Preliminary data from the prospective ABIRISK IBD cohort

Preliminary data from the prospective ABIRISK IBD cohort Clinical response and anti-drug antibodies

The classical objectives of IBD management ü ü ü To induce and maintain remission § Resulting in decreased hospitalizations and surgery To prevent complications § Including treatment adverse events Resulting in improved quality of life
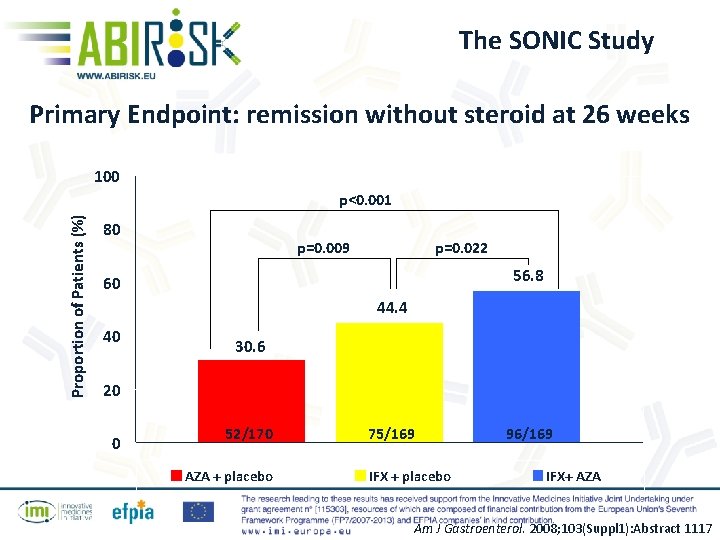
The SONIC Study Primary Endpoint: remission without steroid at 26 weeks 100 Proportion of Patients (%) p<0. 001 80 p=0. 009 p=0. 022 56. 8 60 44. 4 40 30. 6 20 0 52/170 AZA + placebo 75/169 IFX + placebo 96/169 IFX+ AZA Am J Gastroenterol. 2008; 103(Suppl 1): Abstract 1117

Anti-TNF failures in IBD • Approximately 1/3 of patients do not show primary response and 2/3 do not show remission • In placebo-controlled trials, about 50% of patients lost response over 1 year • Treatment optimization with increased dose or shortened interval allowed to recover response in 50 -90% of the patients • In literature reviews, yearly loss of response despite optimization was 13% for IFX and 20% for ADA Allez M, et al. JCC 2010 Gisbert JP, Panes J. Am J Gastroenterol. 2009 Billioud V, et al. Am J Gastroenterol 2011

Immunogenicity Presence of antibodies against anti-TNF m. Abs confers a risk of discontinuation of treatment and a risk of development of hypersensitivity reactions in all immune-mediated inflammatory diseases

Factors affecting the pharmacokinetics of anti-TNF m. Abs Ordas I et al, Clin Pharm Ther 2012

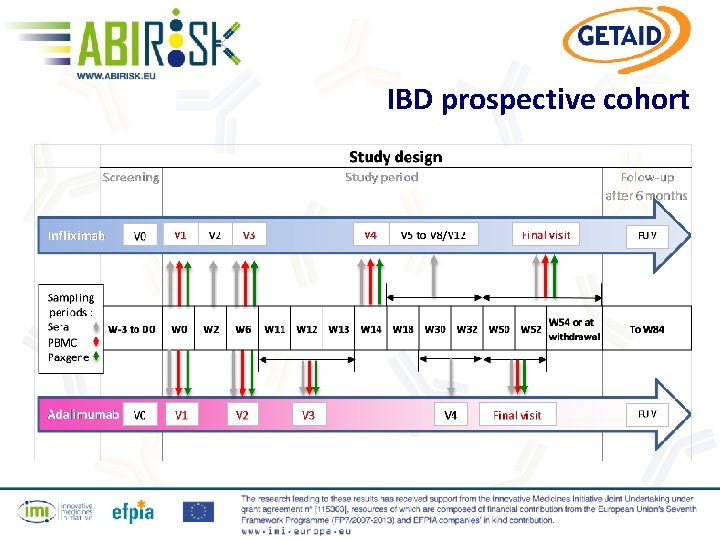
IBD prospective cohort Ø Primary objective § To find early bio-markers able to predict immunization against the biopharmaceuticals within the first year of treatment.

