Predicting Reactions SINGLE DISPLACEMENT GENERAL EQUATION AB C

Predicting Reactions
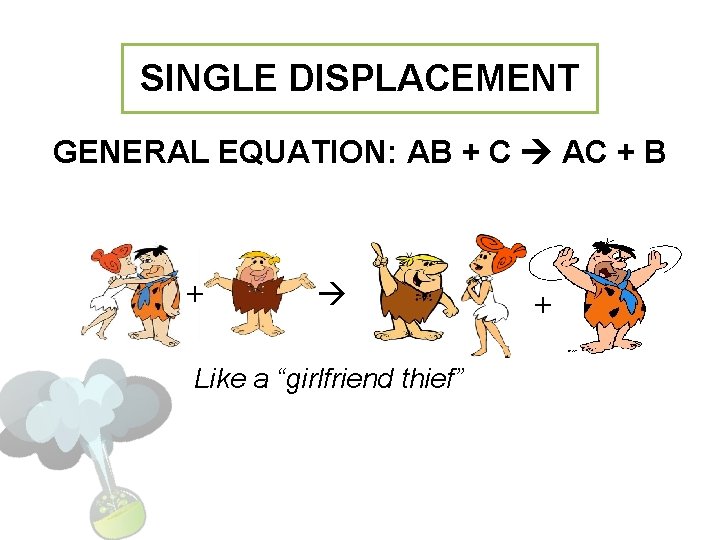
SINGLE DISPLACEMENT GENERAL EQUATION: AB + C AC + B + Like a “girlfriend thief” +

Helpful Hints • Metals replace metals and hydrogen • Halogens replace halogens • Metals + H 2 O metal. OH + H 2
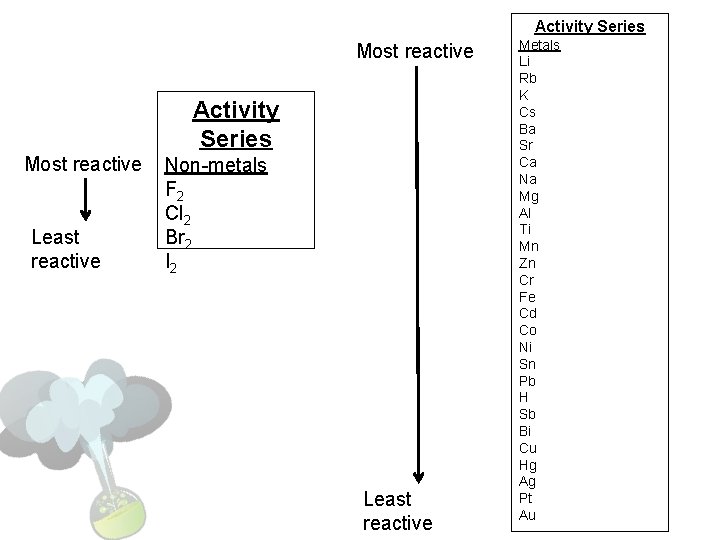
Activity Series Most reactive Least reactive Non-metals F 2 Cl 2 Br 2 I 2 Least reactive Metals Li Rb K Cs Ba Sr Ca Na Mg Al Ti Mn Zn Cr Fe Cd Co Ni Sn Pb H Sb Bi Cu Hg Ag Pt Au

Steps to Predicting Single Displacement Rxns 1. Look at the reactants given and determine if the single element is a metal or a halogen 2. Look at the compound given in the reactants, and find the same type of element (metal or halogen) identified in step 1 3. Find both elements on the reactivity series

