Prebiological synthesis and evolution of macromolecules Sergey Varfolomeyev

Prebiological synthesis and evolution of macromolecules Sergey Varfolomeyev N. M. Emanuel Institute of Biochemical Physics, RAS, Moscow, Russia sdvarf@sky. chph. ras. ru

Origin of Life = Origin of self Proliferating Macromolecules

Life phenomena from point of view of polymer chemistry n Polycondensation – the same reaction for all biopolymers n Three-functional monomers n Immense decrease of number of macromolecules comparing to the number of possible variants

Decrease of macromolecules number Biological systems implemented the extreme low part of possible macromolecular structures Human genome 3. 104 genes Possible variants of protein structures (20 amino acids, 100 monomers in chain) 20 100 ~ 10 130
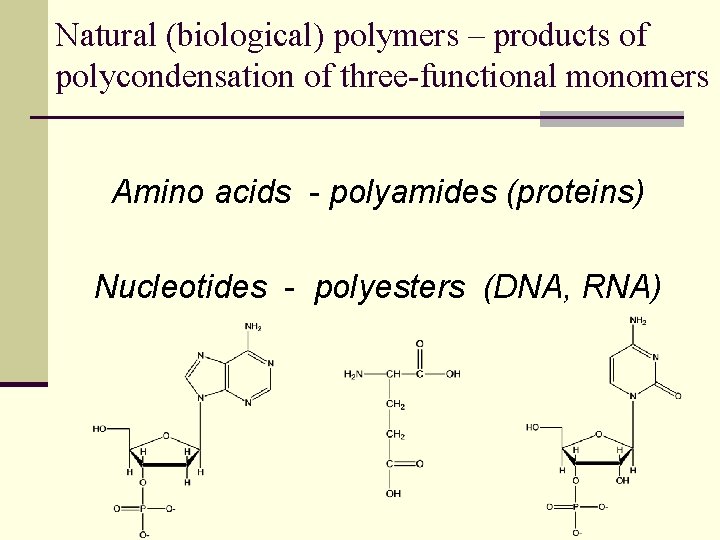
Natural (biological) polymers – products of polycondensation of three-functional monomers Amino acids - polyamides (proteins) Nucleotides - polyesters (DNA, RNA)

Copolymerization of threefunctional monomers -H 2 O

All biomacromolecules (products of polycondensation) are unstable in water

Is it possible to have polycondensation process in water? Thermocycling

Thermocycle – natural global cycle ΔT - 200 - +200 - 20 - + 150 0 - + 120 q Polymerization at high temperatures 120 – 200 OC (statistical number of polymers with different structures and size) q Reduction of temperature, solubilization in water condensate q Macromolecular selection of monomers by polymer matrix q Temperature increasing, polymerization, monomers on matrix (template), formation of partly complementary chain

Natural (biological) polymers – products of polycondensation of three-functional monomers Amino acids - polyamides (proteins) Nucleotides - polyesters (DNA, RNA)


Copolymerization of threefunctional monomers Kinetic theory describing and explain the dramatic reduction of the number of polymer structures in systems. S. D. Varfolomeyev, Kinetic models of the prebiological evolution of macromolecules. Thermocycle as motive force of the process, Mendeleev Communications, 2007, 17, 7 -9

Copolymerization of threefunctional monomers

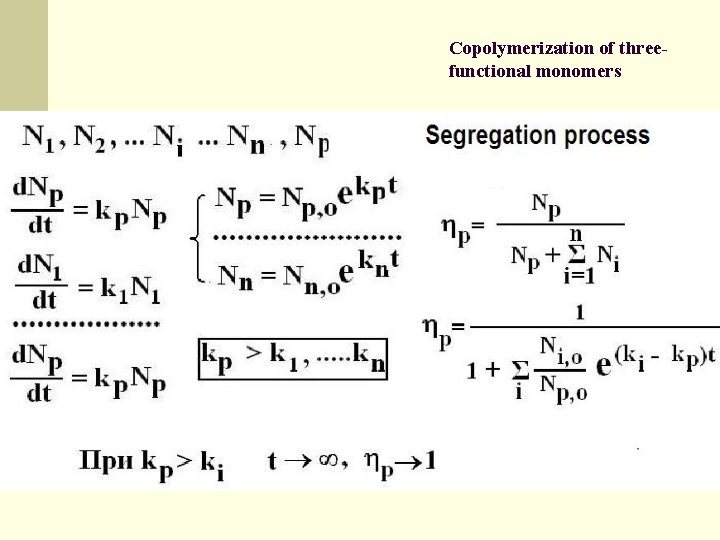
Copolymerization of threefunctional monomers
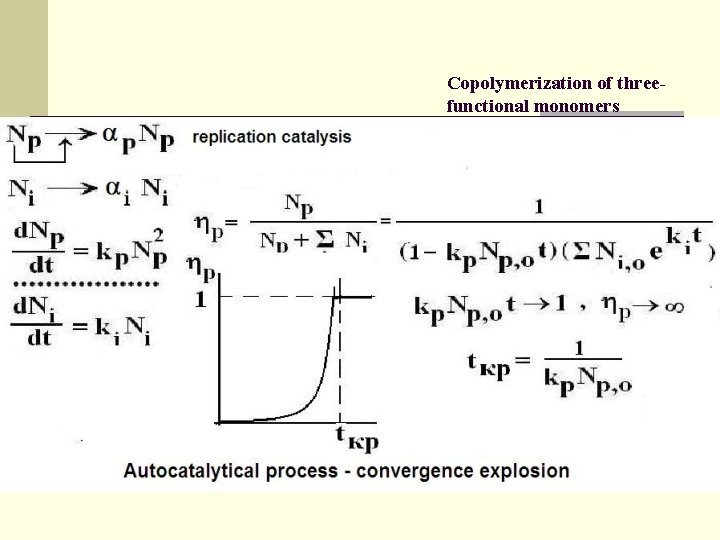
Copolymerization of threefunctional monomers