IBD prospective cohort Ø Secondary objectives • • To find bio-markers able to predict long-term immunization against the biopharmaceuticals To analyze the correlation between immunization to biopharmaceuticals with hypersensitivity reactions, loss of response and biopharmaceuticals levels To identify molecular and cellular biomarkers associated with the development of antidrug-antibodies (ADA) To be able to associate an immunological signature to patients with anti-drug-antibodies

IBD prospective cohort Ø Study design § Multicenter, prospective, non interventional study for collection of biological samples (blood) to be used for in vitro biomarker assay(s) Ø Main selection criteria § Patients in the first line of anti-TNF therapy including adalimumab or infliximab Ø Total expected number of patients : § 200 patients are to be enrolled to have at least 50 ADA+ patients for the final evaluation in 17 centers (France, Belgium and Israel)

IBD prospective cohort Judgment criteria § Immunization against the BP is defined by the presence of ADA within the first 12 months § Quantification of ADA at W 0, W 6, W 12 and W 52 § Clinical response and remission at W 6, W 12, W 52 and at withdrawal if the drug is discontinued § ADA-associated adverse clinical events at any time points
IBD prospective cohort
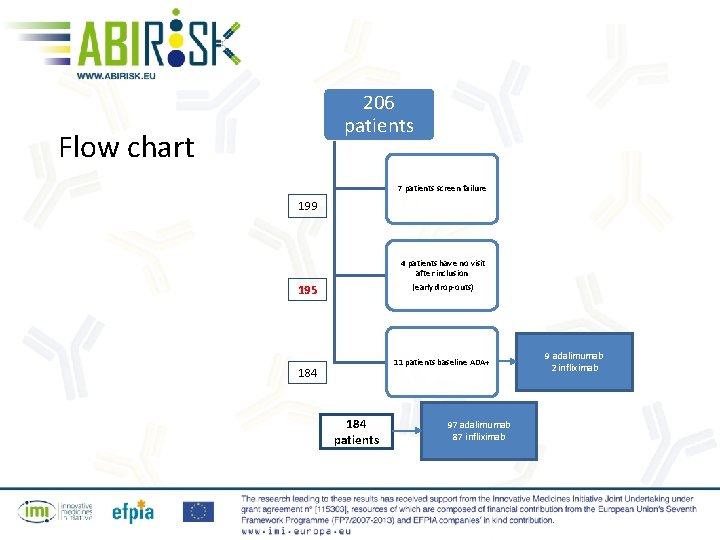
206 patients Flow chart 7 patients screen failure 199 4 patients have no visit after inclusion (early drop-outs) 195 11 patients baseline ADA+ 184 patients 97 adalimumab 87 infliximab 9 adalimumab 2 infliximab

Definitions of remission and response • Crohn’s disease (Harvey-Bradshaw index) – Response : HBIi – HBI 0 ≥ 3 or HBI ≤ 4 – Remission : HBI ≤ 4 • Ulcerative Colitis (Mayo score) – Response : Mayoi – Mayo 0 ≥ 3 or Mayo ≤ 2 – Remission : Mayo ≤ 2

