PRavastatin Or ator Vastatin Evaluation and Infection Therapy









- Slides: 18

PRavastatin Or ator. Vastatin Evaluation and Infection Therapy (TIMI 22) Disclosure Statement: Dr. Cannon currently receives research grant support from Bristol-Myers Squibb, Merck and Sanofi-Synthelabo. He serves as a consultant to Astra. Zeneca, Glaxo Smith Kline, Guilford Pharmaceuticals and Vertex

Background Statin therapy is highly effective vs. placebo in longterm treatment of CHD l l Are statins effective in reducing events in patients with an acute coronary syndrome (ACS)? Does “intensive” LDL-C lowering to an average of 65 mg/d. L achieve a greater reduction in clinical events than “standard” LDL-C lowering to an average of 95 mg/d. L?

PROVE IT - TIMI 22: Study Design 4, 162 patients with an Acute Coronary Syndrome < 10 days Double-blind ASA + Standard Medical Therapy “Standard Therapy” Pravastatin 40 mg “Intensive Therapy” Atorvastatin 80 mg 2 x 2 Factorial: Gatifloxacin vs. placebo Duration: Mean 2 year follow-up (>925 events) Primary Endpoint: Death, MI, Documented UA requiring hospitalization, revascularization (> 30 days after randomization), or Stroke

Patient Population Inclusion Criteria: l Hospitalization for acute MI or high-risk unstable angina < 10 d l Total cholesterol < 240 mg/d. L (< 200 mg/d. L if on Lipid Rx) l Stabilized (i. e. , without ischemia, CHF, post PCI if performed) Major Exclusion Criteria: l Co-morbidity: patient survival < 2 years l Current therapy with simvastatin or atorvastatin 80 mg l Need for, or anticipated use of fibrates or niacin l CABG for treatment of qualifying ACS l Liver disease or unexplained CK elevations l Strong inhibitors of CYP 450 3 A 4 (2 o atorvastatin metabolism)

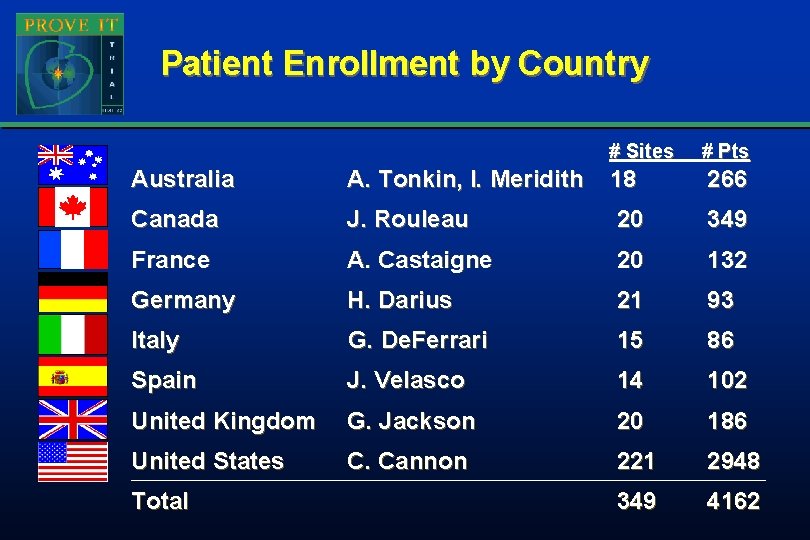
Patient Enrollment by Country # Sites # Pts Australia A. Tonkin, I. Meridith 18 266 Canada J. Rouleau 20 349 France A. Castaigne 20 132 Germany H. Darius 21 93 Italy G. De. Ferrari 15 86 Spain J. Velasco 14 102 United Kingdom G. Jackson 20 186 United States C. Cannon 221 2948 349 4162 Total

Top Ten Enrolling Clinical Centers Hospital Principal Investigator Research Coordinator Huntsville Hospital, Huntsville AL W. Haught K. Griffin Fremantle Hospital, Fremantle WA R. Hendriks D. Greenwell Detar Hospital, Victoria, TX H. Chandna D. Holly St. Francis Hospital, Tulsa, OK J. M. Cassidy N. Ritchie Advanced Health Institute, Galax, VA J. Puma E. Jones Michigan Heart, Ypsilanti, MI J. Bengtson C. Carulli N. Mississippi Medical Center, Tupelo, MS B. Bertolet M. Jones Wilford Hall Med Center, Lackland AFB, TX R. Krasuski U. Ward Queen Elizabeth Hospital, Woodville, Sa J. Horowitz R. Prideaux Moses H. Cone Hospital, Greensboro, NC T. Kelly K. Cochran

