PreLab p GLO Bacterial Transformation AP Biology Ms

Pre-Lab: p. GLO Bacterial Transformation AP Biology Ms. Day

Bacterial Transformation • Step 1 DNA Isolation – Isolation of the “Gene of Interest” (foreign DNA) • Step 2 Recombinant DNA – Insertion of foreign DNA into bacterial plasmid using restriction enzymes and DNA ligase • http: //www. dnalc. org/resources/animations/transformation 1. html • Step 3 Transformation – Insertion of recombinant DNA into bacteria by making bacteria competent (weaken) • Use Ca. Cl 2 and heat shock techniques

How do you make Bacteria competent? • Step 1: Add Calcium Chloride (Ca. Cl 2) – Ca. Cl 2 is in a solution (creates Ca+2 and Cl- ions) – DNA in plasmid is negatively charged due to phosphate groups in the backbone – Cell membrane of E. coli also is negatively charged because phospholipids are made of same phosphate groups (PO 4 -3) – Ca+2 ions neutralize charges so plasmid can get near (and inside) bacterial cell.

How do you make Bacteria competent? • Step 1: Use Heat Shock – Heat Shock is a process that uses warm water (bath) and ice to help get plasmid inside cell • Add recombinant plasmid + host cell + Ca. Cl 2 solution to ice then heat then back on ice – Heat = increases kinetic energy of matter • Molecules/atoms move faster – Ice = decreases kinetic energy of matter • Molecules/atoms move slower • http: //www. dnalc. org/resources/animations/transformati on 2. html
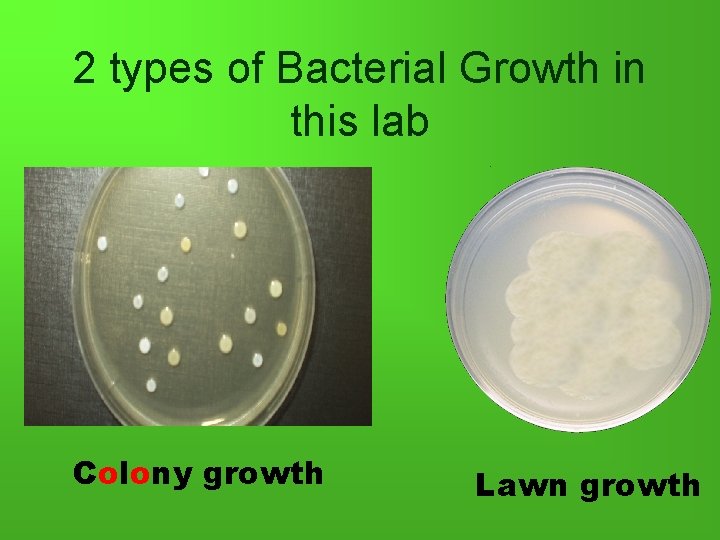
2 types of Bacterial Growth in this lab Colony growth Lawn growth
Step 1: DNA Isolation digesting the ”gene of interest” with restriction enzyme • In the lab, this has been done for you! • Gene of Interest = GFP
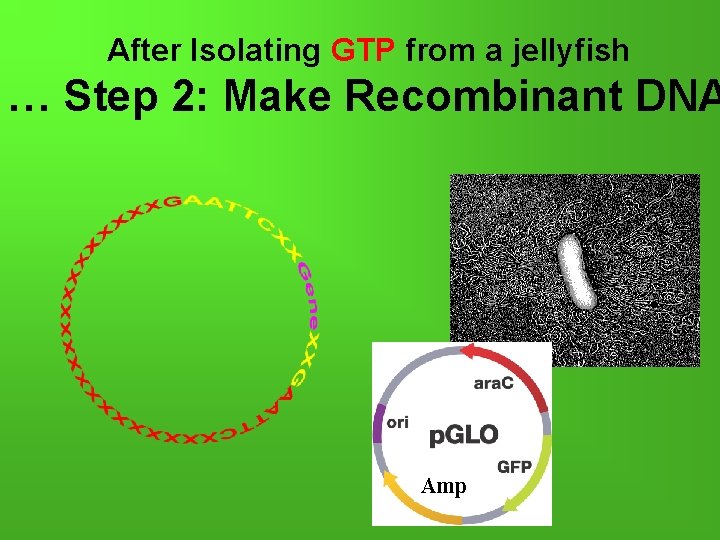
After Isolating GTP from a jellyfish … Step 2: Make Recombinant DNA Amp
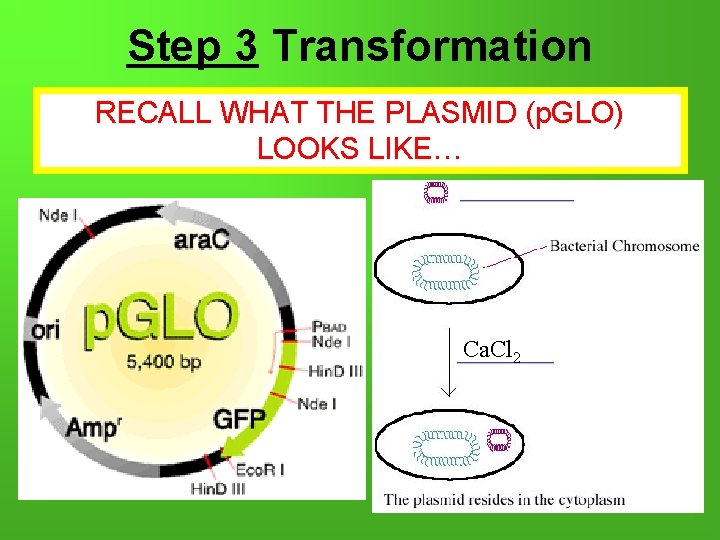
Step 3 Transformation RECALL WHAT THE PLASMID (p. GLO) LOOKS LIKE… Ca. Cl 2
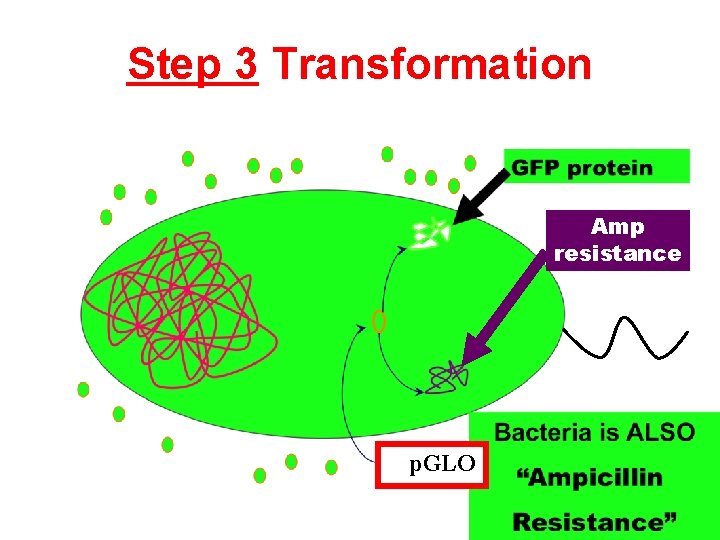
Step 3 Transformation Amp resistance p. GLO
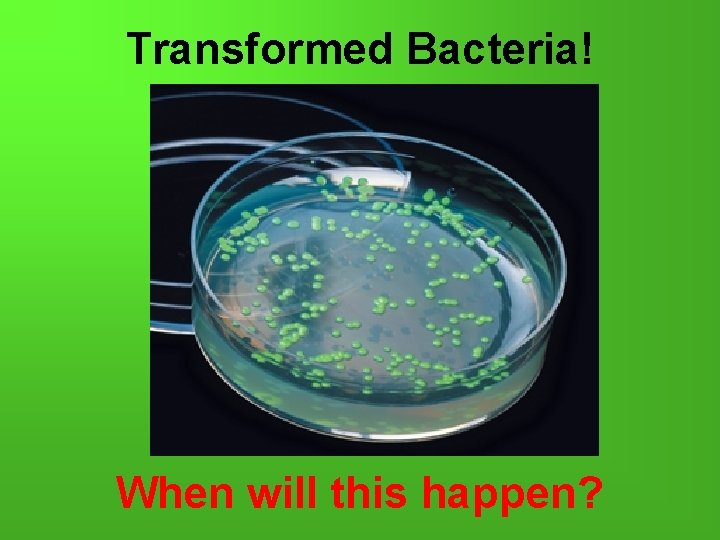
Transformed Bacteria! When will this happen?