Rates of clinical remission

Definitions of remission and response • Crohn’s disease: Harvey-Bradshaw index – Response : HBIi – HBI 0 ≥ 3 or HBI ≤ 4 – Remission : HBI ≤ 4 • Ulcerative Colitis: Mayo score – Response : Mayoi – Mayo 0≥ 3 or Mayo ≤ 2 – Remission : Mayo ≤ 2
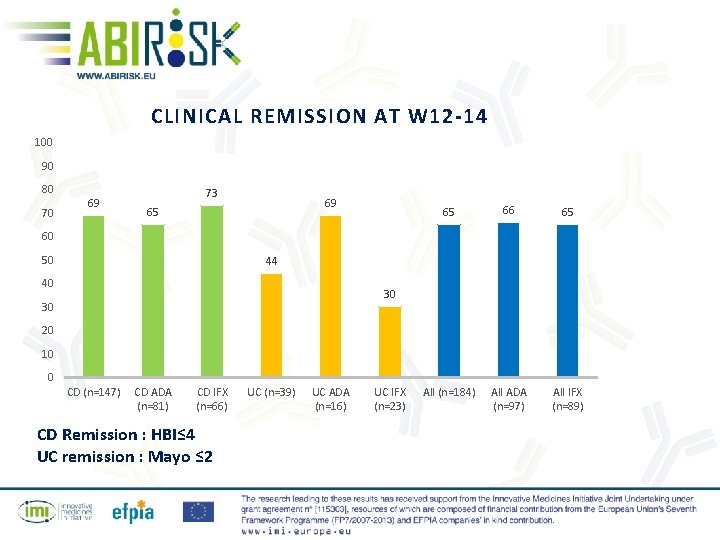
CLINICAL REMISSION AT W 12 -14 100 90 80 70 69 73 69 65 65 66 65 All (n=184) All ADA (n=97) All IFX (n=89) 60 50 44 40 30 30 20 10 0 CD (n=147) CD ADA (n=81) CD IFX (n=66) CD Remission : HBI≤ 4 UC remission : Mayo ≤ 2 UC (n=39) UC ADA (n=16) UC IFX (n=23)
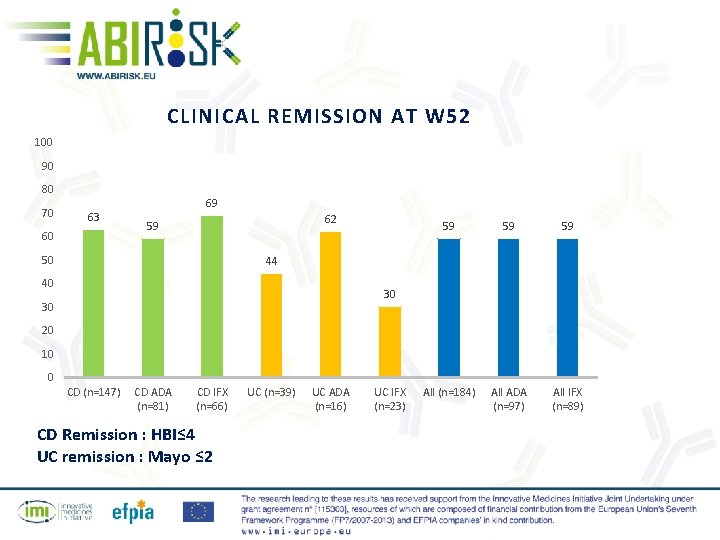
CLINICAL REMISSION AT W 52 100 90 80 70 63 60 69 62 59 50 59 59 59 All (n=184) All ADA (n=97) All IFX (n=89) 44 40 30 30 20 10 0 CD (n=147) CD ADA (n=81) CD IFX (n=66) CD Remission : HBI≤ 4 UC remission : Mayo ≤ 2 UC (n=39) UC ADA (n=16) UC IFX (n=23)

