PREFORMULATION STUDIES Considerations before preformulation studies Before initiation

PREFORMULATION STUDIES


Considerations before pre-formulation studies Before initiation of pre-formulation studies, scientist need to consider the following: 1. The available physicochemical data (including chemical structure, different salts, potency relative to the competitive products and the dosage form etc. ) 2. The therapeutic class of the compound, predictable/anticipated dose and the proposed route of drug administration. 3. Supply situation and development schedule (i. e. the time available). 4. The availability/accessibility of a stability-indicating assay. 5. The nature of the information the formulator should have or would like to have.

GOALS 1. To establish the necessary physico-chemical parameters of new drug substances. 2. To determine kinetic rate and stability profile. 3. To establish compatibility with common excipients 4. To provide insight into how drug products should be processed and stored to ensure their quality. 5. To provide a scientific data to support the dosage form design and evaluation of the product efficacy and stability

OBJECTIVES 1. To generate useful data needed in developing stable and safe dosage form that can be manufactures on a commercial scale. 2. To provide in-dept knowledge and understanding of the physical characteristics of a drug molecule prior to dosage form development. 3. To generate useful information on how to design a drug delivery system with good bioavailability.

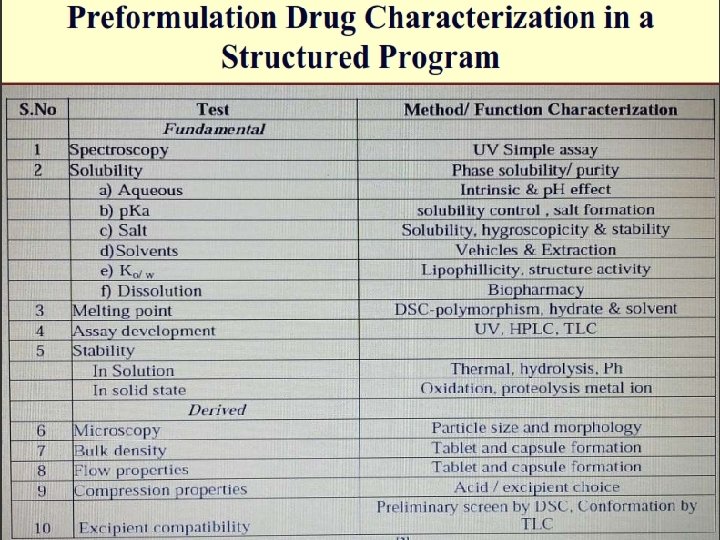
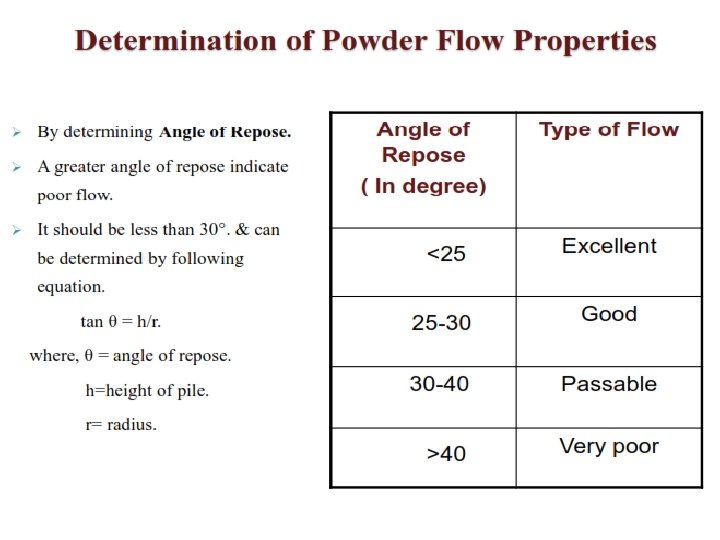