What is the ARA operon? -Clusters of genes located together and transcribed from ONE promoter. -Ara operon = Inducible operon -3 arabinose structural genes present natural (not recombined) plasmid: ara. B, ara. A, ara. D -All 3 genes dependent on promoter (p. BAD) -Arabinose (sugar) changes the shape of the promoter (INACTIVATES REPRESSOR) • INDUCES transcription by allowing RNA polymerase to bind to the DNA
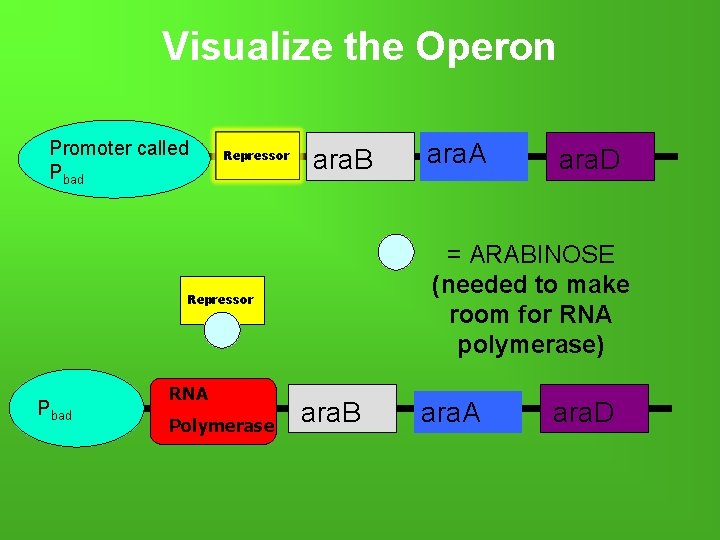
Visualize the Operon Promoter called Pbad Repressor ara. B Pbad Polymerase ara. D = ARABINOSE (needed to make room for RNA polymerase) Repressor RNA ara. B ara. A ara. D
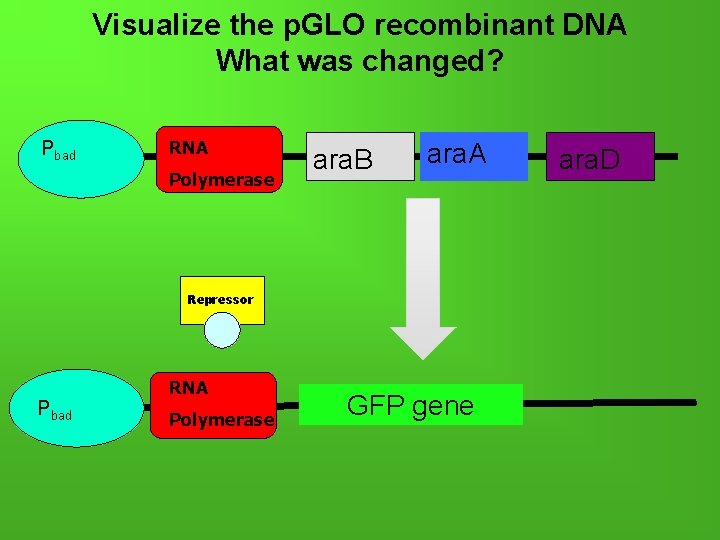
Visualize the p. GLO recombinant DNA What was changed? Pbad RNA Polymerase ara. B ara. A Repressor Pbad RNA Polymerase GFP gene ara. D

Some of your Materials
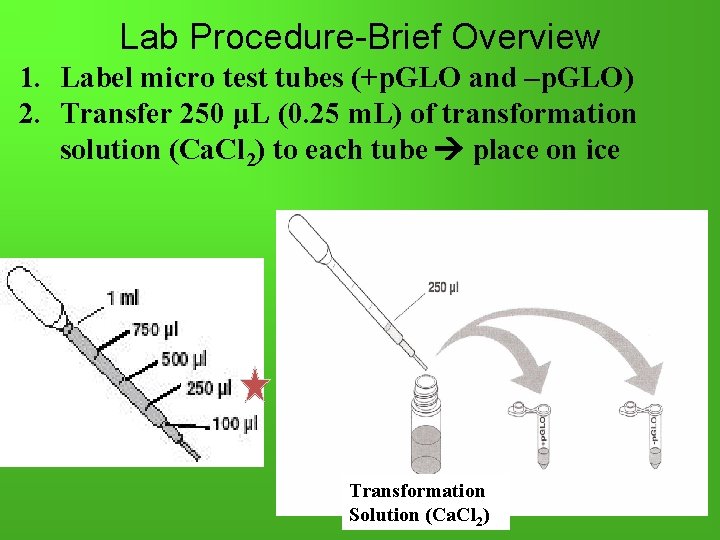
Lab Procedure-Brief Overview 1. Label micro test tubes (+p. GLO and –p. GLO) 2. Transfer 250 μL (0. 25 m. L) of transformation solution (Ca. Cl 2) to each tube place on ice Transformation Solution (Ca. Cl 2)

Lab Procedure-Brief Overview 3. Use sterile inoculating loop to transfer ~2 “fat” colonies of bacteria to +p. GLO tube spin loop to remove bacteria from loop to Ca. Cl 2 solution 4. Use a DIFFERENT sterile inoculating loop to transfer ~2 “fat” colonies of bacteria to -p. GLO tube DO NOT GET TOO MUCH BACTERIA NO chunks!