Copolymerization of threefunctional monomers
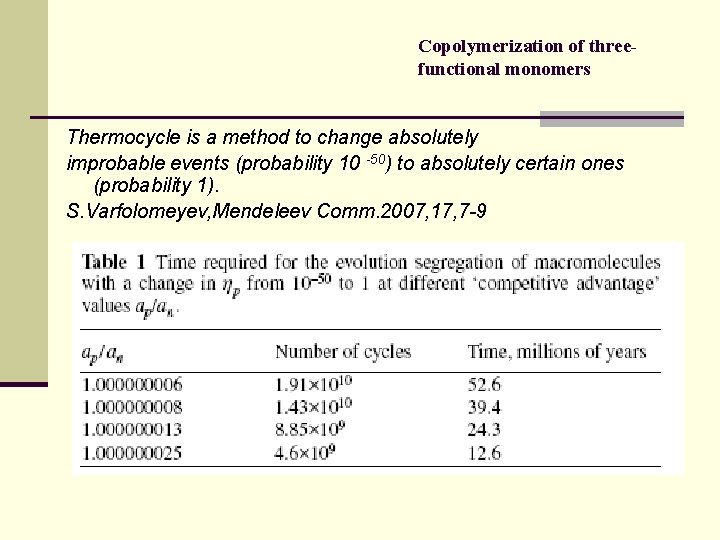
Copolymerization of threefunctional monomers Thermocycle is a method to change absolutely improbable events (probability 10 -50) to absolutely certain ones (probability 1). S. Varfolomeyev, Mendeleev Comm. 2007, 17, 7 -9

Adsorption of monomers over polymer n Formation of a complex between monomer and polymer tends to stabilize macromolecule against thermal destruction and hydrolysis n Synthesis of new polymer is selective
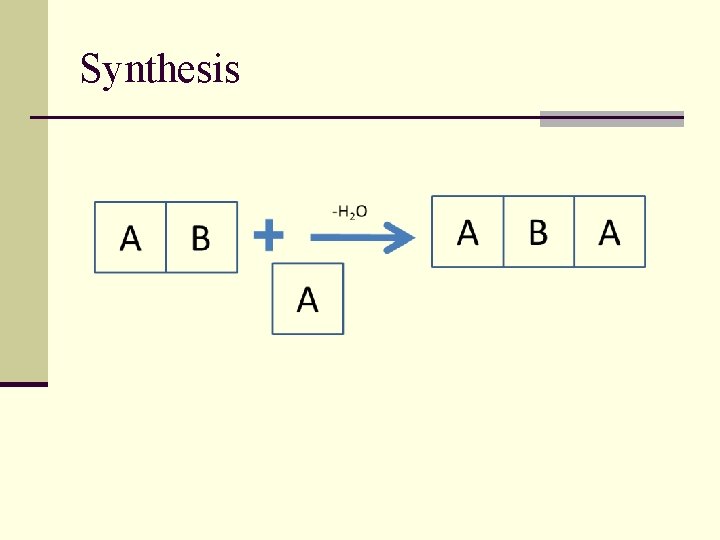
Synthesis
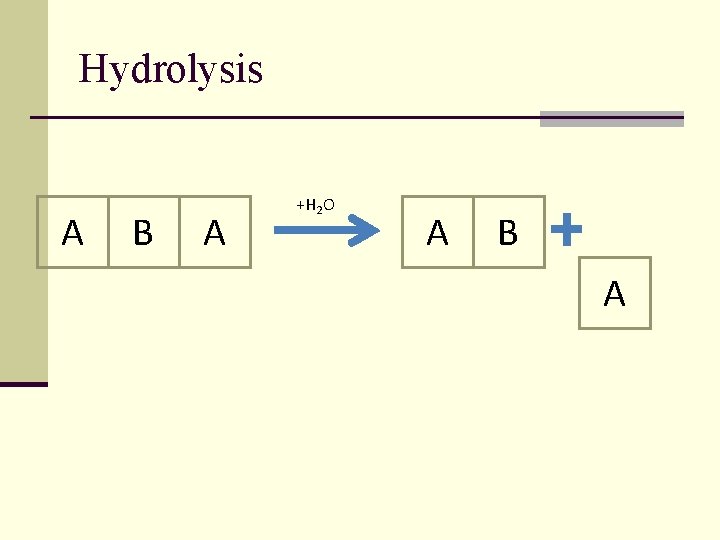
Hydrolysis A B A +H 2 O A B A
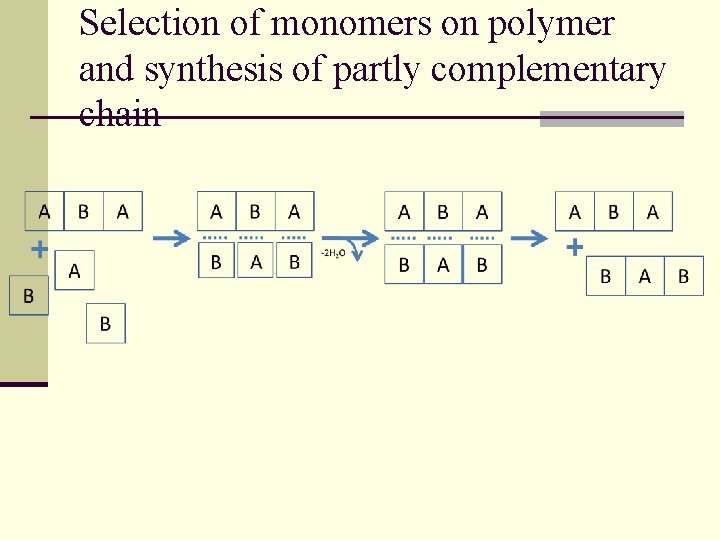
Selection of monomers on polymer and synthesis of partly complementary chain
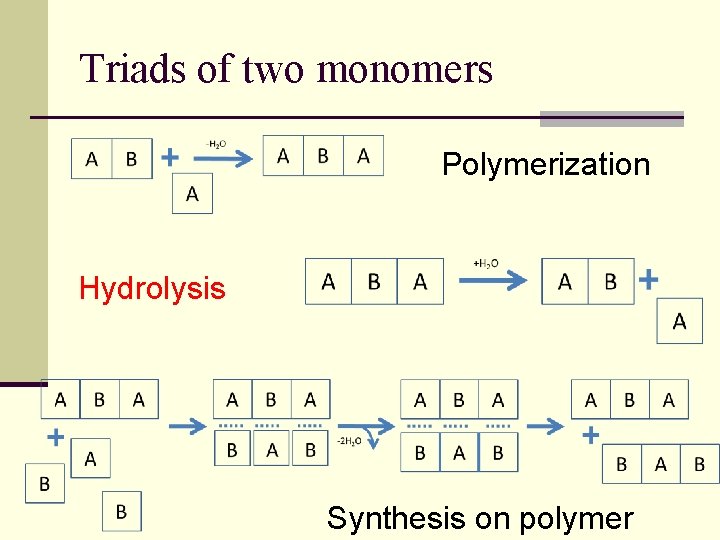
Triads of two monomers Polymerization Hydrolysis Synthesis on polymer