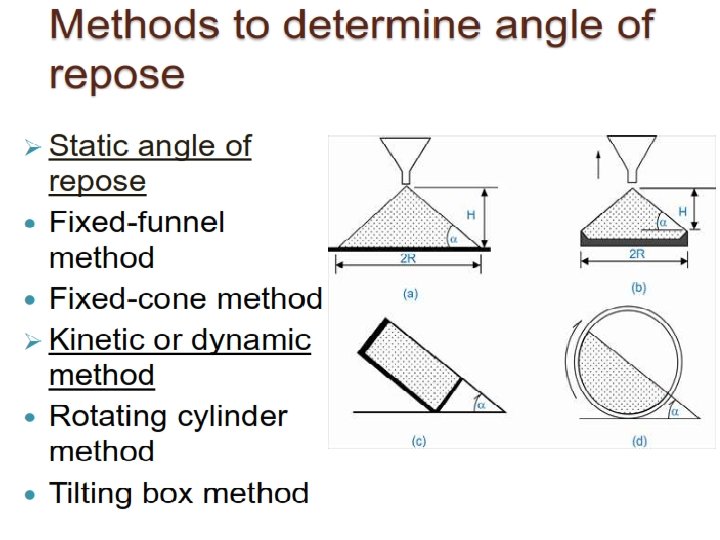
Physicochemical characteristics of Drug substances in Pre-formulation studies 1. 2. Physical Characteristics: Major areas of Pre-formulation Research c. Solubility analysis a. Organoleptic properties 1. Ionization constant/drug p. Ka, 1. Color, odour, taste, 2. Partition co-efficient b. Bulk characterization 3. Solubilization 1. Particle size & surface area 4. Thermal effect 2. Powder flow properties 5. Common ion effect(Ksp) 6. Dissolution. 3. Density 4. Compressibility d. Stability analysis studies 5. Crystallinity 1. Solid-state stability studies 6. Hygroscopicity 2. Solution-state stability studies 7. Polymorphism 3. Drug –excipient compatibility Chemical Characteristics: 1. Hydrolysis 2. Oxidation and reduction 3. Recemization 4. Polymerization
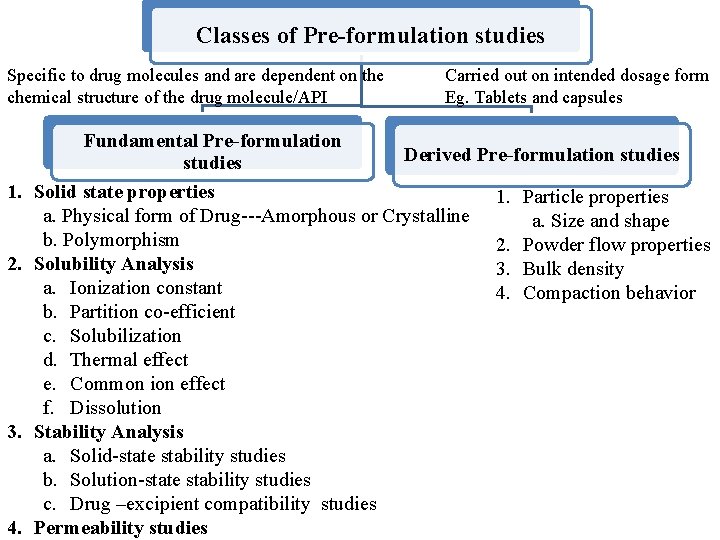
Classes of Pre-formulation studies Specific to drug molecules and are dependent on the chemical structure of the drug molecule/API 1. 2. 3. 4. Carried out on intended dosage form Eg. Tablets and capsules Fundamental Pre-formulation Derived Pre-formulation studies Solid state properties 1. Particle properties a. Physical form of Drug---Amorphous or Crystalline a. Size and shape b. Polymorphism 2. Powder flow properties Solubility Analysis 3. Bulk density a. Ionization constant 4. Compaction behavior b. Partition co-efficient c. Solubilization d. Thermal effect e. Common ion effect f. Dissolution Stability Analysis a. Solid-state stability studies b. Solution-state stability studies c. Drug –excipient compatibility studies Permeability studies