Lab Procedure-Brief Overview 5. You will need p. GLO plasmid… • Your TEACHER will micropipette 10 μL of plasmid into your +p. GLO tube • Mix plasmid into the cell suspension of the by tapping the CLOSED microtube on your desk! • Return the tube it to on ice. • DO NOT add plasmid DNA to the –p. GLO tube.
Lab Procedure-Brief Overview

Lab Procedure-Brief Overview -p. GLO 10: 00 +p. GLO 6. Incubate both +p. GLO and –p. GLO tubes on ice for 10 minutes

Lab Procedure-Brief Overview While the tubes are sitting on ice… 7. Label your 4 LB Nutrient agar plates on the bottom (not the lid) as follows: • Label one LB/amp plate: + p. GLO • Label the LB/amp/ara plate: + p. GLO • Label the other LB/amp plate: - p. GLO • Label the LB plate: -p. GLO

Lab Procedure-Brief Overview TIME TO HEAT SHOCK… 8. Use foam rack as a holder, transfer both the +p. GLO and -p. GLO tubes into the water bath, set at 42°C, for exactly 50 seconds. • Make sure to push the tubes all the way down in the rack so the bottoms of the tubes stick out and make contact with the warm water. When the 50 seconds are done, RAPIDLY place both tubes back on ice. Incubate tubes on ice for 2 minutes


Lab Procedure-Brief Overview 9. Remove the rack with tubes from ice and place on lab bench. Open a tube and, using a new sterile pipet, add 250 µl of LB nutrient broth to EACH tube and reclose it. • Use a new sterile pipet for the other tube. Incubate tubes for 10 minutes at room temperature. LB = FOOD

Lab Procedure-Brief Overview 10. After 10 min have passed, tap the closed tubes with your finger to mix. Using a new sterile pipet for each tube, pipet 100 µl of liquid onto the appropriate LB agar plates

Lab Procedure-Brief Overview 11. Use a new sterile loop for each plate. Spread liquid evenly around surface of LB agar using streaking method. DO NOT PRESS TOO DEEP INTO THE AGAR.