Reactions

Equations

Equations
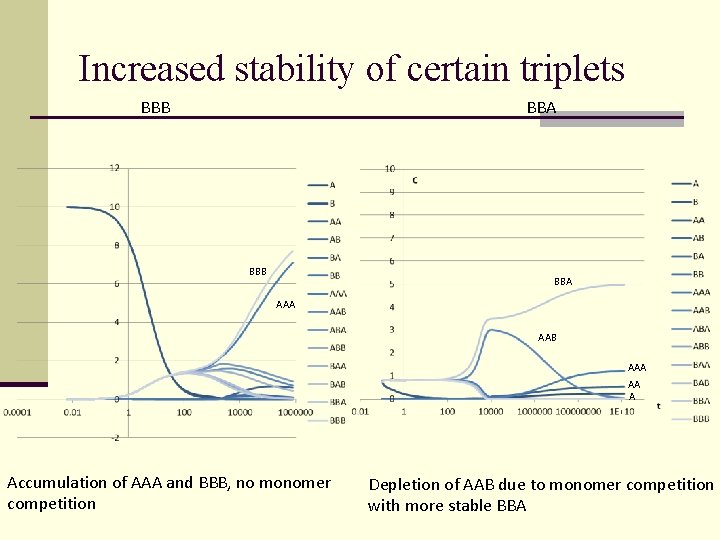
Increased stability of certain triplets BBB BBA AAA AAB AAA AA A Accumulation of AAA and BBB, no monomer competition Depletion of AAB due to monomer competition with more stable BBA

Selection principles (competitive advantages) n. Greater thermodynamical stability n. Greater hydrolysis resistance n. Velocity of catalytic processes: The most stable and «quick» wins!