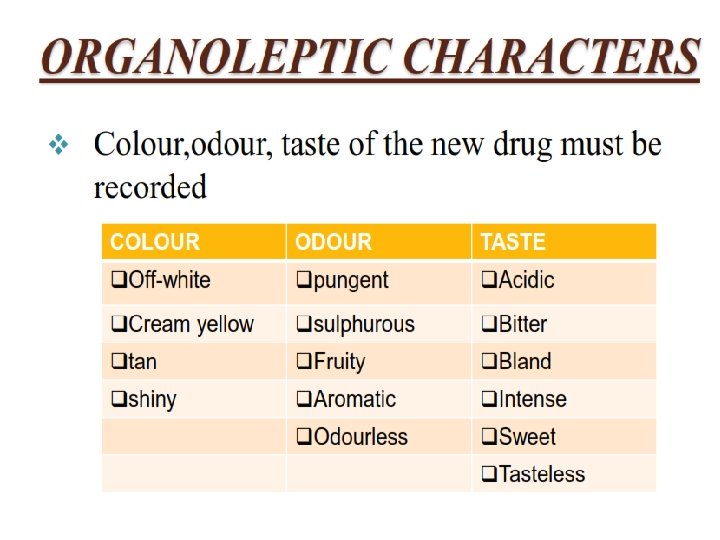

COLOR

ODOUR


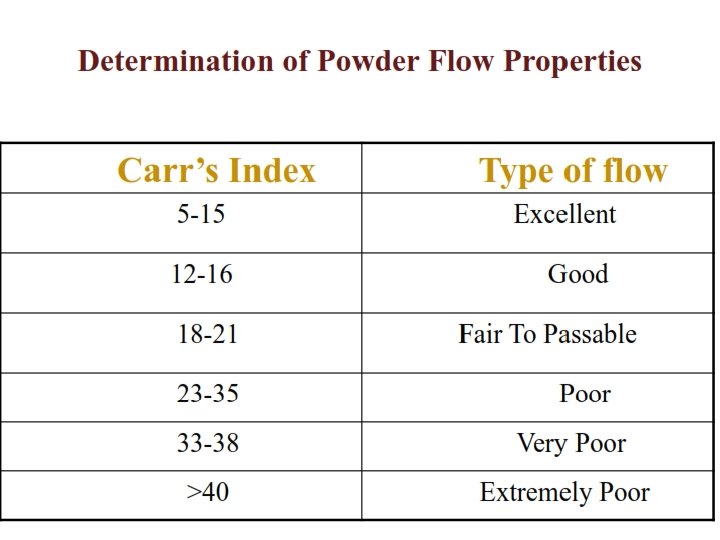
BULK CHARACTERISTICS


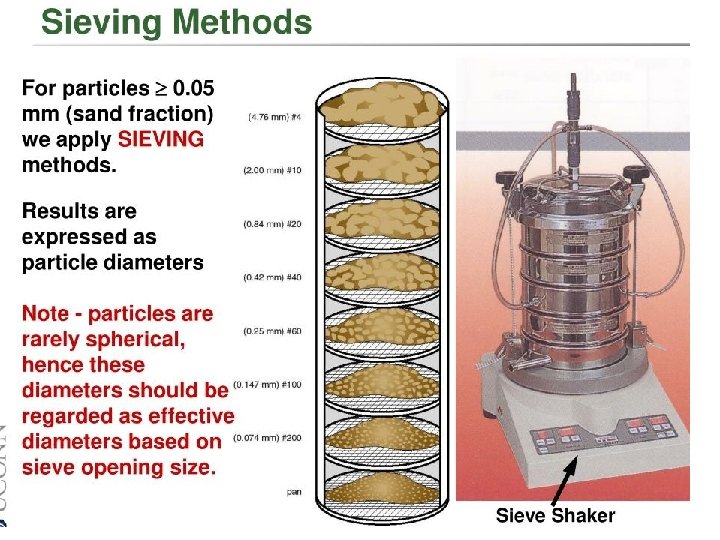
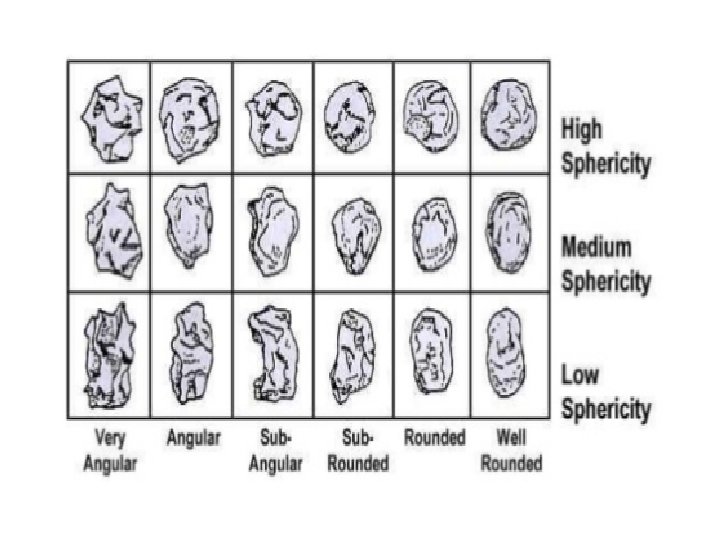
Particle size and shape • Not only effect only physical properties of solid drug but also, in some instances effect on the biopharmaceutical behavior • Size, shape & surface morphology of drug particles affect – Flow property – Dissolution – Chemical reactivity of drugs

Significance of particle size 1. 2. Particle size of drug may affect formulation and product efficacy Certain physical and chemical properties of drug substances are affected by a. The particle size b. Drug dissolution rate c. Content uniformity d. Texture e. Stability f. Flow characteristics g. Sedimentation rates h. Penetrability i. Suspendability 3. Particle size significantly influences the oral absorption profile of certain drugs 4. Satisfactory content uniformity in solid dosage forms depends to a large degree on particle size and the equal distribution of active ingredients throughout the formulation

Or Screening OR Optical Microscopy • Stream Scanning




Stroke’s Law

1. Manual 2. Transmission a. SEM: Scanning Electron microscopy b. TEM: Transmission Electron Microscopy




- Slides: 33