Trial Organization TIMI Study Group Brigham and Women’s Hosp. Eugene Braunwald, MD Christopher Cannon, MD Carolyn Mc. Cabe, BS Data Coordinating Center Nottingham Allan Skene Ph. D. Karen Hill Sponsors: Bristol-Myers Squibb and Sankyo Co. LTD Rene Belder, MD Steven Joyal, MD Gabriella Cucinotta Chen-Sheng Lin, Ph. D Clinical Events Committee: Marc Pfeffer, MD, Ph. D

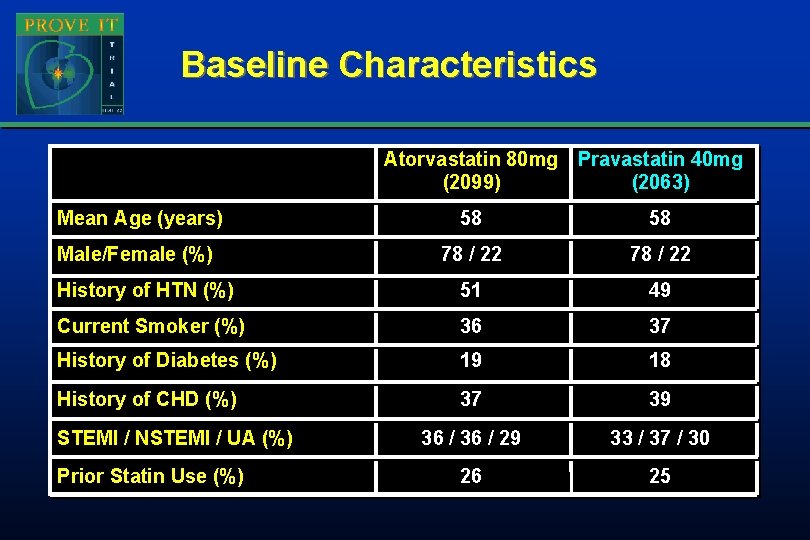
Baseline Characteristics Atorvastatin 80 mg Pravastatin 40 mg (2099) (2063) Mean Age (years) 58 58 Male/Female (%) 78 / 22 History of HTN (%) 51 49 Current Smoker (%) 36 37 History of Diabetes (%) 19 18 History of CHD (%) 37 39 36 / 29 33 / 37 / 30 26 25 STEMI / NSTEMI / UA (%) Prior Statin Use (%)

Concomitant Therapies PCI for initial ACS pre-Rand 69% Aspirin 93% Warfarin 8% Clopidogrel (initial) (at F/U) 72% 20% B-blockers 85% ACE 69% ARB 14%

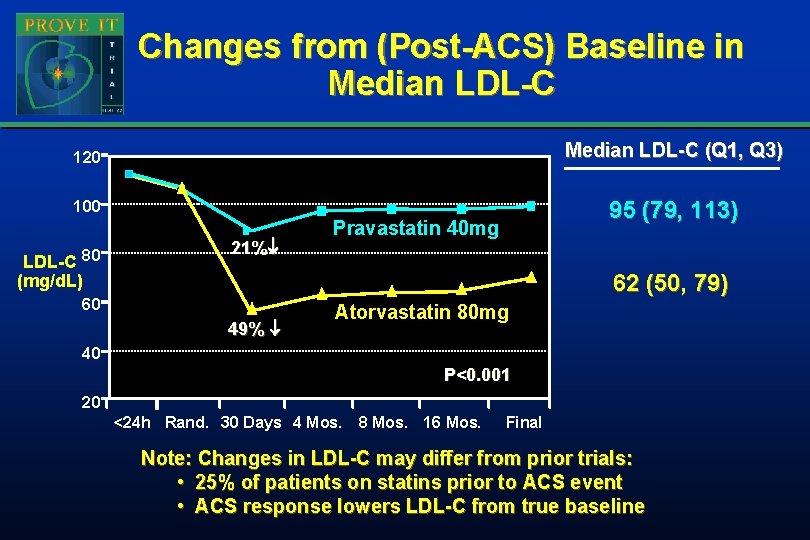
Changes from (Post-ACS) Baseline in Median LDL-C 120 Median LDL-C (Q 1, Q 3) 100 95 (79, 113) LDL-C 80 21% Pravastatin 40 mg 62 (50, 79) (mg/d. L) 60 49% Atorvastatin 80 mg 40 P<0. 001 20 <24 h Rand. 30 Days 4 Mos. 8 Mos. 16 Mos. Final Note: Changes in LDL-C may differ from prior trials: • 25% of patients on statins prior to ACS event • ACS response lowers LDL-C from true baseline

