in alkaline solution. n Cupric ions form a relatively stable chelation complex with EDTA. n Ferrous ions are detremined by permanganate titrimetry. n Similar approach applied by HAGEDORN-JENSEN PROCEDURE—reaction of reducing carbohydrates with ferricyanide.

Continution…. n Aldehyde function can be deternined by selective oxidation with hypoiodous acid. n To detremine aldoses– cyanohydrin reaction is done. n To determine polysaccharides– neutralisation of negatively charged colloidal particles with positively charged ones & relies on metachromatic dyes.

Limitations. n Results depend on the precise reaction times. temp & reagent concentration. n It cannot distinguish b/n different types of reducing sugars. n It cannot directly determine the concentration of non reducing sugars. n It is susceptable to interference from other types of molecules that act as reducing agents.

Gravimetric method n The Menson & walker method –determine the conc of reducing sugars in a sample. n Carbohydrates are oxidised & an excess copper sulfate & alkaline tartarate leads to formation of cuprous oxide ppt n The amount is proportional to conc in the sample. n n Merits Accurate n More reproducible.

Enzymatic method n Analytical method based on enzyme rely on their ability to catalyse specific reaction. n Rapid , highly specific & sensitive to low conc. n Two methods--- n 1. Allowing the reaction to go to completion & measure concentration of the product. n 2. measuring the intial rate of the enzyme catalysed reaction.


Polarimetry n Molecules that contain asymmetric carbon atom have the ability to rotate the plane polarised light. n The extent of polarisation is related to concentration of the optically active molecules in sol by equation a= [a]/c where a=measured angle of rotation a= [a]lc [a] = optical activity l= pathlength , c =589. 3 nm. n n The conc of unknown is determined by measuring its angle of of rotation.

Refractive index n= c/cm Determined by angle of incidence & angle of refraction at a boundary b/n angle & another material of known RI. n n Snell’s law = sin(i)sin(r) = n 2/n 1 n Measurements are made at specific temp (20) & λ (589. 3 nm). n Quick & simple n Determine sugar conc of syrups, honey, molases, tomato products & jams.

Density n d= m/v The density of aq solution increases as the concentration increases. n Thus the carbohydrate concentration can be determined by measuring density. n Routinely used in industry for determination of carbohydrates concentration of juices & beverages. n
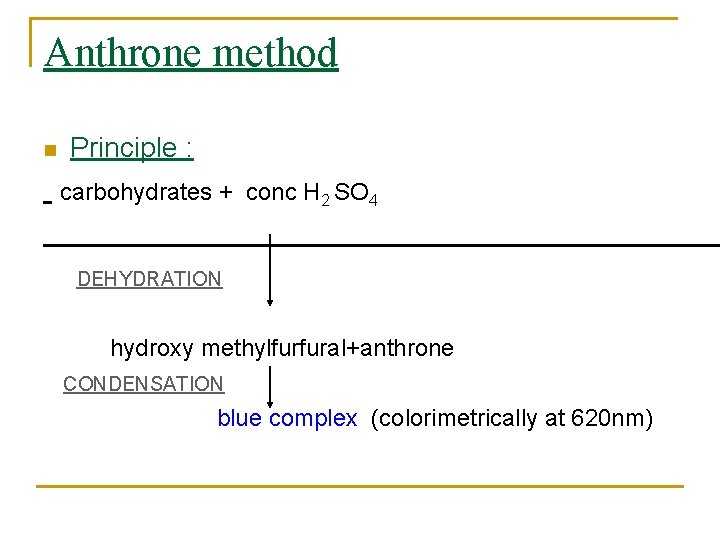
Anthrone method n Principle : carbohydrates + conc H 2 SO 4 DEHYDRATION hydroxy methylfurfural+anthrone CONDENSATION blue complex (colorimetrically at 620 nm)

Procedure n n 1. Anthrone reagent(0. 2 % in conc H 2 SO 4). 2. std glucose solution (10 mg/100 ml) diff volumes of glucose sol make up to 1 ml(water) 4 ml of anthrone reagent –mix well water bath-cool measure optical density at 620 nm

