Pramlintide An analog of amylin that overcomes the


- Slides: 10

Pramlintide – An analog of amylin that overcomes the tendency of human amylin to: • Aggregate, form insoluble particles • Adhere to surfaces – Pharmacokinetic and pharmacodynamic properties similar to human amylin Human amylin Pramlintide (analog of amylin) T A T N C T A Q R L A N F L V Y S K T N S V N S S T L I A G F S N N G Adapted from Young A, et al. Drug Dev Res 1996; 37: 231 -248 Adapted from Westermark P, et al. Proc Natl Acad Sci 1990; 87: 5036 -5040 T N H C Amide A T C A T Q R L A N F L V H C Amide Y S K T N S G V N T P P L I P G F N N S

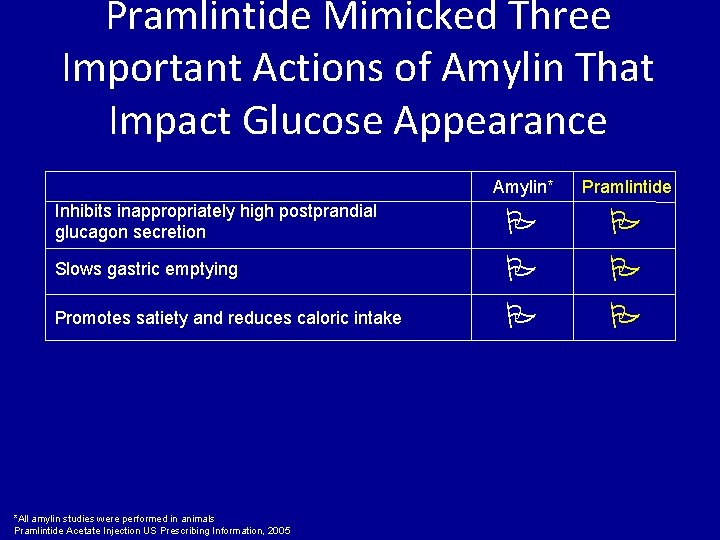
Pramlintide Mimicked Three Important Actions of Amylin That Impact Glucose Appearance Inhibits inappropriately high postprandial glucagon secretion Slows gastric emptying Promotes satiety and reduces caloric intake *All amylin studies were performed in animals Pramlintide Acetate Injection US Prescribing Information, 2005 Amylin* Pramlintide

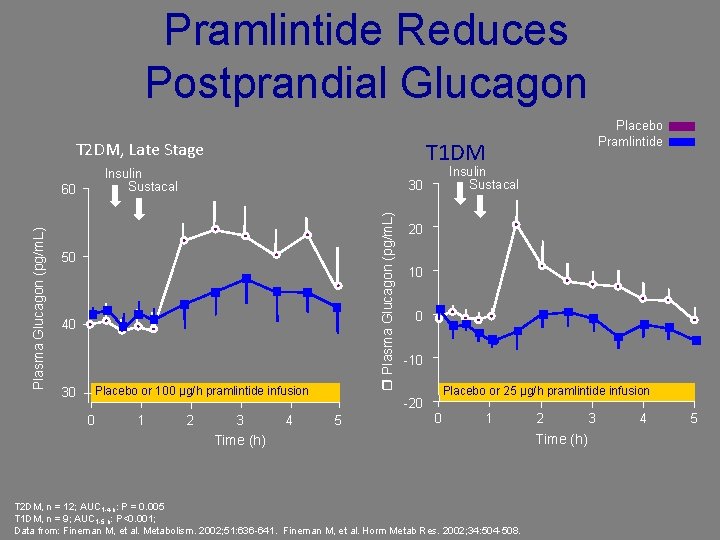
Pramlintide Reduces Postprandial Glucagon T 1 DM T 2 DM, Late Stage Insulin Sustacal 50 40 30 Placebo or 100 µg/h pramlintide infusion 0 1 Insulin Sustacal 30 Plasma Glucagon (pg/m. L) 60 2 Placebo Pramlintide 3 4 20 10 0 -10 Placebo or 25 µg/h pramlintide infusion -20 5 0 1 Time (h) T 2 DM, n = 12; AUC 1 -4 h: P = 0. 005 T 1 DM, n = 9; AUC 1 -5 h: P<0. 001; Data from: Fineman M, et al. Metabolism. 2002; 51: 636 -641. Fineman M, et al. Horm Metab Res. 2002; 34: 504 -508. 2 Time (h) 3 4 5