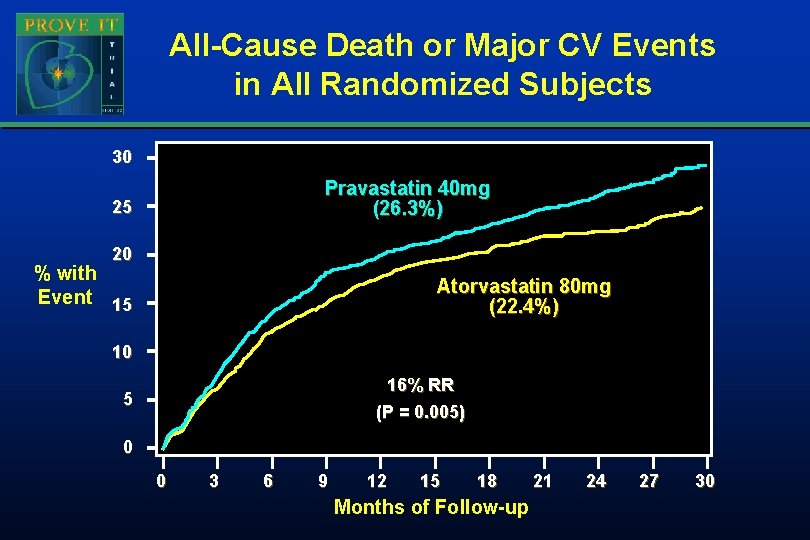
All-Cause Death or Major CV Events in All Randomized Subjects 30 Pravastatin 40 mg (26. 3%) 25 20 % with Event 15 Atorvastatin 80 mg (22. 4%) 10 16% RR (P = 0. 005) 5 0 0 3 6 9 12 15 18 Months of Follow-up 21 24 27 30

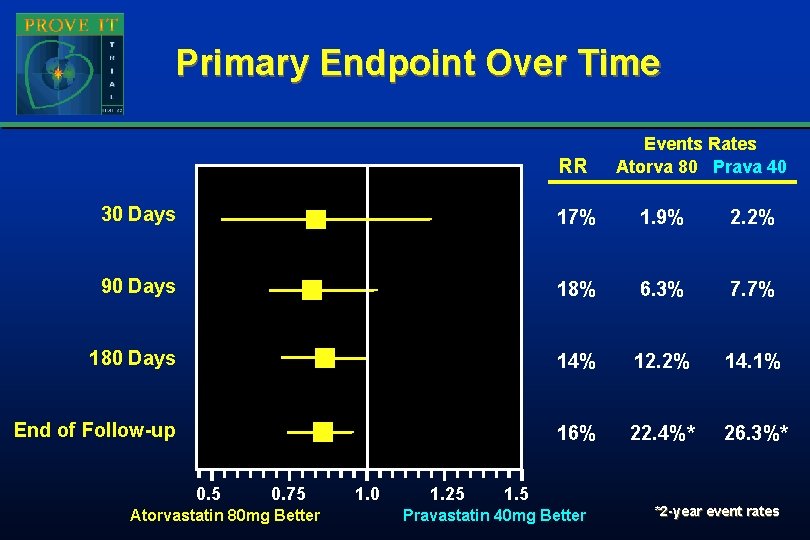
Primary Endpoint Over Time RR Events Rates Atorva 80 Prava 40 30 Days 17% 1. 9% 2. 2% 90 Days 18% 6. 3% 7. 7% 180 Days 14% 12. 2% 14. 1% End of Follow-up 16% 22. 4%* 26. 3%* 0. 5 0. 75 Atorvastatin 80 mg Better 1. 0 1. 25 1. 5 Pravastatin 40 mg Better *2 -year event rates

