PreAward Budget Development for Grants OCGA Updates UCLA

Pre-Award: Budget Development for Grants & OCGA Updates UCLA School of Nursing Research Support Team October 29, 2012

The Purpose of Budgets § Demonstrate that PI has considered all costs of achieving the specific aims and project goals § Dedicate effort and resources to the project § Delineate budget categories which must be adhered to (± 25%) once the project is underway § Describe the scientific necessity of every line item

Timeline for Successful Budgets § Three months before due date § Develop Specific Aims § Meet with Statistics Consultation Group to develop power analysis and study size, statistician effort (if applicable), data management plan § Develop project timeline § § What is the project duration? Who is involved? Does effort fluctuate? Enrollment timeline? Analysis timeline?

Timeline for Successful Budgets § Two months before due date § Identify personnel and performance sites § Rough draft of budget and justification § One month before due date § Final budget and justification

Timeline for Successful Budgets § Secure special arrangements earlier § § Permission to exceed direct cost limitation? Subawards to other Institutions? Projects with an international component? Quotes for equipment, consultants, vendor agreements? § Mandatory cost sharing? § Program income?
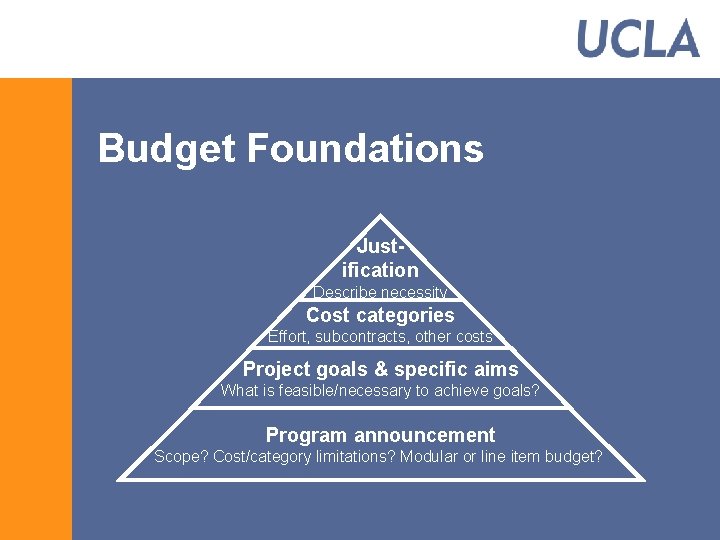
Budget Foundations Justification Describe necessity Cost categories Effort, subcontracts, other costs Project goals & specific aims What is feasible/necessary to achieve goals? Program announcement Scope? Cost/category limitations? Modular or line item budget?

Direct and Indirect costs § Direct Costs identified with a particular sponsored project and directly assigned to that project with a high degree of accuracy Examples: scientific/technical personnel effort, project supplies, subject renumeration, scientific/technical computers § Indirect Costs For common or joint objectives which cannot be identified specifically with a particular project Examples: clerical effort, office supplies, general purpose computers

Direct Cost Categories § § § § Personnel: define effort devoted to the project Materials and supplies Travel (PI to study sites, conferences) Consultants Equipment Subawards Other costs § Define categories and atypical line items at outset to ensure allowability when project is underway § Subject renumeration, TIF, fee remission, animal research, pharmacy costs, rent, project-specific office-type supplies, telephones, mailing, copying, machines (<5 k), core services

Personnel Determinations § § Personnel comprise the largest part of grant budgets 2% escalation per NOT-OD-11 -068 All personnel have a project role, duties, and effort Senior/Key Personnel § individuals who contribute to the scientific development or execution of the project in a substantive, measurable way § Current staff or TBN? § Actual salary/benefits vs institutional scales

Project roles: Senior/Key § Multiple PI § Requires Multiple PI leadership plan § Co-Investigator § Requires stated effort/salary § Faculty Collaborator § Other Significant Contributor § Effort is “as needed”

Subaward, Vendor or Consultant? § Subawards are the most common mechanism for collaboration with another site that brings intellectual value to the project. They have discrete, fully-formed budgets authorized by an institutional official. § Vendor Agreements are established when a site performs work (assays, sample processing) on a feefor-service basis. No intellectual contribution. § Consultants are non-UCLA individuals who perform fee-for-service work at a fixed rate; always requires a letter of support stating the rate, scope and expertise; compliant with SB 1467 “Public Contracts: Conflict of Interest. ”

Human and Animal Subjects § Protocol defines timepoints and processes for subject renumeration which must be included in the budget § Approvals must be in place before award can be issued § Start work on the protocol before award is issued to ensure the project is not delayed or restricted

Financial Characteristics of Contracts & Grants § Defined performance period § Ongoing reporting – progress, effort and technical reports § Auditable financial reports