Influence of anti-drug antibodies on clinical outcome ?
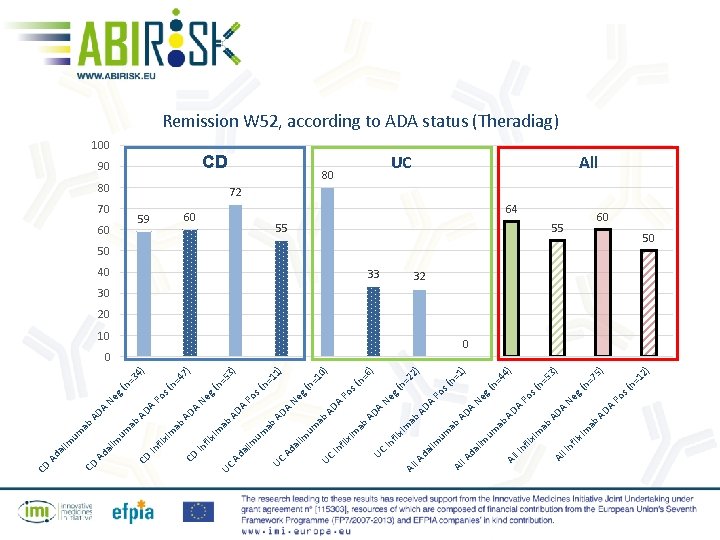
ab AD A A AD ) 55 12 64 s( n= UC Po ) n= 75 g( Ne 3) 4) n= 5 Po s( n= 4 g( Ne 1) 55 xim ab xim l In fli Al l In fli ab A AD s( n= Po ) 22 0 um ab A AD 10 Al um im da lim l. A da l ab Ne g( n= 33 Al l. A xim A n= 6) s( Po 0) 80 Al fli In AD A AD n= 1 40 UC ab ab g( Ne 1) n= 1 ) CD xim um im fli In Ad al AD A s( Po 53 60 UC UC ab A AD (n = 80 um im al ab 90 Ad xim ) 100 UC Ne g 47 59 fli In A AD ) 34 s( n= Po g( n= Ne 60 CD AD A A AD 70 xim ab fli In ab um im al Ad CD CD CD Remission W 52, according to ADA status (Theradiag) All 72 60 50 50 32 30 20 0
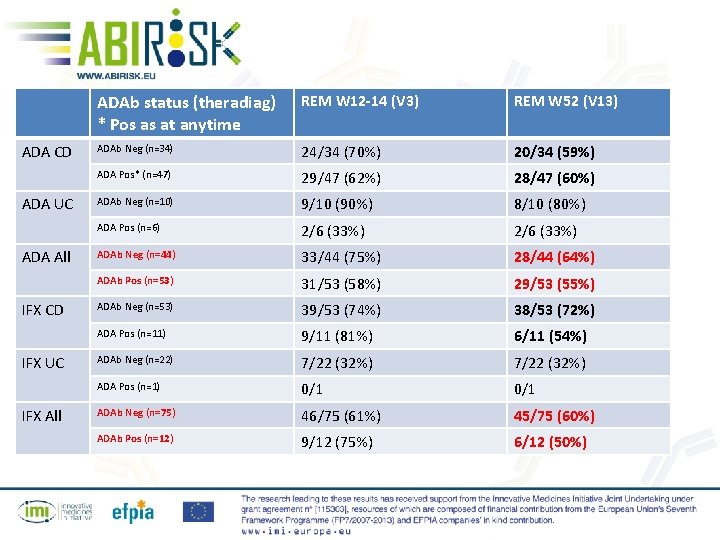
ADA CD ADA UC ADA All IFX CD IFX UC IFX All ADAb status (theradiag) * Pos as at anytime REM W 12 -14 (V 3) REM W 52 (V 13) ADAb Neg (n=34) 24/34 (70%) 20/34 (59%) ADA Pos* (n=47) 29/47 (62%) 28/47 (60%) ADAb Neg (n=10) 9/10 (90%) 8/10 (80%) ADA Pos (n=6) 2/6 (33%) ADAb Neg (n=44) 33/44 (75%) 28/44 (64%) ADAb Pos (n=53) 31/53 (58%) 29/53 (55%) ADAb Neg (n=53) 39/53 (74%) 38/53 (72%) ADA Pos (n=11) 9/11 (81%) 6/11 (54%) ADAb Neg (n=22) 7/22 (32%) ADA Pos (n=1) 0/1 ADAb Neg (n=75) 46/75 (61%) 45/75 (60%) ADAb Pos (n=12) 9/12 (75%) 6/12 (50%)