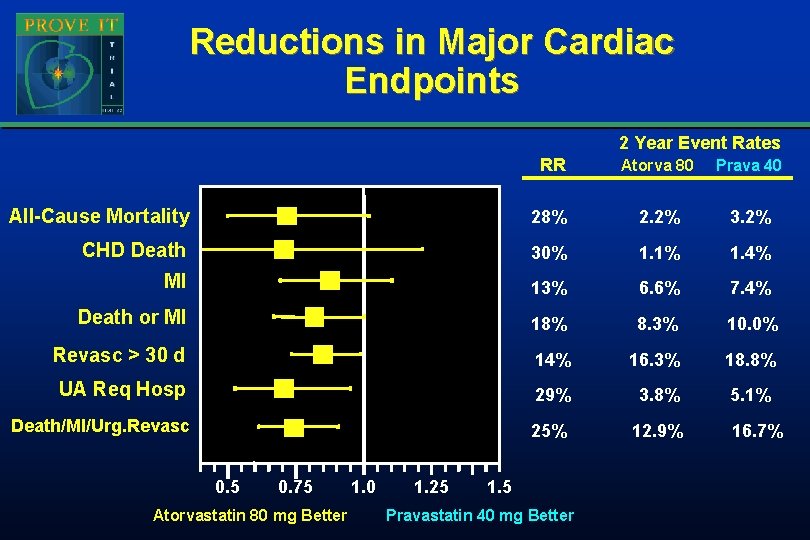
Reductions in Major Cardiac Endpoints 2 Year Event Rates RR Atorva 80 Prava 40 All-Cause Mortality 28% 2. 2% 3. 2% CHD Death 30% 1. 1% 1. 4% MI 13% 6. 6% 7. 4% Death or MI 18% 8. 3% 10. 0% Revasc > 30 d 14% 16. 3% 18. 8% UA Req Hosp 29% 3. 8% 5. 1% Death/MI/Urg. Revasc 25% 12. 9% 0. 5 0. 75 Atorvastatin 80 mg Better 1. 0 1. 25 1. 5 Pravastatin 40 mg Better 16. 7%

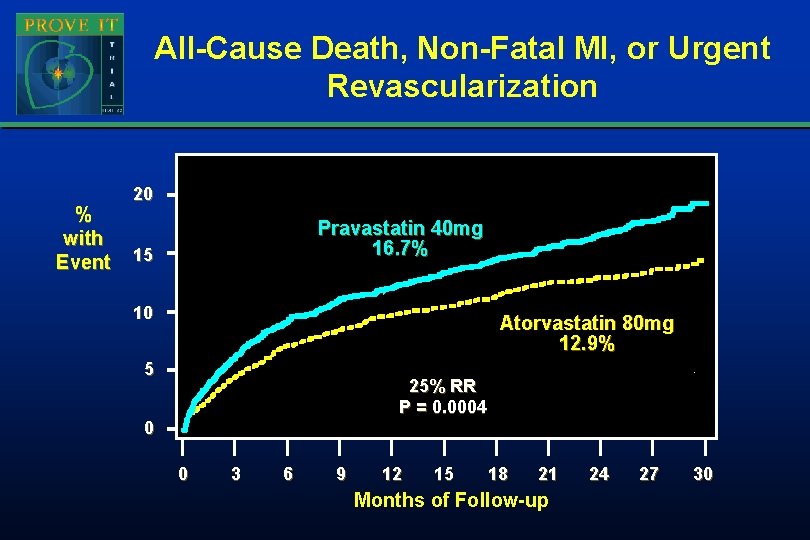
All-Cause Death, Non-Fatal MI, or Urgent Revascularization % with Event 20 Pravastatin 40 mg 16. 7% 15 10 Atorvastatin 80 mg 12. 9% 5 25% RR P = 0. 0004 0 0 3 6 9 12 15 18 21 Months of Follow-up 24 27 30