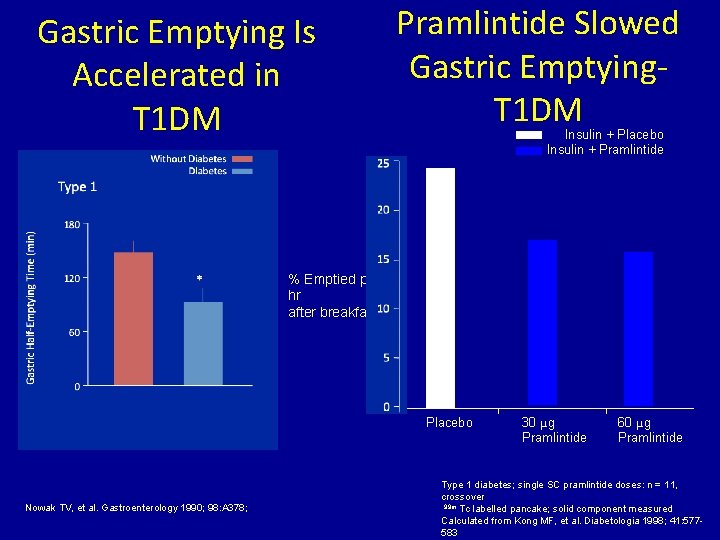
Gastric Emptying Is Accelerated in T 1 DM Pramlintide Slowed Gastric Emptying. T 1 DM Insulin + Placebo Insulin + Pramlintide % Emptied per hr after breakfast Placebo Nowak TV, et al. Gastroenterology 1990; 98: A 378; 30 μg Pramlintide 60 μg Pramlintide Type 1 diabetes; single SC pramlintide doses: n = 11, crossover 99 m Tc labelled pancake; solid component measured Calculated from Kong MF, et al. Diabetologia 1998; 41: 577583

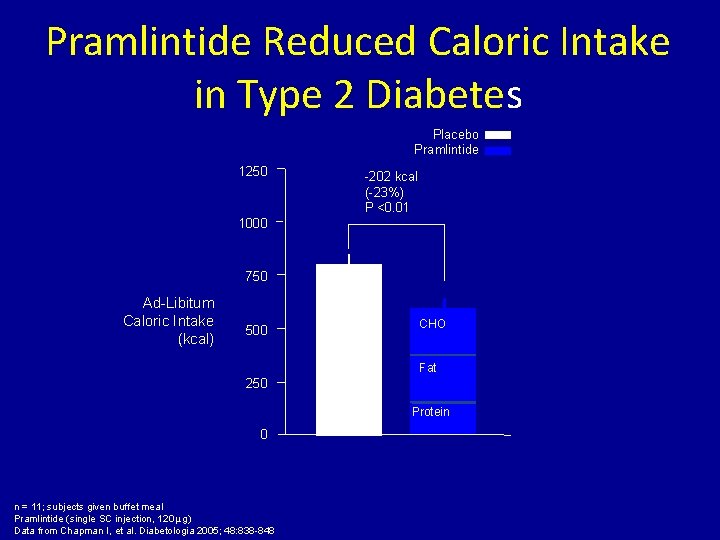
Pramlintide Reduced Caloric Intake in Type 2 Diabetes Placebo Pramlintide 1250 -202 kcal (-23%) P <0. 01 1000 750 Ad-Libitum Caloric Intake (kcal) CHO 500 Fat 250 Protein 0 n = 11; subjects given buffet meal Pramlintide (single SC injection, 120 μg) Data from Chapman I, et al. Diabetologia 2005; 48: 838 -848 Protein

