PREFORMULATION Dr Dinesh M Biyani M Pharm Ph

PREFORMULATION Dr. Dinesh M. Biyani, M. Pharm. Ph. D. DBM, DIRPM, Dip TD Associate Professor, SKB College of Pharmacy, Kamptee dineshbiyani 10@gmail. com

AIDING CANDIDATE DRUG SELECTION • • 1. 2. 3. A medicinal product is developed from : Synthetic chemical compound Chemical extracted from natural source Biotechnical process It involves different disciplines to work together Discovery & Development can be broken into 5 stages Strategic research – feasibility study Exploratory research – identification of “chemical lead” (can be had by using Combinatorial chemistry & high throughput screening. ) even this may be tedious, so QSAR and molecular graphics are better Candidate drug selection – chemical lead generates specific chemical compound with optimal, desired potency, specificity, duration and other pharmaceutical aspects. So, for rapid drug development, a compound with preferred pharmaceutical and chemical synthesis properties should be selected.

Preferred drug synthesis & pharmaceutical properties for compounds intended for Oral Solid Dosage Form Development DRUG SYNTHESIS FACTORS i. FORMULATION FACTORS Least complex structure( none or i. Exists as a stable polymorph form few chiral centers) ii. Non hygroscopic ii. Few synthesis steps as possible iii. Crystalline iii. High yields as possible iv. Acceptable solid state stability of iv. None explosive route or safety in v. candidate drug use v. Acceptable bioavailability Commercial availability of vi. Not highly colored or strong building blocks and contract odour (ensure batch manufacturer reproducibility) vi. Low cost of raw material vii. No predicted problem in scale vii. Compatible with key excipients

4. 5. Exploratory Development – how candidate drug is absorbed and metabolized (phase I) Full Development – (phase II and III) DISCOVERY PREFORMULATION BRIDGE DEVELOPMENT

NEED OR IMPORTANCE In past, although low throughput due to animal testing, but proven results Now HTS, due to recombinant enzyme, but it is in vitro So drug may not absorbed in vivo DMSO, DMA etc. are versatile, but toxicity? THIS IS THE CHALLENGE TO PREFORMULATION SCIENTIST

Integrating Discovery and Development require Preformulation scientist to be aware of molecular mechanisms of unfavourable physical properties such as aqueous solubility Best activity, least blood levels 1 aq. Solubility & due to less solubility phase B activity both optimized simultaneously IN VITRO ACTIVITY 2 recommended for development phase A 0 only activity as criterion AQUEOUS SOLUBILITY Lead for further optimization PREFORMULATION SCIENTIST TO DESIGN API ACTIVE AND TRANSPORTABLE THROUGH LIPOPHILIC MEMBRANE

CRITICAL API DECISIONS Once a NCE is selected for development, the molecular form that will be the ACTIVE PHARMACEUTICAL INGREDIENT will be critical milestone because all subsequent development will be affected by this decision. For Preformulation, physical characterization decision should be like BALANCE PREFORMULATION SOLID STATE: DISSOLUTION PROPERTIES MATERIAL CONSISTANCY: MFG. & STORAGE CONDITIONS Eg. Amorphous drug (greater solubility but conversion to crystalline in presence of heat, moisture and with time) HCl and Na salt (good solubility less toxicity but physical & chemical instability due to moisture and heat) Higher lipophilicity (good for absorption but may have dissolution problems)