Streaking Plates with bacteria
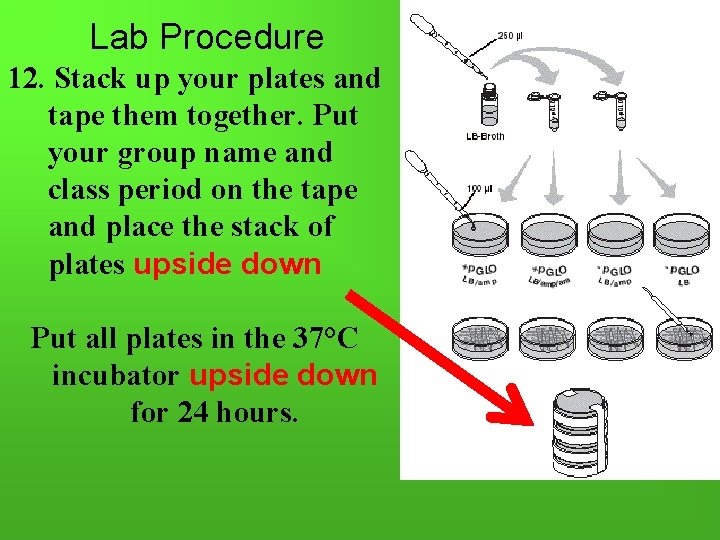
Lab Procedure 12. Stack up your plates and tape them together. Put your group name and class period on the tape and place the stack of plates upside down Put all plates in the 37°C incubator upside down for 24 hours.
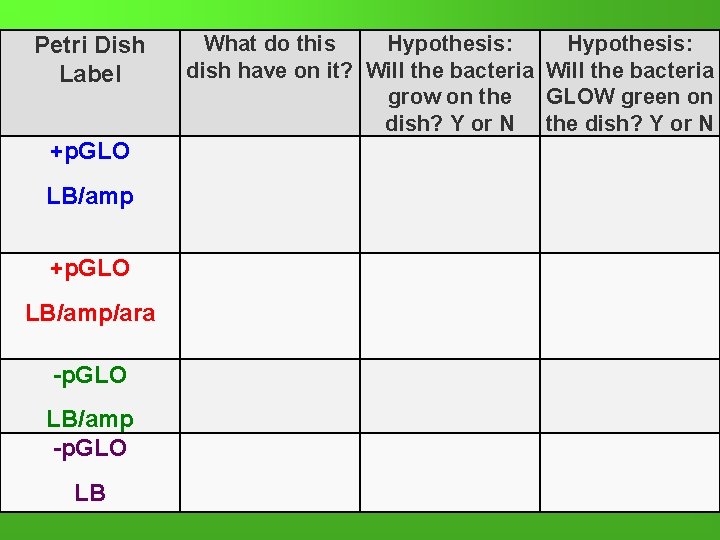
Petri Dish Label +p. GLO LB/amp/ara -p. GLO LB/amp -p. GLO LB What do this Hypothesis: dish have on it? Will the bacteria grow on the GLOW green on dish? Y or N the dish? Y or N
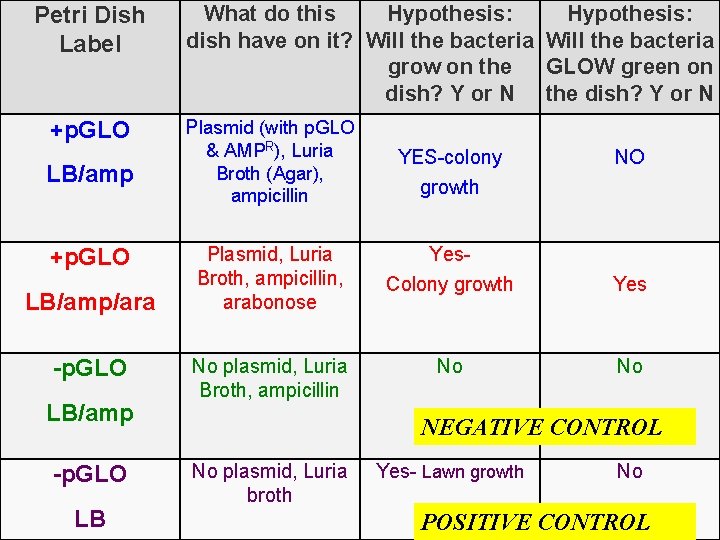
Petri Dish Label +p. GLO LB/amp/ara -p. GLO LB/amp -p. GLO LB What do this Hypothesis: dish have on it? Will the bacteria grow on the GLOW green on dish? Y or N the dish? Y or N Plasmid (with p. GLO & AMPR), Luria Broth (Agar), ampicillin YES-colony Plasmid, Luria Broth, ampicillin, arabonose Yes. Colony growth Yes No plasmid, Luria Broth, ampicillin No No NO growth NEGATIVE CONTROL No plasmid, Luria broth Yes- Lawn growth No POSITIVE CONTROL
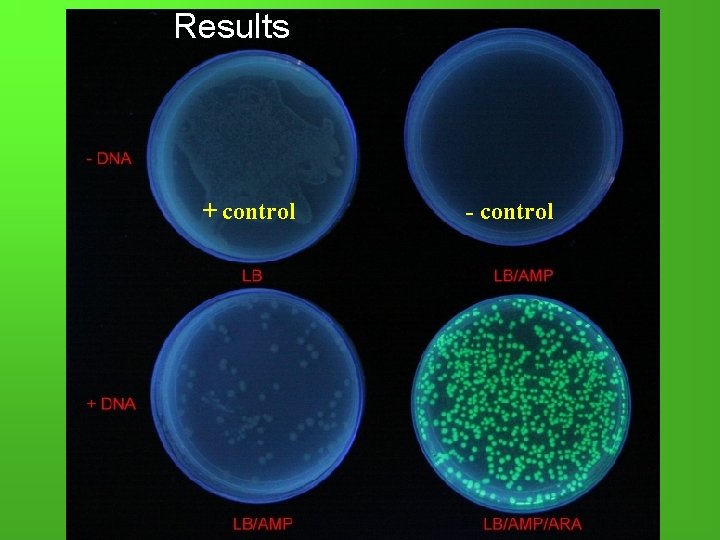
Results + control - control
- Slides: 31