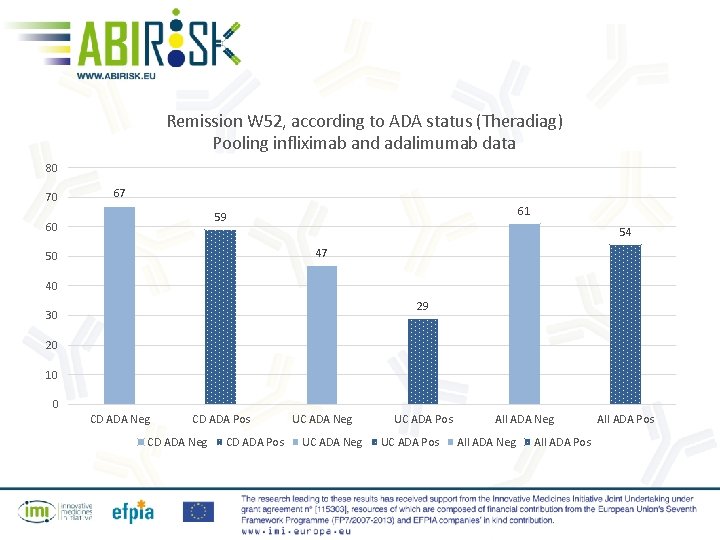
Remission W 52, according to ADA status (Theradiag) Pooling infliximab and adalimumab data 80 70 67 61 59 60 54 47 50 40 29 30 20 10 0 CD ADA Neg CD ADA Pos UC ADA Neg UC ADA Pos All ADA Neg All ADA Pos
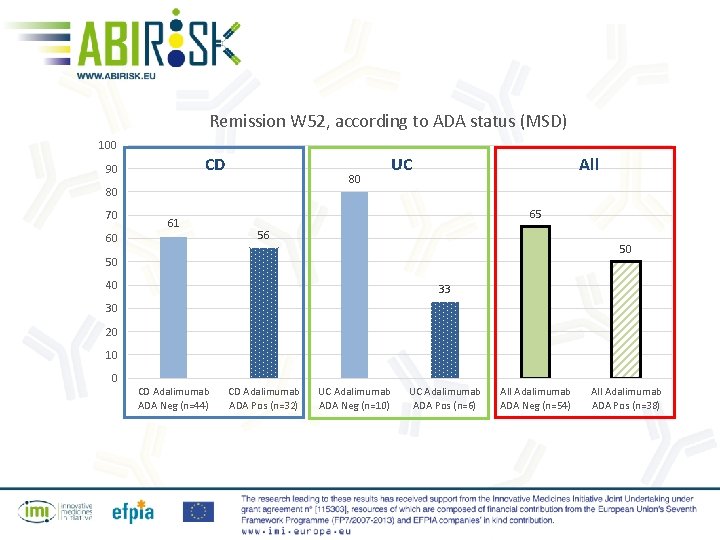
Remission W 52, according to ADA status (MSD) 100 CD 90 80 80 70 61 60 UC All 65 56 50 50 40 33 30 20 10 0 CD Adalimumab ADA Neg (n=44) CD Adalimumab ADA Pos (n=32) UC Adalimumab ADA Neg (n=10) UC Adalimumab ADA Pos (n=6) All Adalimumab ADA Neg (n=54) All Adalimumab ADA Pos (n=38)
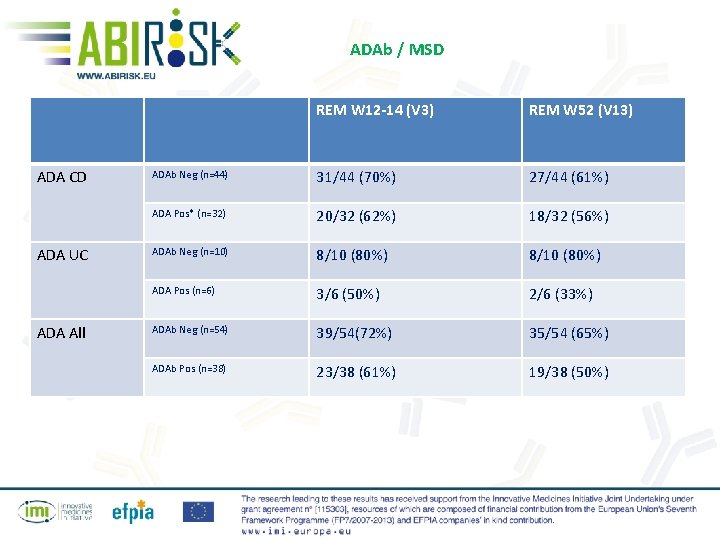
ADAb / MSD ADA CD ADA UC ADA All REM W 12 -14 (V 3) REM W 52 (V 13) ADAb Neg (n=44) 31/44 (70%) 27/44 (61%) ADA Pos* (n=32) 20/32 (62%) 18/32 (56%) ADAb Neg (n=10) 8/10 (80%) ADA Pos (n=6) 3/6 (50%) 2/6 (33%) ADAb Neg (n=54) 39/54(72%) 35/54 (65%) ADAb Pos (n=38) 23/38 (61%) 19/38 (50%)
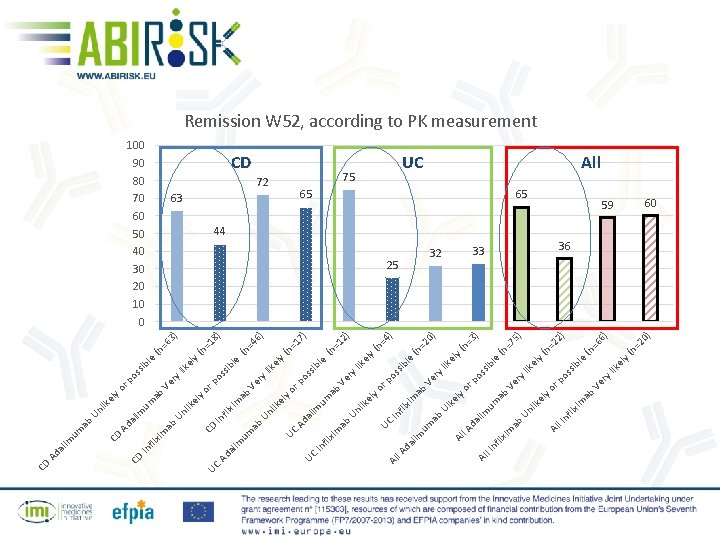
yl ly 0) 65 (n =2 ) 2) 66 (n =2 ) UC ike le sib os rp ly ike 75 (n = ) 33 Ve r yo yl Ve r le ib ss n= 3 44 ab xim fli l In ab el lik Un um po y( 0) =2 (n ) 65 Al ab xim l In fli or el lik le ib 32 Al da lim