Experiments
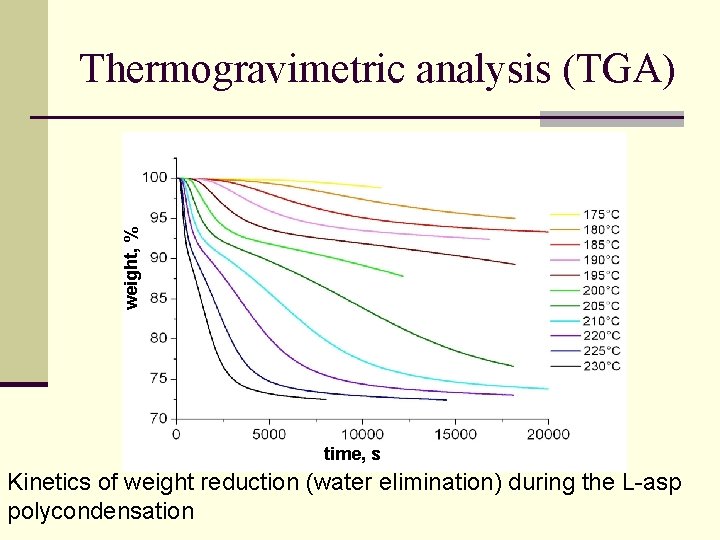
weight, % Thermogravimetric analysis (TGA) time, s Kinetics of weight reduction (water elimination) during the L-asp polycondensation
Unique for Russia combined mass-spectrometer ICR + ionic trap Finnigan LTQ-FT
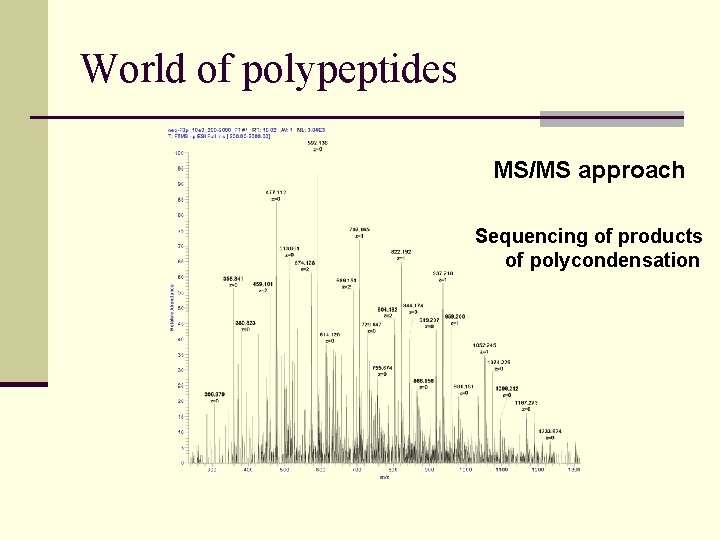
World of polypeptides MS/MS approach Sequencing of products of polycondensation
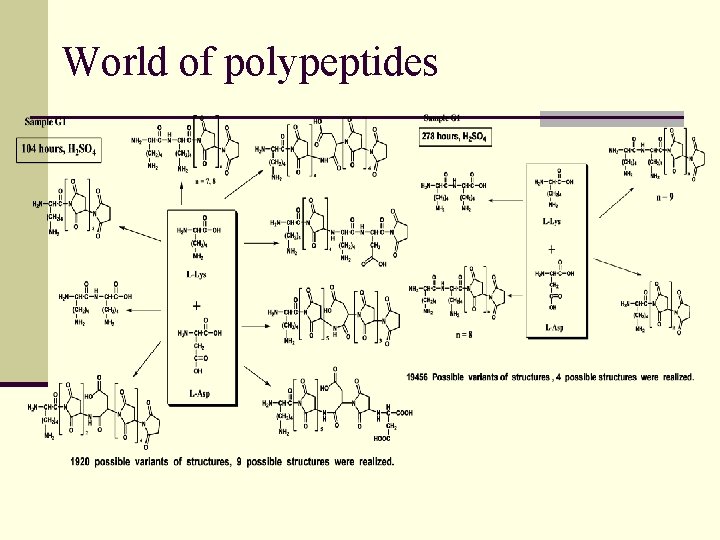
World of polypeptides
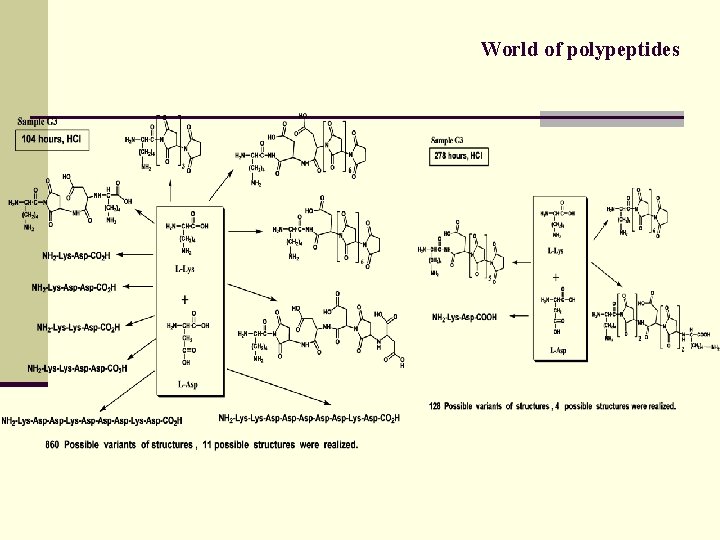
World of polypeptides

