Predicting Products of Reactions Double and Single Replacement

Predicting Products of Reactions Double and Single Replacement

Double replacement • Two main types a. formation of a precipitate b. acid/base reaction (produces a salt plus water) (c. or both!) • use solubility rules and electrolytic properties • only break apart into ions if soluble, in solution, and a strong electrolyte

• Must have a driving force that removes ions from the solution – a precipitate forms and/or – a gas forms and/or – a weak/nonelectrolyte (water usually) forms

• Formation of a precipitate – A precipitate is an insoluble compound (a solid) – Use solubility rules to determine if one or both of the products is a precipitate • Example 3 Ag. NO 3 (aq) + K 3 PO 4 (aq) Ag 3 PO 4 (s) + 3 KNO 3 (aq) insoluble precipitate soluble Net ionic eqn: 3 Ag+(aq) + PO 4 -3(aq) Ag 3 PO 4(s)

• Formation of a gas – Must know common gases • If you get H 2 CO 3 when you do the double replacement it really gives H 2 O + CO 2 (g) • If you get H 2 SO 3 when you do the double replacement it really gives H 2 O + SO 2 (g) • H 2 S is a gas

• For example Na 2 CO 3(aq)+ H 2 SO 4(aq) Na 2 SO 4(aq)+ H 2 CO 3(aq) REALLY IS: Na 2 CO 3(aq)+H 2 SO 4(aq) Na 2 SO 4(aq)+H 2 O(l)+CO 2(g) Net ionic eqn: CO 3 -2(aq) + 2 H+(aq) H 2 O(l) + CO 2(g)

• Formation of a nonelectrolyte – Water is a common nonelectrolyte and is a result of an acid-base reaction HCl(aq) + Na. OH(aq) Na. Cl(aq) + H 2 O(l) Net ionic eqn: H+ (aq)+ OH- (aq) H 2 O(l)

Redox Reactions • Some synthesis, some decomposition, single replacement, and other reactions are oxidation-reductions • OXIDATION-REDUCTION REACTIONS involve electron transfer

• Terms to Know: • • OIL RIG – oxidation is loss, reduction is gain (of electrons) • Oxidation – the loss of electrons, increase in charge (becomes more positive) • Reduction – the gain of electrons, reduction of charge (becomes more negative) • Oxidation number – the assigned charge on an atom • Oxidizing agent (OA) – the species that is reduced and thus causes oxidation • Reducing agent (RA) – the species that is oxidized and thus causes reduction • *note that some in some reactions, the same species can be oxidized and reduced; these are called disproportionation reactions

Rules for Assigning Oxidation States • 1. The oxidation state of an atom in an element is ZERO including allotropes [i. e. N 2, P 4, S 8]. • 2. The oxidation state of a monatomic ion is the same as its charge. • 3. In its compounds, fluorine is always assigned an oxidation state of -1. • 4. Oxygen is usually assigned an oxidation state of -2 in its covalent compounds, such as CO, CO 2, SO 2, and SO 3. – Exceptions to this rule include peroxides (compounds containing the O 22 - group), where each oxygen is assigned an oxidation state of -1, as in hydrogen peroxide (H 2 O 2), – and OF 2 in which oxygen is assigned a +2 oxidation state. • 5. In its covalent compounds with nonmetals and acids, hydrogen is assigned an oxidation state of +1. – Metal hydrides are an exception; H is at the end of the chemical formula since it has an oxidation state of -1. (such as Na. H) • 6. The sum of the oxidation states must be zero for an electrically neutral compound. For a polyatomic ion, the sum of the oxidation states must equal the charge of the ion.

• There can be non-integer oxidation states like in Fe 3 O 4. There’s a -8 for the 4 oxygens divided across 3 iron ions, therefore Fe’s charge is Fe 8/3+

Single Replacement Redox • How can we predict if a single replacement reaction will occur? – Reading the reduction potential chart • elements that have the most positive reduction potentials are easily reduced (in general, non-metals) • elements that have the least positive reduction potentials are easily oxidized (in general, metals) • Can also be used as an activity series. Metals having less positive reduction potentials are more active and will replace metals with more positive potentials. • The MORE POSITIVE reduction potential gets to indeed be reduced IF you are trying to set up a cell that can act as a battery.
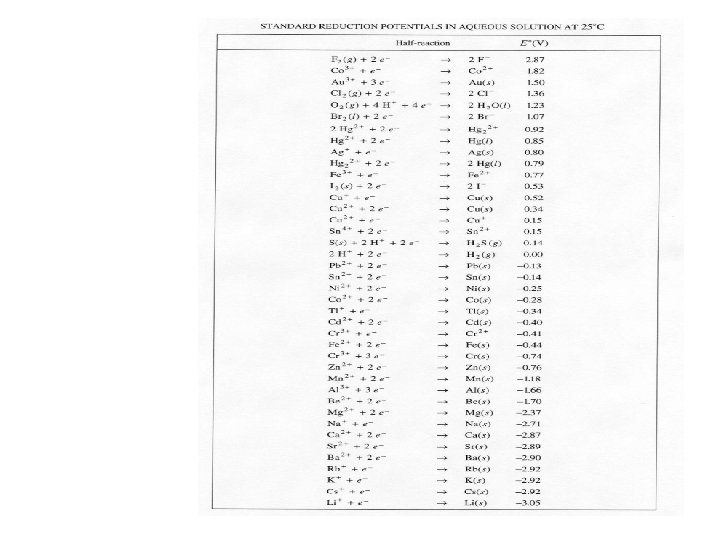

• For example, Will the following reaction occur? Mg(s) + 2 KCl(aq) 2 K(s) + Mg. Cl 2(aq) Step 1: Determine which element is reduced by assigning oxidation numbers before and after Mg(s) + 2 KCl(aq) 2 K(s) + Mg. Cl 2(aq) 0 +1 -1 0 +2 -1 So K was reduced b/c it went from +1 to 0

• Step 2: Look up the reduction potentials of the elements whose oxidation #’s changed K= -2. 92 Mg=-2. 37 • Step 3: Determine if the element with the most +reduction potential was reduced – NO! K was reduced in the reaction as written, but Mg has the most positive reduction potential! – K cannot be reduced by Mg, but Mg could be reduced by K • Step 4: Does this reaction occur? – NO! (but note that means that the reverse reaction would occur!)
- Slides: 15