Financial Characteristics of Contracts & Grants § Most research grants are cost-reimbursable § Specific scope of work § Unobligated funds returned to agency § Some contracts (clinical trials, subcontracts for services) are fixed-rate § Funds provided for services rendered § Unobligated funds retained under Policy 913 § Research Office will advise PI regarding the contract type

Federal Costing Principles § OMB A-21 § Should I charge this to my grant? § Allowability- Does the agency or announcement prohibit certain costs? § Allocability-Is the expense tied directly and proportionally to the project? § Reasonableness- Are the cost and units reasonable in light of the study aims and scope of work? § Consistency- Are the costs consistent across projects and campus units? § OMB A-110 § Competitive procurement § Report submission and records retention § Prior approval for rebudgeting beyond established thresholds

Managing Project Expenses § Partnership between PI, requestor, purchaser and grant analyst to adhere to principles and facilitate timely correction § Continuous, dynamic, collaborative review § Review Process § Research Office provides ad hoc, real-time reports on expenditures § PI defines steps to align expenses with budget and goals § Fund manager and requester take action § Repeat

Managing Project Expenses § By approving a purchase, PI certifies that the expense is necessary, directly benefits and is used exclusively for the project § Use exception processes sparingly or never § Out-of-pocket expenses, cost transfers, solesource purchases § Expenses >90 days old cannot be moved to or between research projects (NIH GPS 7. 5)

Examples of reporting flags § Categorical variances ± 25% § Rebudgeting into/out of IDC-exclusion categories § Effort variances § Unallowable/misattributed items § Carryforward >25% to continuation year

Reporting § Effort reports § PI submits reports quarterly § Prevents overlap and ensures effort is consistent with stated levels § Personnel effort must be ≥ pay § Prior approval required if senior/key effort decreases >25% from proposed effort § Three reporting points: Other Support page, All Personnel Report, Effort Reporting System

Reporting § Progress reports § Submitted to agency annually to secure continuation funding § Publications? Results? Project delays? Changes to project? Enrollment? Effort? Carryforward over threshold? § Sloppy reports are frequently returned and will delay continuation funding

Pre-Award: OCGA Updates UCLA School of Nursing Research Support Team October 29, 2012

OCGA Reorganization § Proposal and Award Intake Teams Created § Grant Analysts § Grants and Cooperation Agreements § Grant Officers § Contracts, Incoming Subs, and Multi Campus Agreements § New Assistant Director § Grants and Cooperative Agreements

OCGA Proposal Intake Process § Proposal Intake Team receives all proposals to review § Team reviews documents for completeness § Complete application § Minimum documents § Less than minimum § some level of review possible § Incomplete/insufficient § Systematic reminders will be issued to follow-up on missing documents

OCGA Proposal Requirements § Minimum for Review § § § Completed EPASS with PI signature Sponsor guidelines All sponsor forms requiring OCGA signature Final budget with budget justification Brief description of proposal aims or proposal abstract § Subaward Documents (if applicable)

OCGA Proposal Requirements § Review, Approval, and Submission § Completed EPASS with all required signatures * § Sponsor guidelines § Final Proposal (budget, science, agency required signatures, biosketches, etc. ) § Subaward Documents (if applicable) * § Signed/Completed COI disclosure (if applicable) § For PHS agencies or agencies that have adopted PHS regulations, all investigators report in e. DGE *

PHS Conflict of Interest Regulations § Applies to all research supported by PHS agencies (NIH, CDC, AHRQ, etc. ) § Applies to non profits that have adopted the PHS COI regulations (American Heart Association, American Cancer Society, Susan G. Komen, etc. ) § Regulations apply to proposals submitted and awards received on or after August 24, 2012.

UCLA Electronic Disclosure Gateway (e. DGE) § By law, the PI and other Investigators must disclose their significant financial interests to UCLA before a proposal can be submitted § Disclosures are required for all research awards, including some that previously did not require disclosure (i. e. conference, equipment, or training grants) § Could be found at: https: //coi. research. ucla. edu § Help Desk for e. DGE: 310 -206 -8546 or rpchelp@research. ucla. edu

UCLA Electronic Disclosure Gateway (e. DGE)

UCLA Electronic Disclosure Gateway (e. DGE)