World of polypeptides n Lys-Asp, basic solution Chain 5 monomers: 9 structures of 60 possible Chain 7 monomers: 11 structures of 252 possible KK DK DDK KKK DKDK KDKK DKDDK DKKDK KDKKK
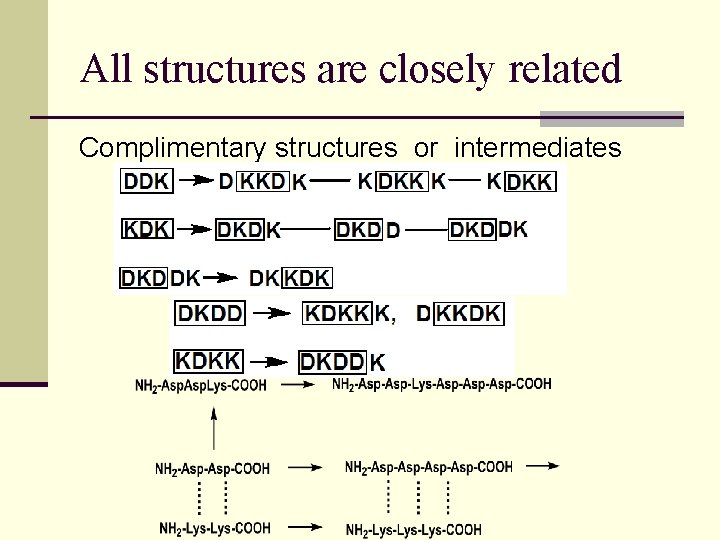
All structures are closely related Complimentary structures or intermediates