l. A ly ke Ui Ve ry os s n= 4 y( 12 ) (n = 25 Al ab xim In fli el lik le ) 75 ab um lim da l. A Al ry Ve rp yo el Un lik ab um 17 n= y( 6) CD UC ab xim sib rp os el lik 63 In fli im al Ad ry (n =4 ) ) 18 (n = 63 (n = 72 UC UC yo el Un lik Ve le sib ike ly yl os rp yo ab xim In fli ab um im Ad al CD el lik Un Ve r le ib os s rp 100 90 80 70 60 50 40 30 20 10 0 UC ab ab um im yo el lik Un Ad al xim In fli CD ab um im al Ad CD CD Remission W 52, according to PK measurement All 59 60 36
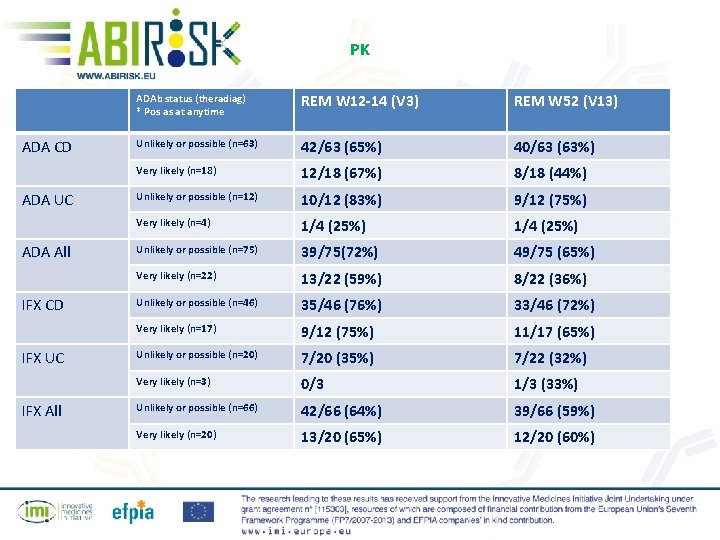
PK ADA CD ADA UC ADA All IFX CD IFX UC IFX All ADAb status (theradiag) * Pos as at anytime REM W 12 -14 (V 3) REM W 52 (V 13) Unlikely or possible (n=63) 42/63 (65%) 40/63 (63%) Very likely (n=18) 12/18 (67%) 8/18 (44%) Unlikely or possible (n=12) 10/12 (83%) 9/12 (75%) Very likely (n=4) 1/4 (25%) Unlikely or possible (n=75) 39/75(72%) 49/75 (65%) Very likely (n=22) 13/22 (59%) 8/22 (36%) Unlikely or possible (n=46) 35/46 (76%) 33/46 (72%) Very likely (n=17) 9/12 (75%) 11/17 (65%) Unlikely or possible (n=20) 7/20 (35%) 7/22 (32%) Very likely (n=3) 0/3 1/3 (33%) Unlikely or possible (n=66) 42/66 (64%) 39/66 (59%) Very likely (n=20) 13/20 (65%) 12/20 (60%)