OCGA EPASS Form § Replaced the Goldenrod § EPASS has been mandatory since September 1 § The PI/Co. PI signature(s) on the EPASS incorporate required PHS certification § NIH/PHS PI signature Form is no longer needed § Utilizes dropdowns and hyperlinks § Form could be found at: http: //www. research. ucla. edu/ocga/Forms/EPASS_Form. pdf
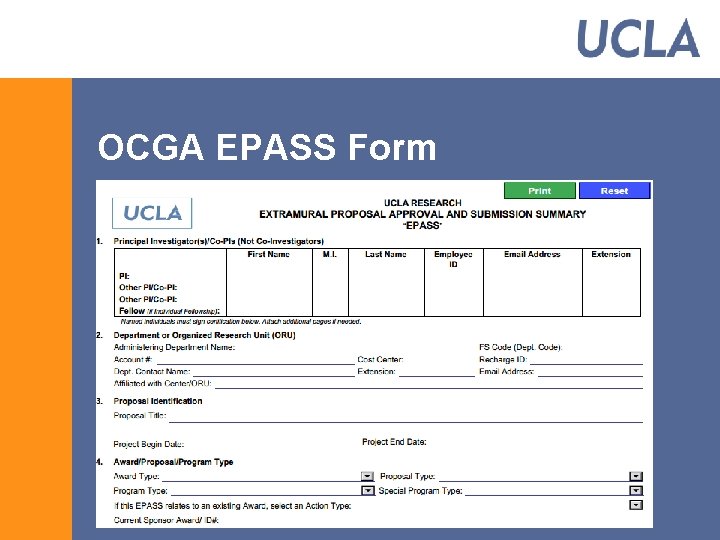
OCGA EPASS Form
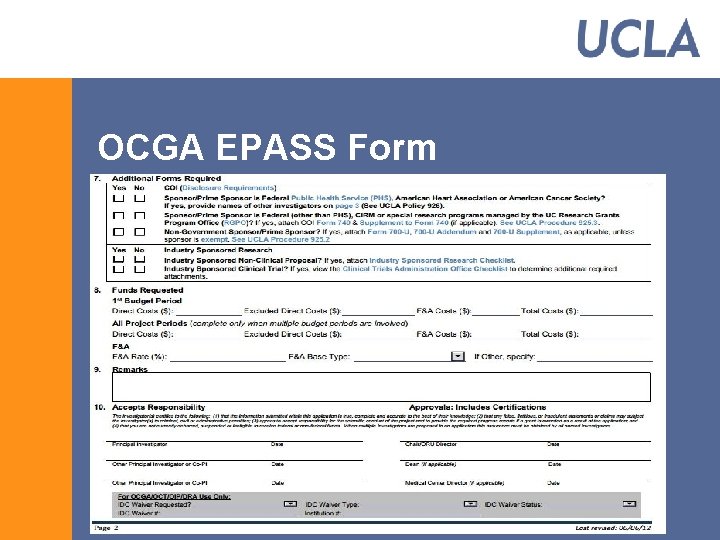
OCGA EPASS Form
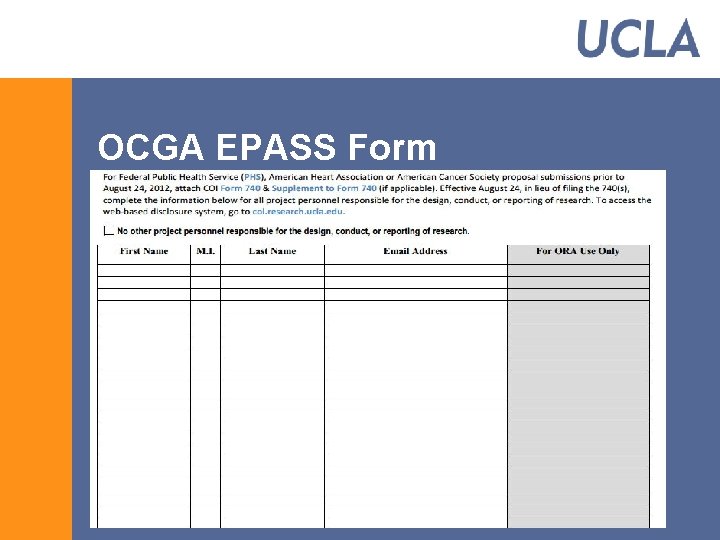
OCGA EPASS Form

OCGA Subrecipient Requirements § Prior to proposal submission subs need to submit: § Statement of work § Budget and budget justification § Subrecipient Commitment Form § Now incorporates new PHS COI regulations § Form could be found at: http: //www. research. ucla. edu/ocga/Forms/Sub_Commit ment_Form_rev. Sept_2012. pdf

Subrecipient Commitment Form

Subrecipient Commitment Form

Subrecipient Commitment Form

Summary § Budgets and expenses are a function of the project goals and specific aims § Budgets should be as realistic as possible to minimize post-award changes § PI and departmental coordinators are jointly responsible for adhering to costing principles and managing project expenses § OCGA form/process changes reflect the revised PHS funding regulations.

Summary § Proposal submission tips: § Familiarize yourself with UCLA Policies, Procedures, and Forms (as well as Sponsor Application Instructions and Forms) § Alert the SON Research Office regarding all upcoming submissions § Meet deadlines for a successful submission § Start Early!

Questions? § Slides will be available on the web (click Research, Past Presentations) § Email researchoffice@sonnet. ucla. edu
- Slides: 40