Preliminary Data on U S DNABased Patents and

Preliminary Data on U. S. DNA-Based Patents and Plans for a Survey of Licensing Practices Robert Cook-Deegan, Le. Roy Walters, Derrick Pau, Bi Ade, Stephen Mc. Cormack, Janella Gatchalian, and Richard Burgess Kennedy Institute of Ethics, Georgetown University Collaborators are involved in the empirical studies but do not necessarily concur with analysis or conclusions

Themes u. Trends in US DNA-based patents u. Why licensing is where the action is u. Public-Private interactions in genomics, using DNA Patent Database

What is a DNA-based Patent? n n n One or more claims based on DNA or RNA Algorithm initially developed by James Martinell, USPTO, for OTA Revising search algorithm, doing sensitivity analysis, improving free public acess

Search algorithm (November 2002) n Patent classes n n Terms n n 047 (plant), 119 (animal), 260 (food), 426 (organic), 435 (molecular biology and microbiology), 536/22. 1 through 536/25. 5 (organic compounds/nucleic acids), and 800 (multi-celled organisms) Antisense, c. DNA, centromere, deoxyoligonucleotide, deoxyribonucleic, deoxyribonucleotide, DNA, exon, gene or genes, genetic, genome or genomic, genotype, haplotype, intron, mt. DNA, nucleic, nucleotide, oligodeoxynucleotide, oligoribonucleotide, plasmid, polymorphism, polynucleotide, polyribonucleotide, ribonucleic, recombinant DNA, RNA, m. RNA, r. RNA, si. RNA, sn. RNA, or t. RNA, ribonucleoprotein, hn. RNP, or sn. RNP, SNP Terms tested and rejected after sensitivity analysis n Seq. ID, sequence, atg*, telomere, dideoxy*
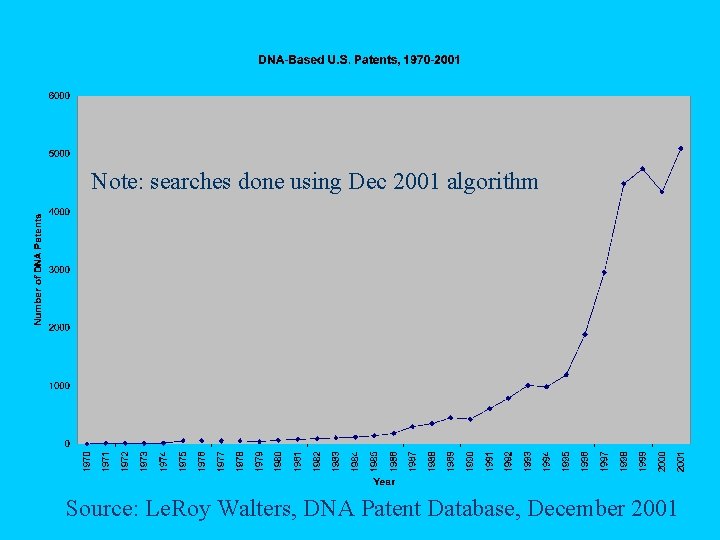
Note: searches done using Dec 2001 algorithm Source: Le. Roy Walters, DNA Patent Database, December 2001