General principles of evolution of multifunctional macromolecules q Non-statistical distribution of monomers along the polymeric chain q The primary synthesized polymers works as a template for sorption and subsequent polymerization q Selection factors (stability, catalysis, etc. ) Evolution to unique structure, dramatic reduction of the number of structural variants.
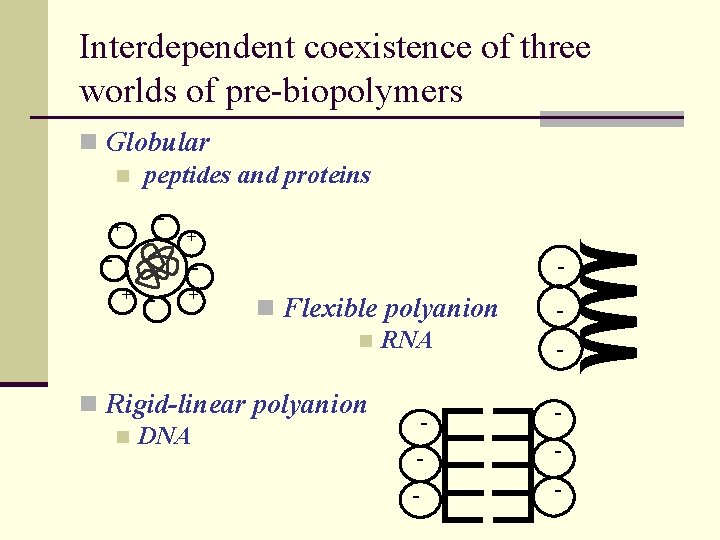
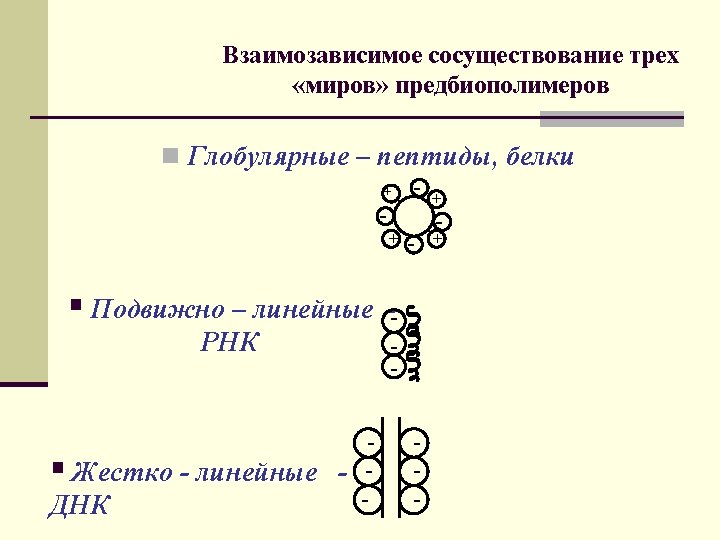
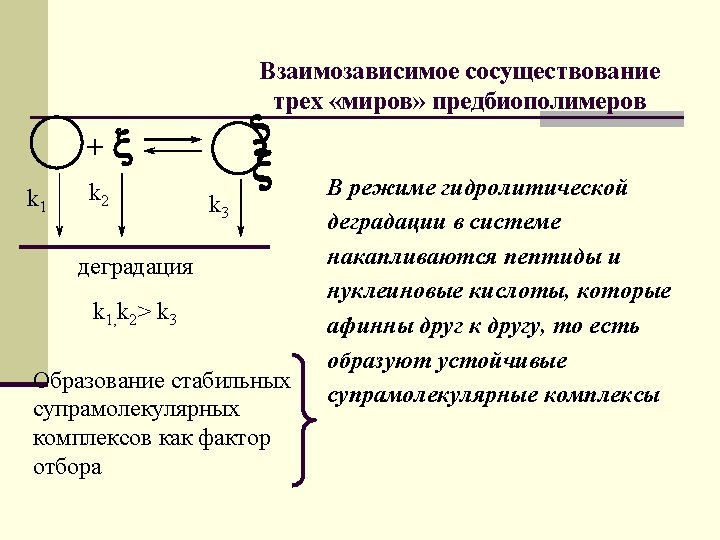
Interdependent coexistence of three worlds of pre-biopolymers n Globular n peptides and proteins - + - - + n Flexible polyanion n RNA n Rigid-linear polyanion n DNA - - + - -
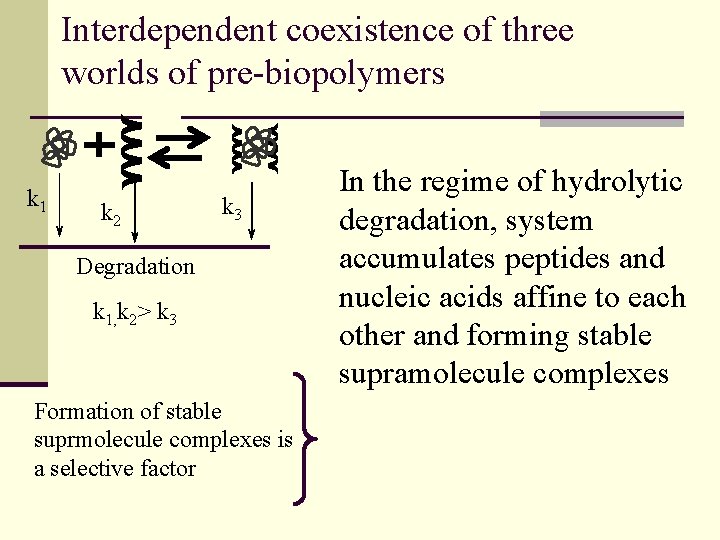
Interdependent coexistence of three worlds of pre-biopolymers k 1 k 2 k 3 Degradation k 1, k 2> k 3 Formation of stable suprmolecule complexes is a selective factor In the regime of hydrolytic degradation, system accumulates peptides and nucleic acids affine to each other and forming stable supramolecule complexes
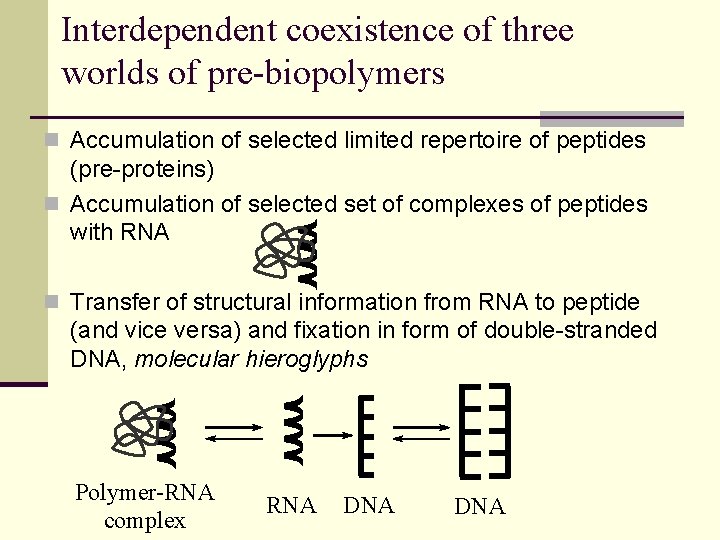
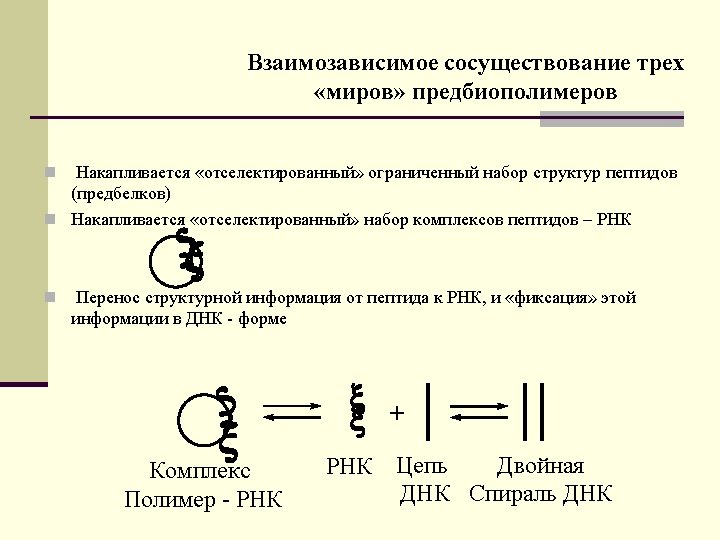
Interdependent coexistence of three worlds of pre-biopolymers n Accumulation of selected limited repertoire of peptides (pre-proteins) n Accumulation of selected set of complexes of peptides with RNA n Transfer of structural information from RNA to peptide (and vice versa) and fixation in form of double-stranded DNA, molecular hieroglyphs Polymer-RNA complex RNA DNA
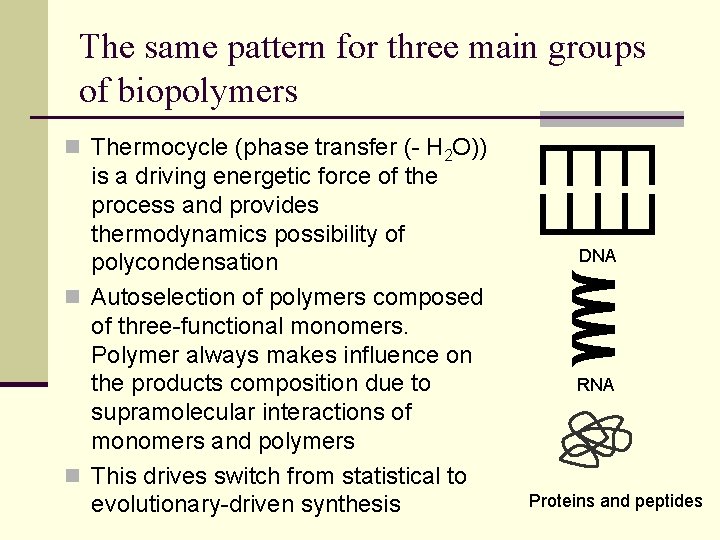
The same pattern for three main groups of biopolymers n Thermocycle (phase transfer (- H 2 O)) is a driving energetic force of the process and provides thermodynamics possibility of polycondensation n Autoselection of polymers composed of three-functional monomers. Polymer always makes influence on the products composition due to supramolecular interactions of monomers and polymers n This drives switch from statistical to evolutionary-driven synthesis DNA RNA Proteins and peptides

