Precipitation Reactions Graphic Wikimedia Commons User Tubifex Double

Precipitation Reactions Graphic: Wikimedia Commons User Tubifex

Double Replacement Reactions The ions of two compounds exchange places in an aqueous solution to form two new compounds. AX + BY AY + BX One of the compounds formed is usually a precipitate (an insoluble solid), an insoluble gas that bubbles out of solution, or a molecular compound, usually water.

Double replacement forming a precipitate… Lead(II) nitrate + potassium iodide lead(II) iodide + potassium nitrate Double replacement (ionic) equation Pb(NO 3)2(aq) + 2 KI(aq) Pb. I 2(s) + 2 KNO 3(aq) Complete ionic equation shows compounds as aqueous ions Pb 2+(aq) + 2 NO 3 -(aq) + 2 K+(aq) +2 I-(aq) Pb. I 2(s) + 2 K+(aq) + 2 NO 3 -(aq) Net ionic equation eliminates the spectator ions Pb 2+(aq) + 2 I-(aq) Pb. I 2(s)
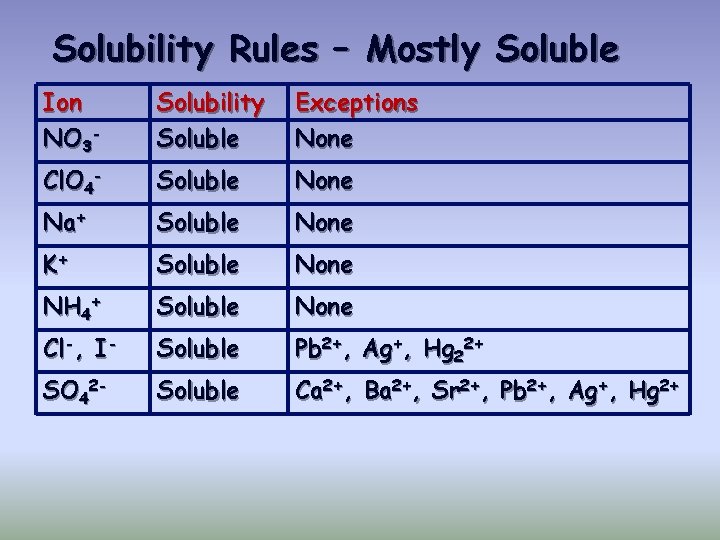
Solubility Rules – Mostly Soluble Ion NO 3 - Solubility Soluble Exceptions None Cl. O 4 - Soluble None Na+ Soluble None K+ Soluble None NH 4+ Soluble None Cl-, I- Soluble Pb 2+, Ag+, Hg 22+ SO 42 - Soluble Ca 2+, Ba 2+, Sr 2+, Pb 2+, Ag+, Hg 2+
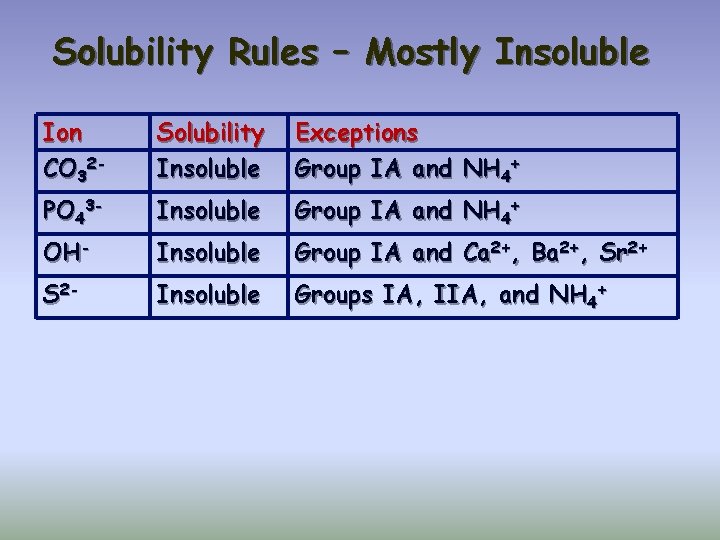
Solubility Rules – Mostly Insoluble Ion CO 32 - Solubility Insoluble Exceptions Group IA and NH 4+ PO 43 - Insoluble Group IA and NH 4+ OH- Insoluble Group IA and Ca 2+, Ba 2+, Sr 2+ S 2 - Insoluble Groups IA, IIA, and NH 4+
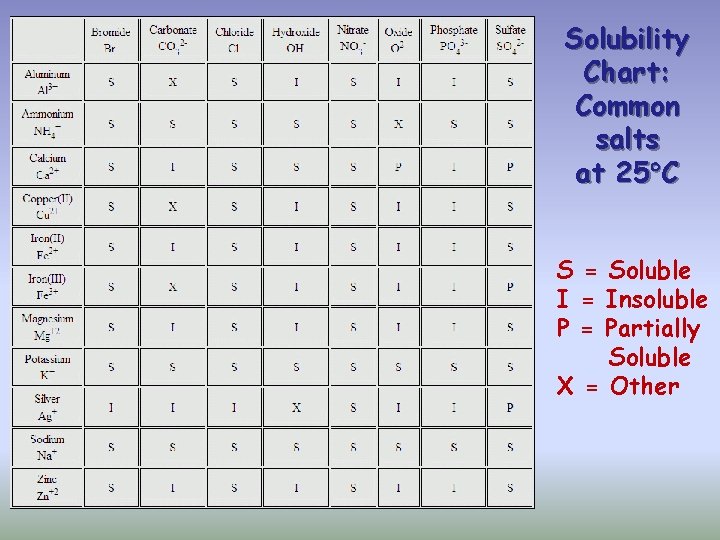
Solubility Chart: Common salts at 25 C S = Soluble I = Insoluble P = Partially Soluble X = Other
- Slides: 6