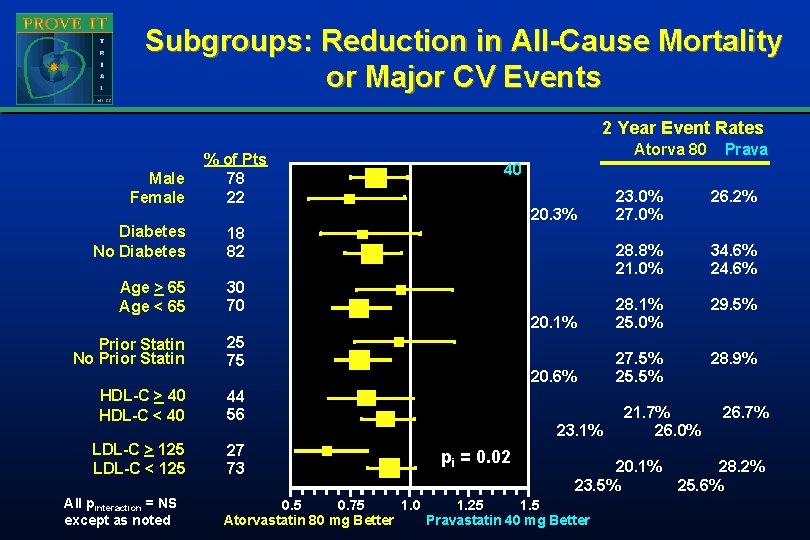
Subgroups: Reduction in All-Cause Mortality or Major CV Events 2 Year Event Rates Male Female % of Pts 78 22 Diabetes No Diabetes 18 82 Age > 65 Age < 65 30 70 Prior Statin No Prior Statin 25 75 HDL-C > 40 HDL-C < 40 44 56 LDL-C > 125 LDL-C < 125 27 73 All pinteraction = NS except as noted Atorva 80 Prava 40 23. 0% 27. 0% 26. 2% 28. 8% 21. 0% 34. 6% 29. 5% 20. 1% 28. 1% 25. 0% 28. 9% 20. 6% 27. 5% 25. 5% 20. 3% 23. 1% pi = 0. 02 21. 7% 26. 0% 20. 1% 23. 5% 0. 5 0. 75 1. 0 1. 25 1. 5 Atorvastatin 80 mg Better Pravastatin 40 mg Better 26. 7% 28. 2% 25. 6%

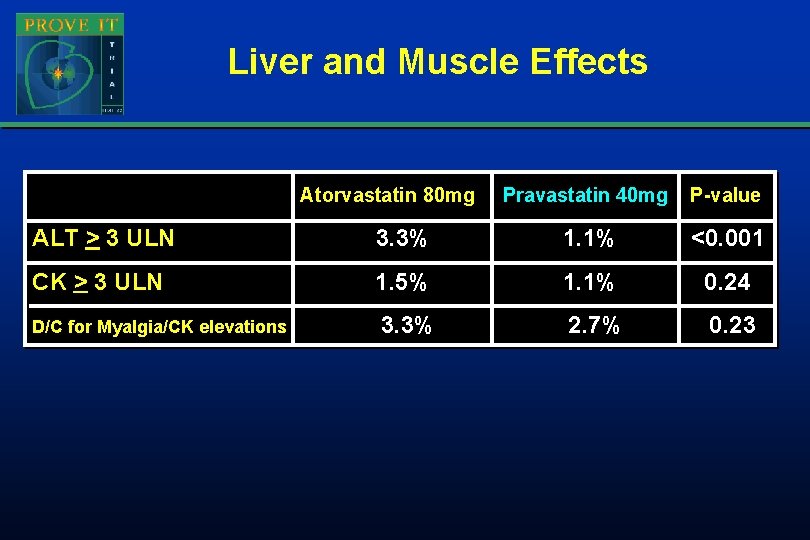
Liver and Muscle Effects Atorvastatin 80 mg Pravastatin 40 mg P-value ALT > 3 ULN 3. 3% 1. 1% <0. 001 CK > 3 ULN 1. 5% 1. 1% 0. 24 3. 3% 2. 7% 0. 23 D/C for Myalgia/CK elevations

Summary In patients recently hospitalized within 10 days for an acute coronary syndrome: · “Intensive” high-dose LDL-C lowering (median LDL-C 62 mg/d. L) compared to “moderate” standard-dose lipid-lowering therapy (median LDL-C 95 mg/d. L) reduced the risk of all cause mortality or major cardiac events by 16% (p=0. 005) · Benefits emerged within 30 days post ACS with continued benefit observed throughout the 2. 5 years of follow-up · Benefits were consistent across all cardiovascular endpoints, except stroke, and most clinical subgroups

PROVE-IT Conclusion MARCH 8, 2004 Our findings indicate that patients recently hospitalized for an acute coronary syndrome benefit from early and continued lowering of LDL-C to levels substantially below current target levels. Cannon CP, Braunwald E, Mc. Cabe CH, et al. N Engl J Med 2004; 350: 15 www. nejm. org