Practice Write in shorthand noble gas notation for

Practice: Write in shorthand noble gas notation for: F, K, Co, Kr, Ce

Classifying Elements by Electron Configuration

Classifying Elements by Electron Configuration • Blocks on the periodic table (s, p, d, f) • Group number equals the number of electrons in the outermost energy level

Classifying Elements by Electron Configuration • Each period number corresponds to the outermost principle energy level


Practice: Identify the block (s, p, d, f) of the periodic table that this element is located. A) Zinc B) Sodium C) Uranium D) Nitrogen

Periodic Trends There are 5 trends (patterns in groups or periods) on the periodic table to consider: atomic size, ionization energy, ionic size, electronegativity, and electron affinity. Read the descriptions in the notes and then answer the questions on the worksheet.

Trend #1: Atomic Size • Measured by the radius of an atom via x-ray diffraction
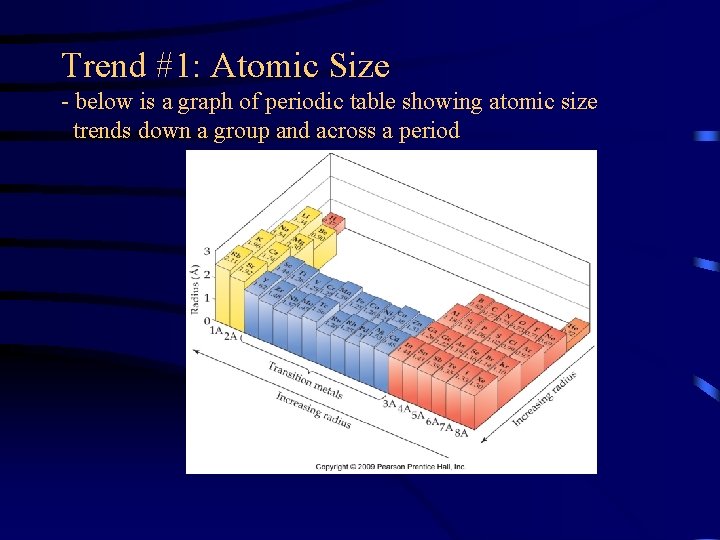
Trend #1: Atomic Size - below is a graph of periodic table showing atomic size trends down a group and across a period

Trends in Atomic Size • Atomic size increases down a group because electrons are added to higher energy levels, which are further away from the nucleus. Since there is less of an attraction of the nucleus for the electrons, the atom is larger.

Trend #1: Atomic Size • Atomic size decreases across a period because within the same energy level, each element has one more p+ and ethan the preceding element. Therefore, outmost e’s are attracted more closely to the nucleus making it smaller.

Ion • Define ion • Predict the charge of the ion typically formed by the elements in groups 1 -8 on the periodic table