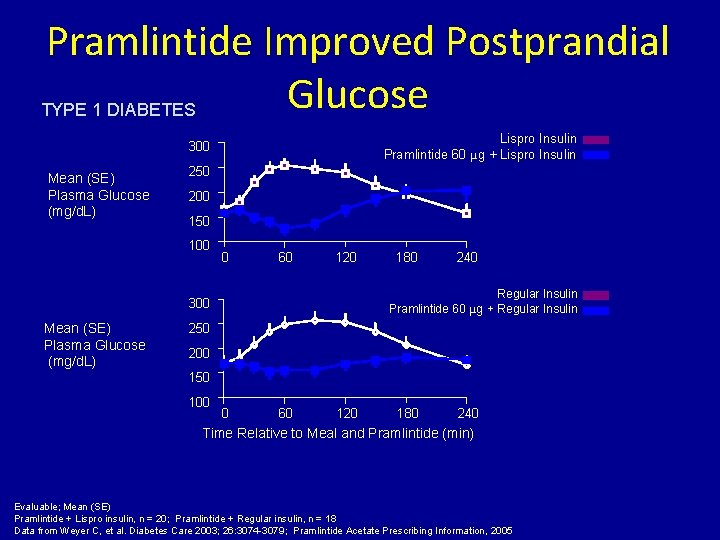
Pramlintide Improved Postprandial Glucose TYPE 1 DIABETES Lispro Insulin Pramlintide 60 μg + Lispro Insulin 300 Mean (SE) Plasma Glucose (mg/d. L) 250 200 150 100 0 60 120 240 Regular Insulin Pramlintide 60 μg + Regular Insulin 300 Mean (SE) Plasma Glucose (mg/d. L) 180 250 200 150 100 0 60 120 180 240 Time Relative to Meal and Pramlintide (min) Evaluable; Mean (SE) Pramlintide + Lispro insulin, n = 20; Pramlintide + Regular insulin, n = 18 Data from Weyer C, et al. Diabetes Care 2003; 26: 3074 -3079; Pramlintide Acetate Prescribing Information, 2005

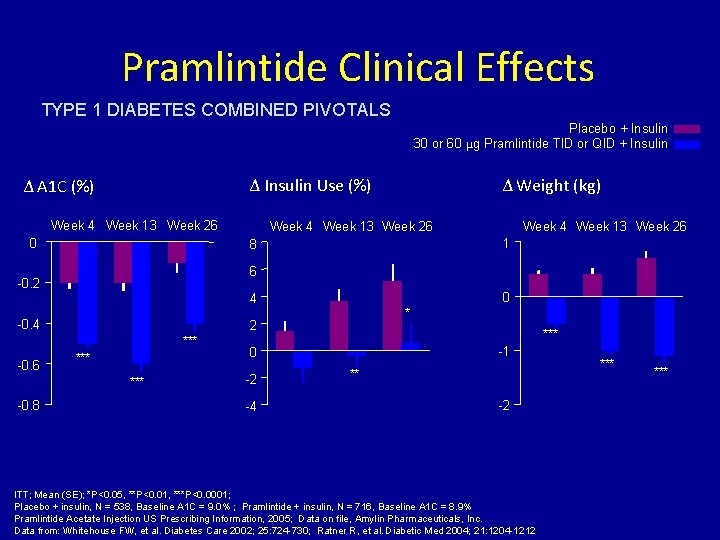
Pramlintide Clinical Effects TYPE 1 DIABETES COMBINED PIVOTALS Placebo + Insulin 30 or 60 μg Pramlintide TID or QID + Insulin Δ Insulin Use (%) Δ A 1 C (%) Week 4 Week 13 Week 26 0 Week 4 Week 13 Week 26 1 6 0 4 -0. 4 * 2 *** *** -0. 8 Week 4 Week 13 Week 26 8 -0. 2 -0. 6 Δ Weight (kg) *** -1 0 -2 -4 ** -2 ITT; Mean (SE); *P<0. 05, **P<0. 01, ***P<0. 0001; Placebo + insulin, N = 538, Baseline A 1 C = 9. 0% ; Pramlintide + insulin, N = 716, Baseline A 1 C = 8. 9% Pramlintide Acetate Injection US Prescribing Information, 2005; Data on file, Amylin Pharmaceuticals, Inc. Data from: Whitehouse FW, et al. Diabetes Care 2002; 25: 724 -730; Ratner R, et al. Diabetic Med 2004; 21: 1204 -1212 ***