Growth of DNA Patents 1970 s 1980 s 1990 s Patents Issued 292 1, 869 19, 082

DPD Patent Coding Sheet n Categorizes n n n n Inventor information (#, country of origin) Patent filing information Assignee information (#, country of origin, type of organization) US government involvement (Bayh-Dole) Types of claims (c. DNA [gene], vector, transgenic animal, vaccine, etc. ) DNA-based patents issued between 1980 -1993 were manually coded into the DPD Patent Coding Sheet
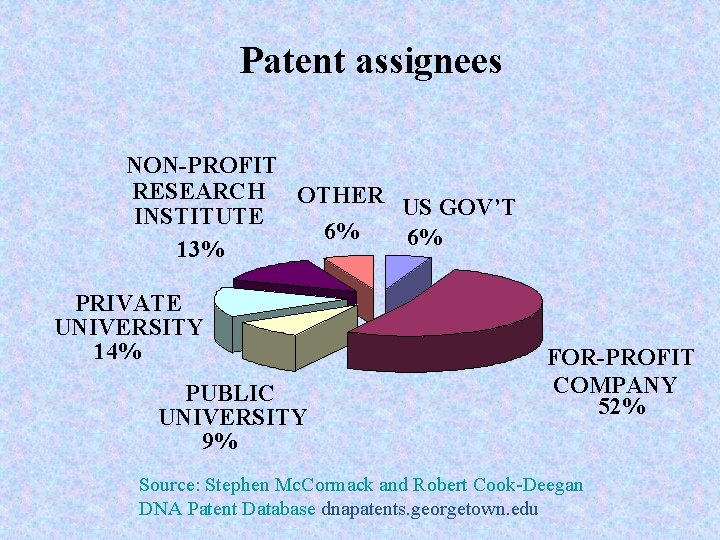
Patent assignees NON-PROFIT RESEARCH OTHER US GOV’T INSTITUTE 6% 6% 13% PRIVATE UNIVERSITY 14% PUBLIC UNIVERSITY 9% FOR-PROFIT COMPANY 52% Source: Stephen Mc. Cormack and Robert Cook-Deegan DNA Patent Database dnapatents. georgetown. edu
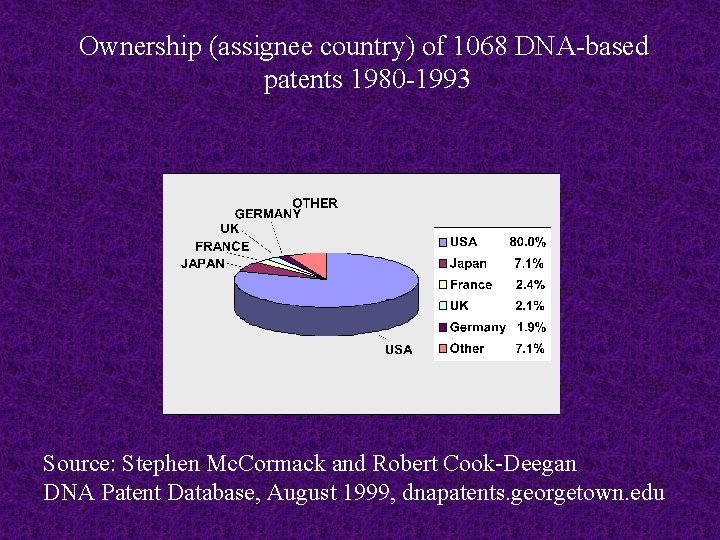
Ownership (assignee country) of 1068 DNA-based patents 1980 -1993 Source: Stephen Mc. Cormack and Robert Cook-Deegan DNA Patent Database, August 1999, dnapatents. georgetown. edu
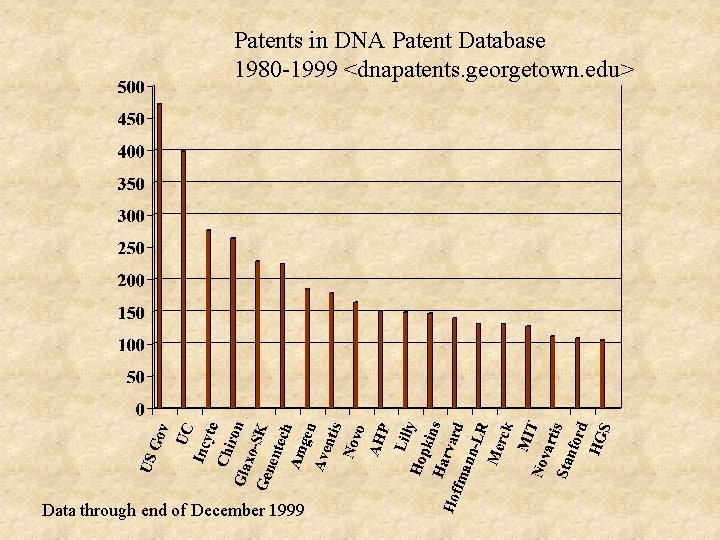
Patents in DNA Patent Database 1980 -1999 <dnapatents. georgetown. edu> Data through end of December 1999

Why study licensing? Move debate beyond patentability n Presumption that patent law should apply to all kinds of inventions equally n Question becomes how patent rights are exercised, not whether they exist What if EPO not patented? What if Cohen-Boyer licensed exclusively? n

Gedanken experiment: Sanger sequencing patent License with provision that sequence data be publicly disclosed per Bermuda rules n. No policy change in high-throughput sequencing centers n. What would Incyte, Human Genome Sciences, Celera, etc. , have done? n. What would universities do with other sequences outside HGP?