4. If the element identified in step 1 is higher on the reactivity series than the element in step 2, then they will switch places to form new products 5. Look at the products formed • If the single element is a diatomic molecule, it needs a subscript of 2 • If your compound is not neutral; look at the charges and add subscripts to make it neutral
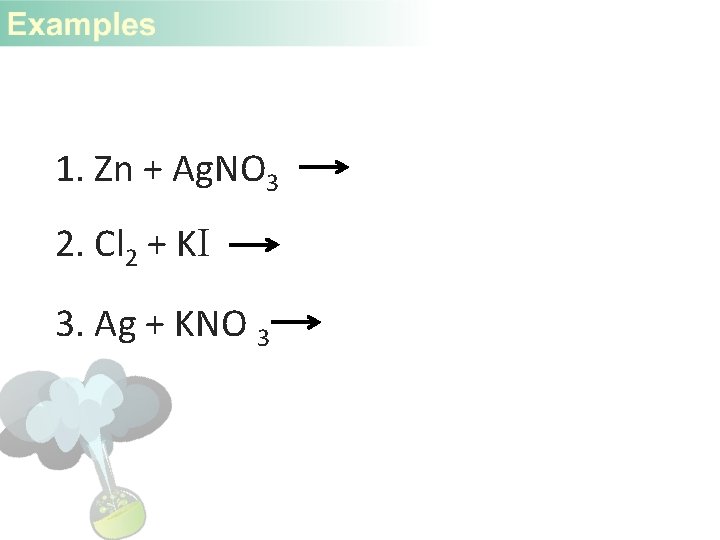
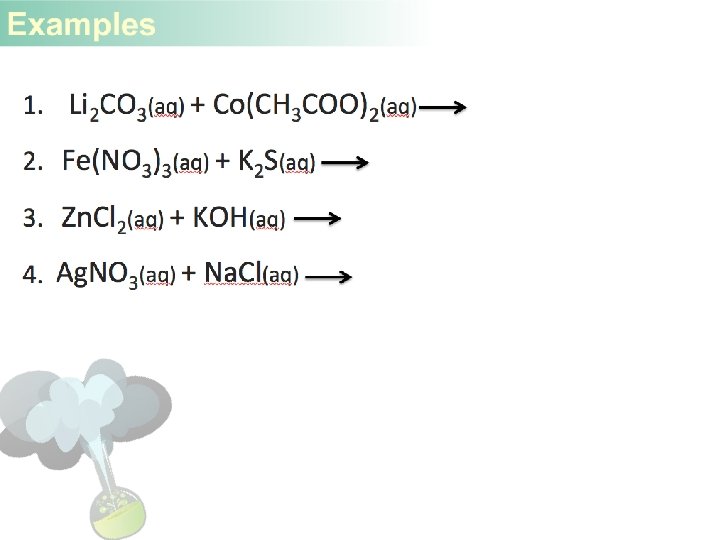
1. Zn + Ag. NO 3 2. Cl 2 + KI 3. Ag + KNO 3


Steps to Predicting Double Displacement Rxns 1. Rewrite the cations from the reactants on the product side 2. Switch the anions, make sure new compounds are neutral!! 3. Use the solubility chart to identify the state of the products

Driving forces Formation of… Precipitate How do you recognize it? Results in the formation of an insoluble product; use the solubility chart Gas You must memorize the combinations that decompose into gases (there are 4). You must also memorize the gaseous products that form. Water Reaction between an acid and base that results in a salt and water. You must memorize the short list of strong acids and strong bases so you will recognize all the weak acids and bases that dissolve, but do not dissociate into ions.

PRECIPITATE RXNS

Solubility is the property of a substance (solute) to dissolve in another substance (solvent).

Examples Classify the following ionic compounds as soluble or insoluble: (a) silver sulfate (Ag 2 SO 4) (b) calcium carbonate (Ca. CO 3) (c) sodium phosphate (Na 3 PO 4).