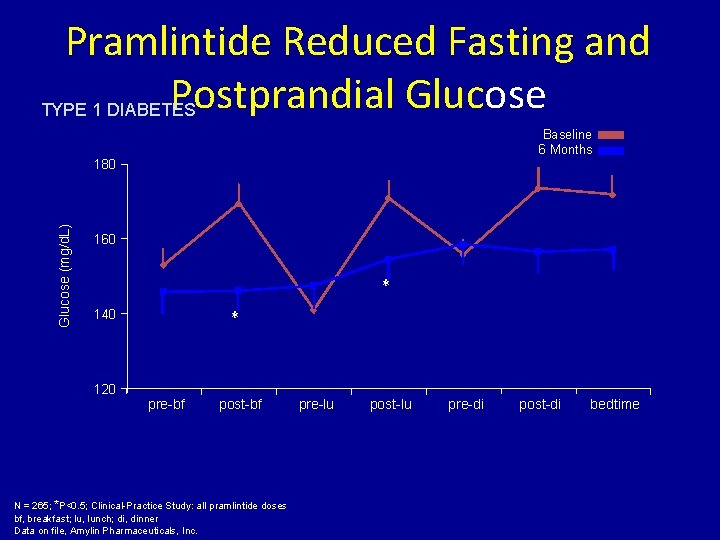
Pramlintide Reduced Fasting and Postprandial Glucose TYPE 1 DIABETES Baseline 6 Months Glucose (mg/d. L) 180 160 * * 140 120 pre-bf post-bf N = 265; *P<0. 5; Clinical-Practice Study: all pramlintide doses bf, breakfast; lu, lunch; di, dinner Data on file, Amylin Pharmaceuticals, Inc. pre-lu post-lu pre-di post-di bedtime

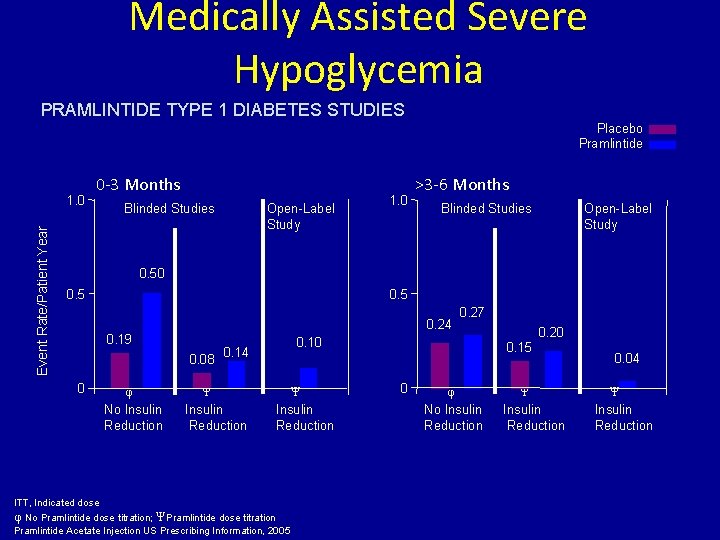
Medically Assisted Severe Hypoglycemia PRAMLINTIDE TYPE 1 DIABETES STUDIES Placebo Pramlintide Event Rate/Patient Year 1. 0 0 -3 Months Blinded Studies Open-Label Study 1. 0 >3 -6 Months Blinded Studies Open-Label Study 0. 50 0. 5 0. 24 0. 19 0. 08 0 φ No Insulin Reduction 0. 10 0. 14 Ψ Insulin Reduction 0. 27 Ψ Insulin Reduction ITT, Indicated dose φ No Pramlintide dose titration; Ψ Pramlintide dose titration Pramlintide Acetate Injection US Prescribing Information, 2005 0. 20 0. 15 0 φ No Insulin Reduction Ψ Insulin Reduction 0. 04 Ψ Insulin Reduction

Pramlintide Safety and Tolerability in Type 1 Diabetes • Insulin-Induced Severe Hypoglycemia: – More common in type 1 diabetes; risk reduced by appropriate patient selection, careful patient instruction and insulin dose adjustments as stated in the Boxed Warning • Nausea: – Mostly mild-to-moderate nausea. Occurred more frequently during initiation and then decreased with time but can increase risk of hypoglycemia. – Nausea reduced by dose titration – Could increase risk of insulin-induced severe hypoglycemia due to reduced food intake Pramlintide Acetate Injection US Prescribing Information, 2005