General principles of evolution of multifunctional macromolecules (1) Non-statistical distribution of monomers along the polymeric chain (2) The primary synthesized polymers work as the template for sorption and subsequent polymerization (3) Selection (stability, catalysis, etc. ) (4) Evolution to unic structure, dramatic reduction of the number of structural variants.





The great paradox of enzymes origin Very restricted number of catalytical sites structures
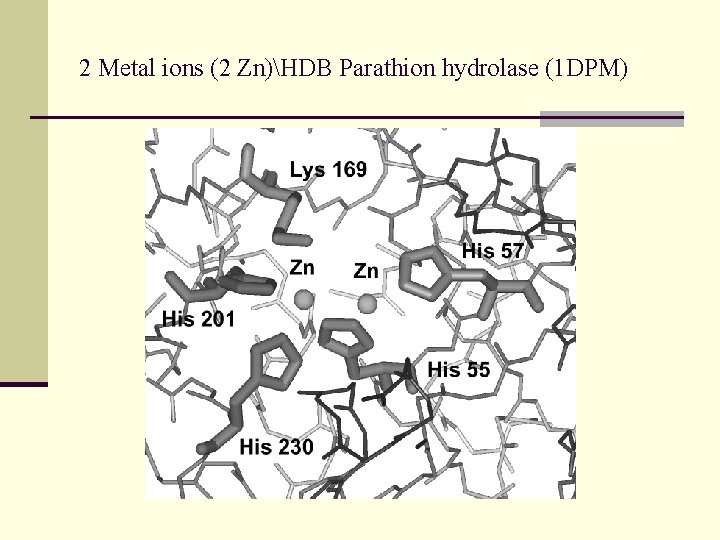
2 Metal ions (2 Zn)HDB Parathion hydrolase (1 DPM)
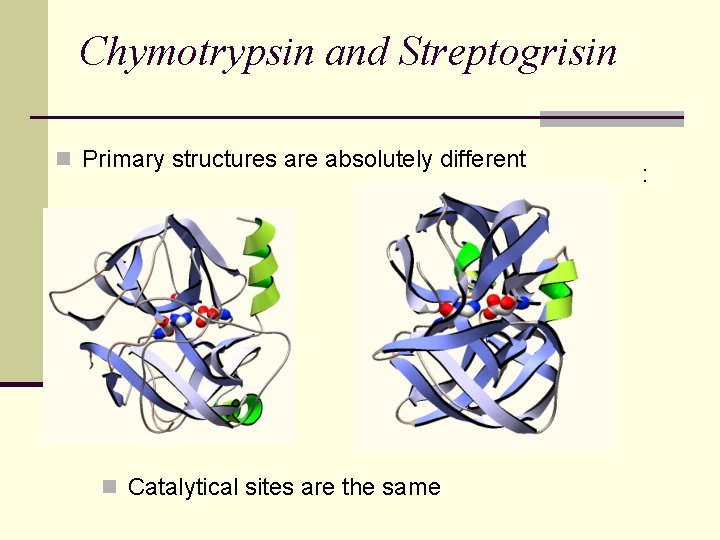
Chymotrypsin and Streptogrisin n Catalytical sites are the same : n Primary structures are absolutely different
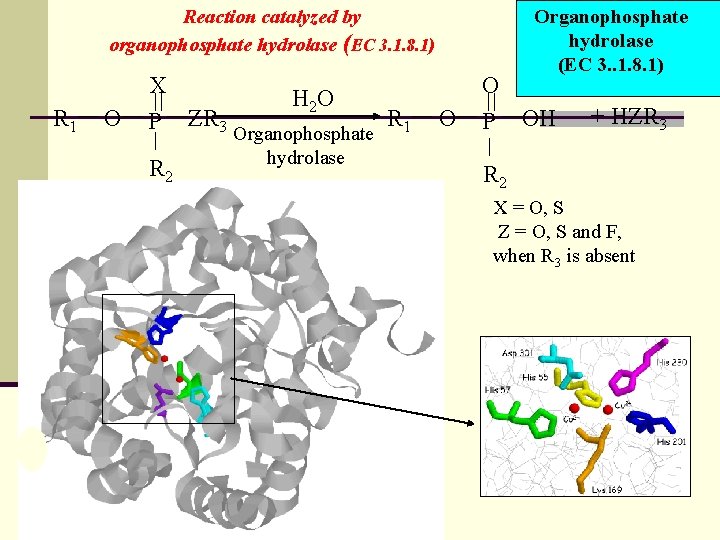
Reaction catalyzed by organophosphate hydrolase (EC 3. 1. 8. 1) X R 1 O P R 2 ZR 3 H 2 O Organophosphate hydrolase O R 1 O P Organophosphate hydrolase (EC 3. . 1. 8. 1) OH + HZR 3 R 2 X = O, S Z = O, S and F, when R 3 is absent
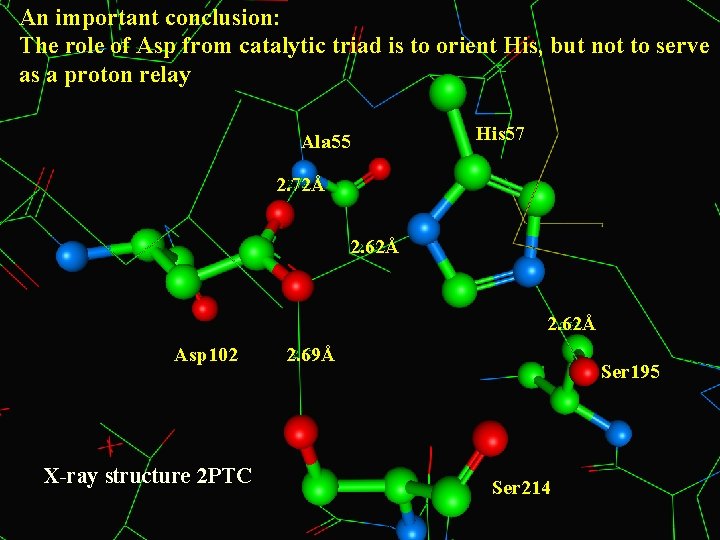
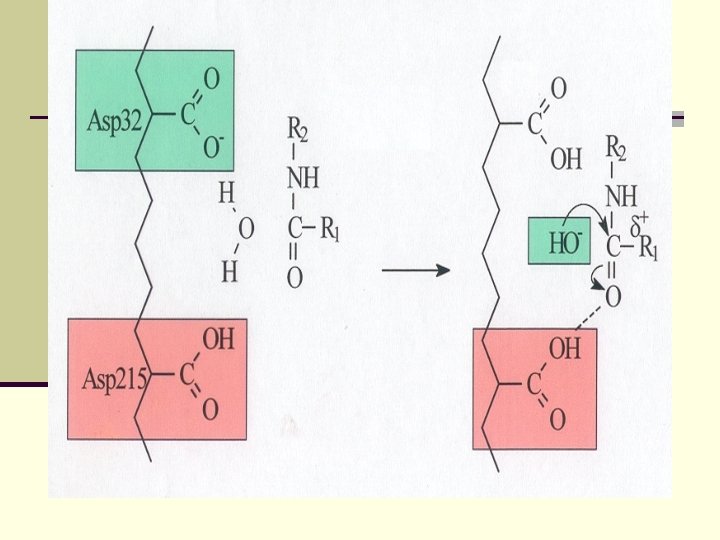
An important conclusion: The role of Asp from catalytic triad is to orient His, but not to serve as a proton relay Ala 55 His 57 2. 72Å 2. 62Å Asp 102 X-ray structure 2 PTC 2. 69Å Ser 195 Ser 214