Preformulation is the physicochemical characterization of the solid and solution properties of compounds for subsequent formulation of a stable and biopharmaceutically suitable dosage form. Preformulation typically begins in the earlier stages of Lead optimization (LO), however, there may be some involvement during Lead identification (LI) In LI, it supports to the medicinal chemist for solid state screening to ensure the consistency in polymorphism. During last 3 - 6 months of LO (of total 2 years) total compounds are narrowed to around 3 for final nomination. This is known as prenomination.
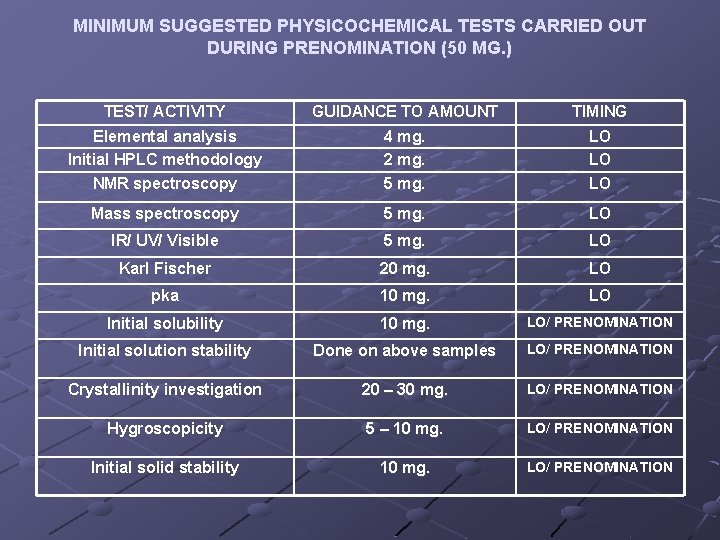
MINIMUM SUGGESTED PHYSICOCHEMICAL TESTS CARRIED OUT DURING PRENOMINATION (50 MG. ) TEST/ ACTIVITY GUIDANCE TO AMOUNT TIMING Elemental analysis Initial HPLC methodology NMR spectroscopy 4 mg. 2 mg. 5 mg. LO LO LO Mass spectroscopy 5 mg. LO IR/ UV/ Visible 5 mg. LO Karl Fischer 20 mg. LO pka 10 mg. LO Initial solubility 10 mg. LO/ PRENOMINATION Initial solution stability Done on above samples LO/ PRENOMINATION Crystallinity investigation 20 – 30 mg. LO/ PRENOMINATION Hygroscopicity 5 – 10 mg. LO/ PRENOMINATION Initial solid stability 10 mg. LO/ PRENOMINATION
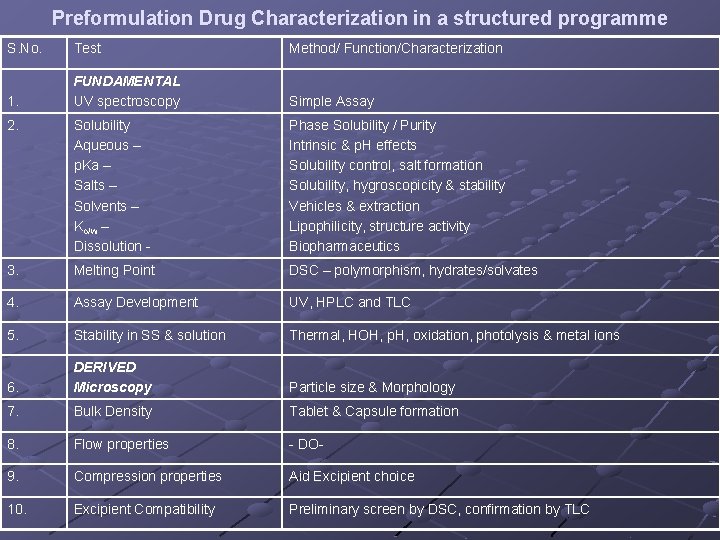
Preformulation Drug Characterization in a structured programme S. No. Test Method/ Function/Characterization 1. FUNDAMENTAL UV spectroscopy Simple Assay 2. Solubility Aqueous – p. Ka – Salts – Solvents – Ko/w – Dissolution - Phase Solubility / Purity Intrinsic & p. H effects Solubility control, salt formation Solubility, hygroscopicity & stability Vehicles & extraction Lipophilicity, structure activity Biopharmaceutics 3. Melting Point DSC – polymorphism, hydrates/solvates 4. Assay Development UV, HPLC and TLC 5. Stability in SS & solution Thermal, HOH, p. H, oxidation, photolysis & metal ions 6. DERIVED Microscopy Particle size & Morphology 7. Bulk Density Tablet & Capsule formation 8. Flow properties - DO- 9. Compression properties Aid Excipient choice 10. Excipient Compatibility Preliminary screen by DSC, confirmation by TLC
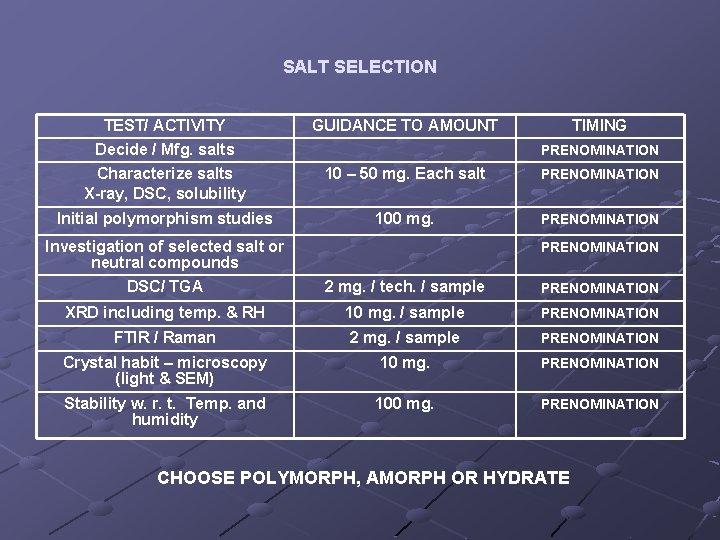
SALT SELECTION TEST/ ACTIVITY Decide / Mfg. salts GUIDANCE TO AMOUNT TIMING Characterize salts X-ray, DSC, solubility 10 – 50 mg. Each salt PRENOMINATION Initial polymorphism studies 100 mg. PRENOMINATION Investigation of selected salt or neutral compounds PRENOMINATION DSC/ TGA 2 mg. / tech. / sample PRENOMINATION XRD including temp. & RH 10 mg. / sample PRENOMINATION FTIR / Raman 2 mg. / sample PRENOMINATION Crystal habit – microscopy (light & SEM) 10 mg. PRENOMINATION Stability w. r. t. Temp. and humidity 100 mg. PRENOMINATION CHOOSE POLYMORPH, AMORPH OR HYDRATE
- Slides: 11