14
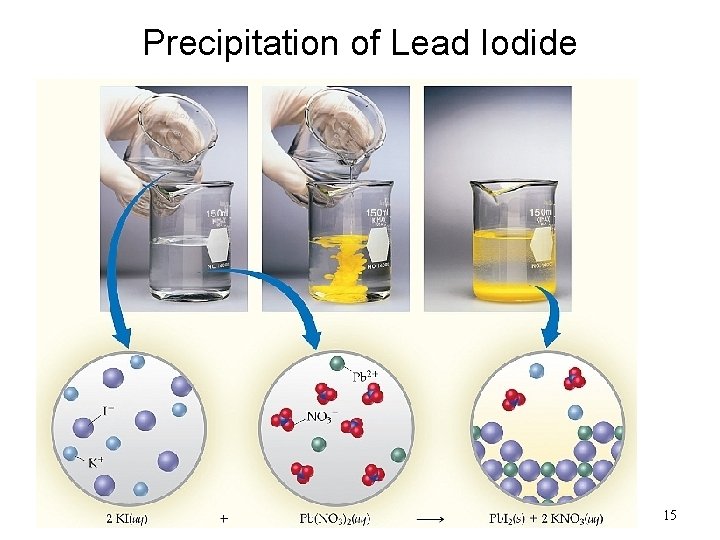
Precipitation of Lead Iodide 15

ACID-BASE RXNS acid + base salt + water

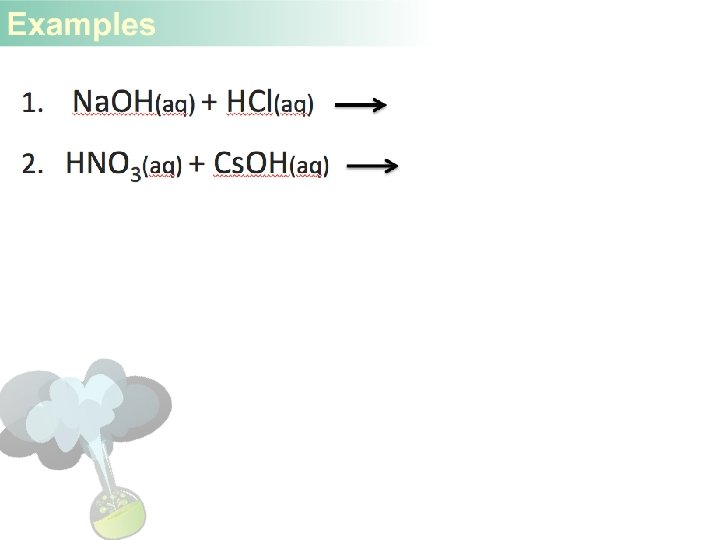
STRONG ACIDS STRONG BASES Strong acids/bases are strong electrolytes and will completely dissociate in water.

Writing Net Ionic Equations 1. Write the balanced molecular equation. 2. Write the ionic equation showing the strong electrolytes completely dissociated into cations and anions. 3. Cancel the spectator ions on both sides of the ionic equation. 4. Check that charges and number of atoms are balanced in the net ionic equation. 20