How Do We Know Universities Are Important? n n n Biotech/pharma highest fraction of products and services dependent on academic research (Edwin Mansfield surveys) Patent citation literature (Cockburn, Henderson, Jaffe, Trajtenberg) Literature citation studies (Narin) Spillovers (Gambardella, Henderson-Cockburn) Synthesis (Nelson, Mowery)

What’s funny about universities? n n n Mission = research + teaching Some publicly owned, all publicly supported for research Don’t make products Do sell services, run component businesses Take money > make money What to maximize? Net revenue or net social good?

Funding Sources for Licensing Survey n n U. S. Department of Energy National Human Genome Research Institute, U. S. National Institutes of Health Lori Pressman to do survey; who has done AUTM surveys

Licensing Survey n n 24 US and Canadian “academic” institutions (universities and nonprofit) Biggest recipients of US Federal R&D Their U. S. DNA-patent holdings Licensing practices Complement to Cho/Merz and Mowery/Nelson studies

Licensing practices n n n Exclusive, co-exclusive, non-exclusive agreements Patent clusters Licensing income Research tools treated differently? 9 -10 qualitative case studies of “interesting” patent clusters or licensing practices Look for patents not reported to federal government (cross-check to Ra. Di. US and Pub. Biomed databases)

Institutions of Interest n Most DNA-based patents: UC, UTx, Hopkins, Scripps, Mass. General, Harvard, Stanford, Salk, Wash. U, Columbia, MIT, Rockefeller, Cornell, UPenn, UWisc n High fraction of patents are DNA-based Salk, Dana Farber, Baylor, Scripps, Rockefeller, Wash. U, Harvard, Jefferson, Mass. General, NYU, Yale, Hopkins, Columbia, Brigham&Women’s

Valuable Gene Patents u. Human Insulin u. Clot-dissolving enzymes u. Growth Factors –Erythropoietin –White blood cell lineages Gene patent for making therapeutic protein

Techniques Cohen-Boyer r. DNA u Polymerase chain reaction u 4 -color fluorescence sequencing u Multiplex sequencing u Microarray techniques u Gene transfer u Public domain technologies u Sanger sequencing u Maxam-Gilbert sequencing u Monoclonal antibody production

Diagnostics Huntington’s by RFLP u Cystic Fibrosis u – Research exemption u Apo. E for Alzheimer’s – Exclusively licensed u Br. CA 1 & 2 – One company Hemochromatosis u Many genes and mutations u Most single nucleotide polymorphisms u Many genes and mutations u

Patents induce private development investment u Automated sequencing u Therapeutic proteins: gene patents protect post-discovery development investment – long clinical trial testing period – Trials and manufacturing startup expensive u Microarray applications

Patents were not essential to induce health research investment u Br. CA 1 & 2 and hemochromatosis u Cohen-Boyer u PCR u Sequencing methods u Cell fusion and monoclonal antibody techniques u Most university-based discoveries

Patents a source of revenue u Startups (intellectual “capital”) u Universities (licensing income)

Patents a tool for policy change u PXE International model – Sharon Terry co-inventor u Indigenous Peoples movement – Licensing rights to access u NIH and universities as major “property managers” – Create norms through licensing – Increase transparency – Initiate pooling?

Bayh-Dole Act: Purpose u Commercially exploit inventions developed using federal funds u Legal mandate and financial incentive to do business with firms that can develop an invention into a product or service

Policy Options: Bayh-Dole u Transparency – Stop flying blind u u u Research exemption (Dreyfus) Diagnostic use exemption (Rivers bill) More flexibility to pre-empt patenting – Lower threshold than “exceptional circumstances” (Rai/Eisenberg) – Simplify process u u Licensing oversight and march-in (Mc. Garey/Cell. Pro) Earlier release of sequence data (BCD/Mc. Cormack)

Policy Options: Universities u Informational “markets” on licensing terms u Explicit attention to where scientific and academic norms conflict with business interests and revenuemaximization u Due diligence on licensees u Patent pools?

Policy options: NIH u Guidance on disposition of research tools u Guidance on licensing practices u Develop criteria for “exceptional circumstances” u Develop criteria for “march-in” u Patent pool?

Policy Options: Companies u Consortia for preserving public domain research tools – EST – SNP u Patent pools u Guidance on licensing practices – BIO and Ph. RMA

Policy Options: IP law u Research exemption for all patents – What about academic health centers as major service providers? Exemption kills that market. u u Shift to “opposition” rather than re-examination Data exclusivity for FDA approval as a substitute for patents (for therapeutics and devices) Liability rules and contract practices; unfair competition law (per Reichman/Uhlir) Database protection
- Slides: 31