Benedict’s method This method is value in clinical analysis of glucose in blood & urine. 1. Benedicts sol+ sodium carbonate(conical flask) n heat to boil. 2. Std solution is taken in burette. On titration white bulky ppt is formed (cuprousthiocyanate) Note the vol of sugar solution required.
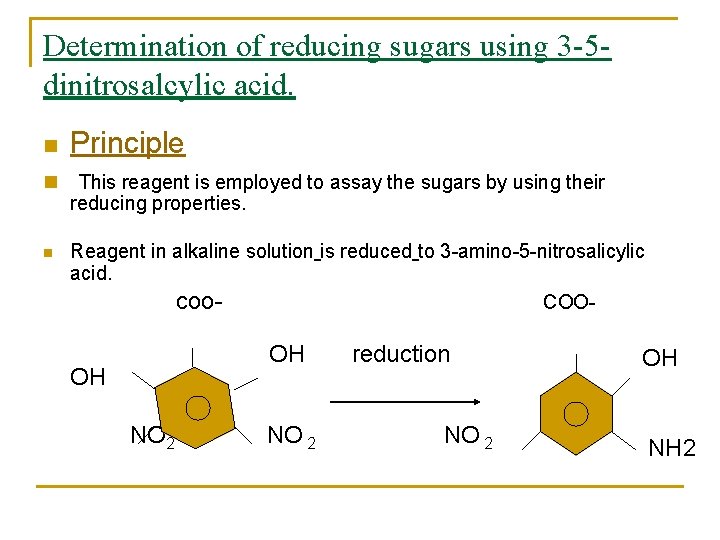
Determination of reducing sugars using 3 -5 dinitrosalcylic acid. n n Principle This reagent is employed to assay the sugars by using their reducing properties. Reagent in alkaline solution is reduced to 3 -amino-5 -nitrosalicylic acid. coo- COO- n OH reduction OH OH NO 2 NH 2

Procedure Prepare the DNS reagent just before use. n 1 ml of the reagent to 3 ml of sugar solution in a test tube. n Prepare blank boil n cool measure at 510 nm estimate concentration
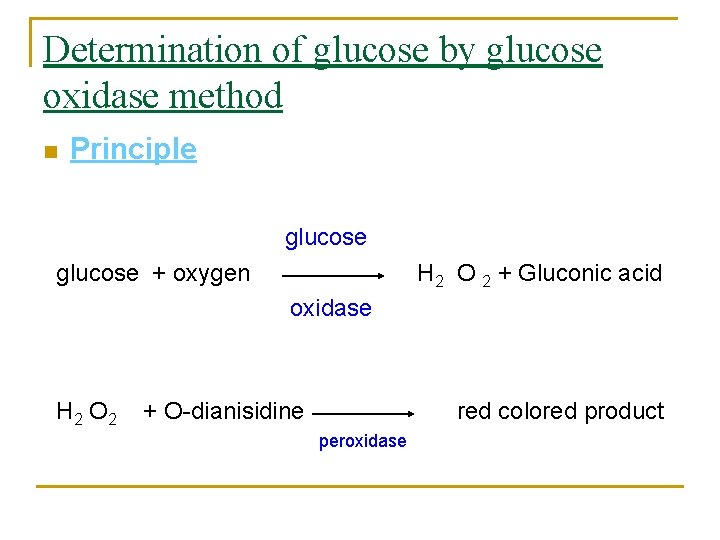
Determination of glucose by glucose oxidase method n Principle glucose + oxygen H 2 O 2 + Gluconic acid oxidase H 2 O 2 + O-dianisidine red colored product peroxidase

Procedure n n Materials; Glucose oxidase peroxidase reagent. Working standard solution. Procedure 0. 5 ml deprotinised plant extract + 0. 5 ml dw+1 ml reagent Incubate all tubes at 35 c for 40 min. Add 2 ml of 6 N HCL Read the color intensity at 540 nm

Phenol sulphuric acid method for total carbohydrate determination. n Principle; Glucose acidmedium hydroxymethyl furfural phenol green color product. Materials ; 1. Phenol 5% 2. Sulphuric acid 96% reagent grade. 3. Standard glucose.

Procedure n Pipette out 0. 2 , 0. 4, 0. 6, 0. 8 & 1 ml of working std. pipette out 0. 1 & 0. 2 ml of sample sol. Makeup to 1 ml. H 2 O set a blank with 1 ml water Add 1 ml of phenol sol to each tube. Add 5 ml of sulphuric acid to each tube & shake well. After 10 min shake & place in water bath for 25 -30 c. Read the color at 490 nm.

Estimation of starch by anthrone reagent. Sample + 80% alcohol sugars get remove. hydrolysed dehydrated n starch glucose hydroxymethy furfural anthrone n green color product Materials : Anthrone sol 80% ethanol 52% perchloric acid Std glucose sol.