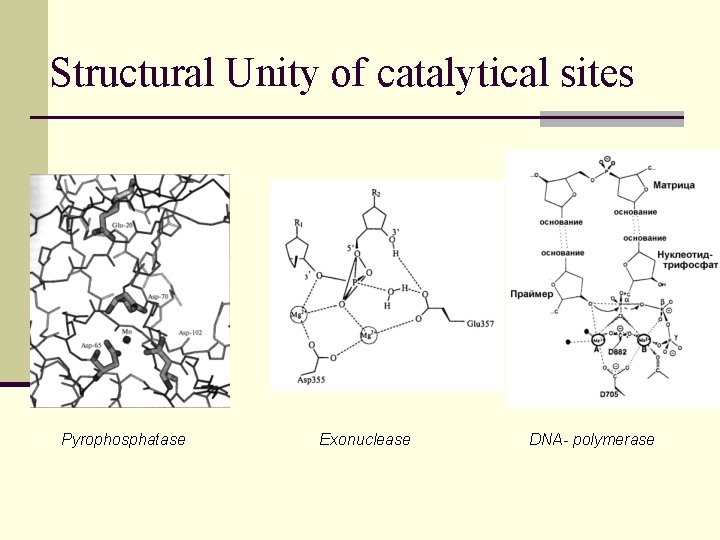
Structural Unity of catalytical sites Pyrophosphatase Exonuclease DNA- polymerase

Prebiological synthesis and evolution of macromolecules S. D. Varfolomeev N. M. Emanuel Institute of Biochemical Physics, RAS, Moscow, Russia sdvarf@sky. chph. ras. ru
- Slides: 58