Conclusion - 1 • These are preliminary results (full monitoring of the prospective cohort finished last week) – Statistical analysis … • High rates of clinical remission in this prospective cohort – The lower rates of remission with infliximab in UC may be related to a higher proportion of acute severe colitis – Analysis to be done: Clinical remission without steroid, Clinical remission and CRP normalization, statistics, predictors?

Conclusion - 2 • Assessment of immunogenicity – Different assays … – Significant correlation between clinical outcome and ATI in CD (to be confirmed) – Immunogenicity was defined by the presence of ADAb at anytime • These are preliminary results which must be completed: – Primary Vs. secondary failures, Impact on the early detection of ADAb on clinical outcome? Exclusion of transient ADAb? Impact of the optimization on the detection of ADAb, – Comparison of MSD and Theradiag assays, Correlation with PK – Analysis pooled with RA and JIA

Acknowledgments • Co-investigators Franck Carbonnel, Jean-Marc Gornet, Arnaud Bourreille, Yoram Bouhnik, Maria Nachury, Benjamin Pariente, Guillaume, Anthony Buisson, Gilles Bommelaer, Marion Simon, Laurent Beaugerie, Stéphane Nancey, Elise Chanteloup, … Yehuda Chowers • GETAID Sabrina Williams, all co-investigators • ABIRISK Marc Pallardy, Sebastian Spindeldreher, Philippe Broet, Delphine Bachelet, Signe Hassler, Agnes Hincelin-Mery, Vincent Mikol, Sophie Tourdot, Pierre Doennes, Anna Fogdell-Hahn, Florian Deisenhammer, Xavier Mariette Denis Mulleman, Gilles Paintaud, David Ternant, Salima Hacein-Bey, Niek de Vries, Bernard Maillere, Claudia Mauri • All partners
- Slides: 28