Pb. I 2 Precipitate Reaction precipitate Pb(NO 3)2 (aq) + 2 KI (aq) Pb. I 2 (s) + 2 KNO 3 (aq) molecular equation Pb 2+(aq) + 2 NO 3 -(aq) + 2 K+(aq) + 2 I-(aq) Pb. I 2(s) + 2 K+(aq) + 2 NO 3 -(aq) ionic equation Pb 2+(aq) + 2 I-(aq) Pb. I 2 (s) net ionic equation K+ and NO 3 - are spectator ions 21
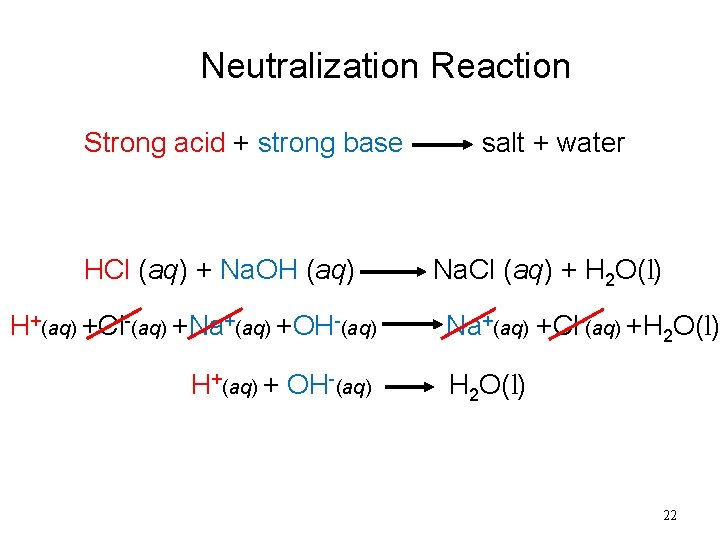
Neutralization Reaction Strong acid + strong base HCl (aq) + Na. OH (aq) H+(aq) +Cl-(aq) +Na+(aq) +OH-(aq) H+(aq) + OH-(aq) salt + water Na. Cl (aq) + H 2 O(l) Na+(aq) +Cl-(aq) +H 2 O(l) 22
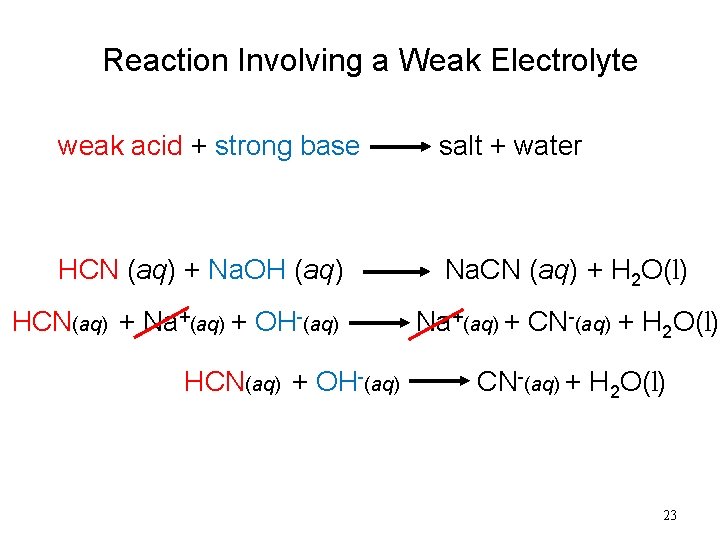
Reaction Involving a Weak Electrolyte weak acid + strong base salt + water HCN (aq) + Na. OH (aq) Na. CN (aq) + H 2 O(l) HCN(aq) + Na+(aq) + OH-(aq) HCN(aq) + OH-(aq) Na+(aq) + CN-(aq) + H 2 O(l) 23
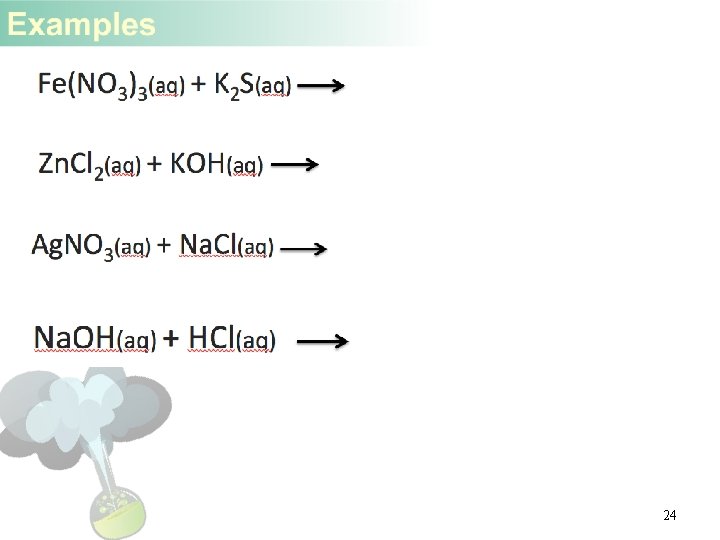
24

Review of Concepts Which of the diagrams here accurately describes the reaction between Ca(NO 3)2(aq) and Na 2 CO 3(aq)? For simplicity, only the Ca 2+ (yellow) and CO 32− (blue) ions are shown.

