Precipitation Reactions Graphic Wikimedia Commons User Tubifex Double

Precipitation Reactions Graphic: Wikimedia Commons User Tubifex

Double Replacement Reactions The ions of two compounds exchange places in an aqueous solution to form two new compounds. AX + BY AY + BX One of the compounds formed is usually a precipitate (an insoluble solid), an insoluble gas that bubbles out of solution, or a molecular compound, usually water.

Double replacement forming a precipitate… Lead(II) nitrate + potassium iodide lead(II) iodide + potassium nitrate Double replacement (ionic) equation Pb(NO 3)2(aq) + 2 KI(aq) Pb. I 2(s) + 2 KNO 3(aq) Complete ionic equation shows compounds as aqueous ions Pb 2+(aq) + 2 NO 3 -(aq) + 2 K+(aq) +2 I-(aq) Pb. I 2(s) + 2 K+(aq) + 2 NO 3 -(aq) Net ionic equation eliminates the spectator ions Pb 2+(aq) + 2 I-(aq) Pb. I 2(s)

Solubility Rules – AP Chemistry All sodium, potassium, ammonium, and nitrate salts are soluble in water. Memorization of other “solubility rules” is beyond the scope of this course and the AP Exam. Therefore, the following slides are only for your amusement, and will not be tested
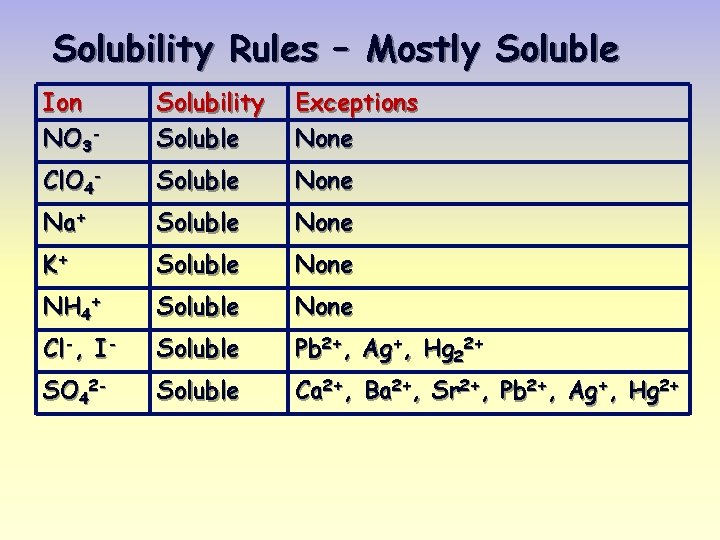
Solubility Rules – Mostly Soluble Ion NO 3 - Solubility Soluble Exceptions None Cl. O 4 - Soluble None Na+ Soluble None K+ Soluble None NH 4+ Soluble None Cl-, I- Soluble Pb 2+, Ag+, Hg 22+ SO 42 - Soluble Ca 2+, Ba 2+, Sr 2+, Pb 2+, Ag+, Hg 2+
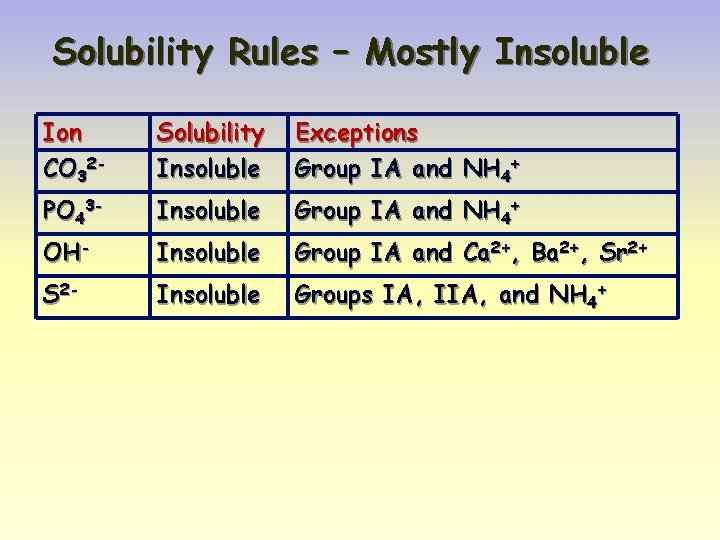
Solubility Rules – Mostly Insoluble Ion CO 32 - Solubility Insoluble Exceptions Group IA and NH 4+ PO 43 - Insoluble Group IA and NH 4+ OH- Insoluble Group IA and Ca 2+, Ba 2+, Sr 2+ S 2 - Insoluble Groups IA, IIA, and NH 4+
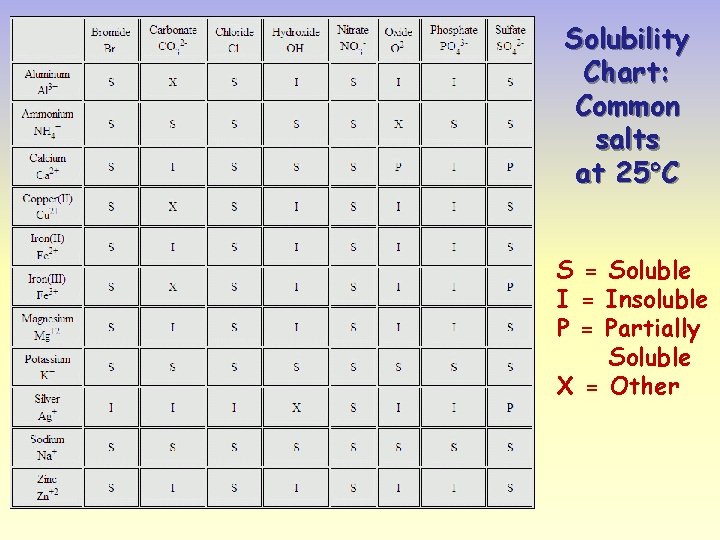
Solubility Chart: Common salts at 25 C S = Soluble I = Insoluble P = Partially Soluble X = Other
- Slides: 7