Procedure n Homogenise 0. 1 to 0. 5 g of sample in 80% alcohol. n Centrifuge & retain residue, wash & dry. n Add 5 ml of water & 6. 5 ml of 52% perchloric acid. n Extract at 0 c for 20 min. Centrifuge & save supernatant. n Repeat the extraction using fresh perchloric acid, centifuge , makeup n Pipette out 0. 1 or 0. 2 ml of supernatant & make up to 1 ml. n Prepare the std & add 4 ml of anthrone reagent. heat & cool.

Determination of amylose n Principle The iodine is adsorbed within the helical coils of amylose to produce a blue colored complex. Materials Distilled ethanol 1 N NAOH 0. 1% phenolphthalein Iodine reagent Standard solution.

Procedure Weigh 100 mg ofsample, &add 1 ml ofethanol, add 10 ml of 1 n NAOH. Make up to vol to 100 ml. 2. 5 extract , add 20 ml dw & 3 drops of phenolphthalein. Add 0. 1 n HCLdrop by drop until pink color just disappears. Add 1 ml of iodine reagent & make up to 50 ml. Take 0. 2, 0. 4, 0. 6, &1 ml of std amylose & develop color. Calculate the amt of amylose.

Determination of fructose. n Principle n Hydroxymethyl furfural + resorcinol red color product Materials Resourcinol reagent Dilute HCL Standard fructose solution.

Procedure 2 ml of solution , add 1 ml of resourcinol reageant Add 7 ml of dil HCL Pipette out 0. 2, 0. 4, 0. 6, 0. 8 & 1 ml of std & makeup to 2 ml with H 2 O Add 1 ml of resourcinol & 7 ml of dilute HCL heat all tubes in water bath , cool Read the color at 520 nm within 3 omin.


Column chromatography Adsorbent materials – siliceous earths or charcoal mono & disaccharides & higher carbohydrates - charcoal Methyl mannosides – cellulose powder n Mobile solvent – butanol: pyridine: water (10: 3: 3)

Estimation by HPLC n Problems with detection system for carbohydrates have limited the application of HPLC to carbohydrates. n The ploar bonded phases – lichrosorb – NH 2 n Mobile phases– acetonitrile – water mixture. n This system is used for determination of sugar content of soyabeans, dairy roducts , molases. n The determination of glycoproteins levels & of protein/carbohydrate ratio in glycoprotein is very imp in diagnosis of cancer patients.

Ion exchange chromatography n Derivitization ----bisulphite addtion product of a carbonyl compound & the production of carbohydrates – substituted boric acid. n Separation of larger quantities of sugar - anion exchange columns. n Mixtures of sugars – on the bases of the relative stability of their borate complexes.

Paper chromatography n Developing solvent – mixture of butanol , acetic acid& water. n Reagents– aniline, diphenyl amine, phosphoric acid. n Ascending paper chromatography – separation of monosaccharides. n Chromatogram sprayed with p-anisidine, eluted with aq. stannous chloride. n Benzidine with stannous chloride ---aldose analysis. n Triphenyltetrazolium, benzidine – direct photometric examination of paper chromatography spots.

Estimation of sugars in ice cream

Thin layer chromatography n Principle. Thin even layer of adsorbent is coated on glass plate. spots are applied Development takes place & spots are identified BINDING MATERIALS. silicis acid, alumina, keiselguhr THICKNESS 250µ is maintained with controllable spreader.

Procedure Prepare silica gel plate – activates at 105 c for 30 min. Solvent preparation & std sugar solution preparation. Spot sugars & unknown Development Spray coating reagent Calculate Rf. Rf = distance travelled by compound distance moved by glucose.

Miscellaneous methods n Gasometry is applicable to carbohydrate analysis. n Hagedorn-jenson reaction--- reaction of sugars with ferricyanide. n Best range 10 – 120 mcg of reducing sugar Biochemical procedures also prove useful for identification of carbohydrates. n

n Electrochemical procedures have been adopted to carbohydrate analysis. n Reduction of carbonyl function with a sodium borohydride followed by titration of residual borohydride with acid to produce hydrogen gas. .

Significances n Generally available as immediate energy source. n Cellulose, polysaccharides is an important structural component of plant cells. n Glucose is essential for cell function. n Significance of carbohydrates for gastrointestinal function. n Majore role in working process of immune system, fertilisation, pathogenesis, blood clotting.

k. Gowthami Keerthana Sirisha M. pharm(analysis)
- Slides: 51