GAS FORMING RXNS Certain salts react with acids to produce gaseous products, you need to recognize these products as gases, and gases do not ionize

Secondary Decomposition If you get one of these as a product in your molecular equation, they immediately breakdown
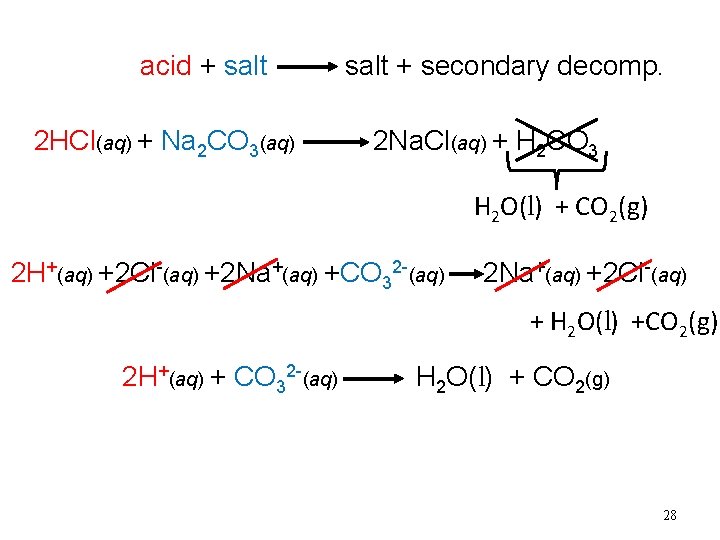
acid + salt 2 HCl(aq) + Na 2 CO 3(aq) salt + secondary decomp. 2 Na. Cl(aq) + H 2 CO 3 H 2 O(l) + CO 2(g) 2 H+(aq) +2 Cl-(aq) +2 Na+(aq) +CO 32 -(aq) 2 Na+(aq) +2 Cl-(aq) + H 2 O(l) +CO 2(g) 2 H+(aq) + CO 32 -(aq) H 2 O(l) + CO 2(g) 28
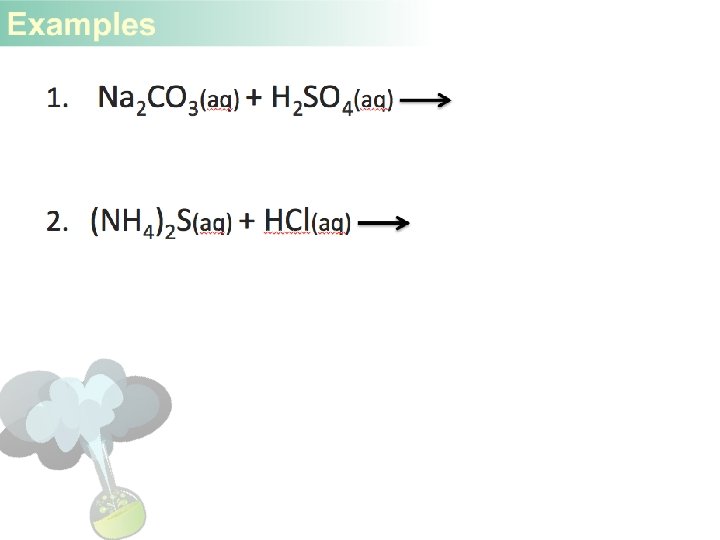
- Slides: 29