Trend #2: Ionization Energy (IE) • IE is the energy required to overcome attraction of nuclear charge and remove an electron from an atom.
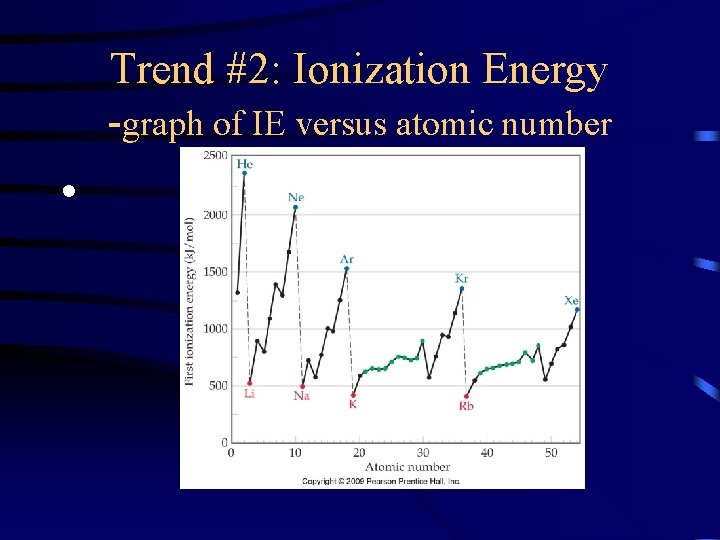
Trend #2: Ionization Energy -graph of IE versus atomic number •
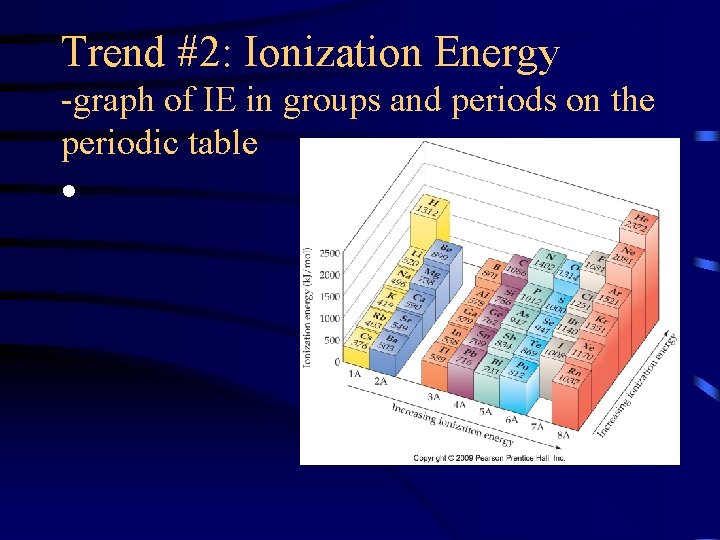
Trend #2: Ionization Energy -graph of IE in groups and periods on the periodic table •
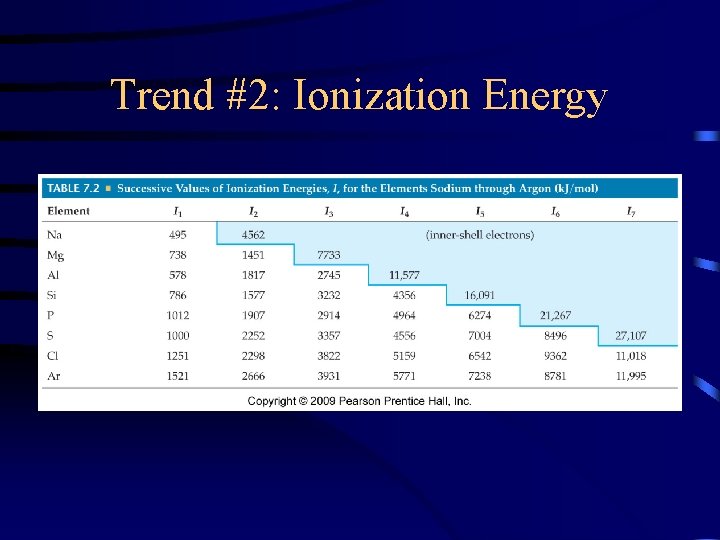
Trend #2: Ionization Energy •

Trend #2: Ionization Energy • IE decreases as you move down a group because the outermost electron is not as attracted to the nucleus so it is easier to remove

Trend #2: Ionization Energy • IE increases as you move left to right across a period because more protons in the nucleus lead to greater attraction for the electrons in the same energy level

Trend #3: Ionic Size • Cations < Neutral < Anions • Consider the attraction of the nucleus for the outermost electrons to see why this is true

Trend #3: Ionic Size - table of ionic size and atomic size •

Trend #3: Ionic Size • Ionic size increases down a group because the electrons are in energy levels further away from the nucleus

Trend #3: Ionic Size • Across a period ionic size of positive ions and negative ions decreases • But keep in mind that anions have bigger radii than cations.

Trend #4: Electronegativity (EN) • EN is a measure of the ability of an atom in a chemical compound to attract electrons • Measured by the Pauling electronegativity scale.
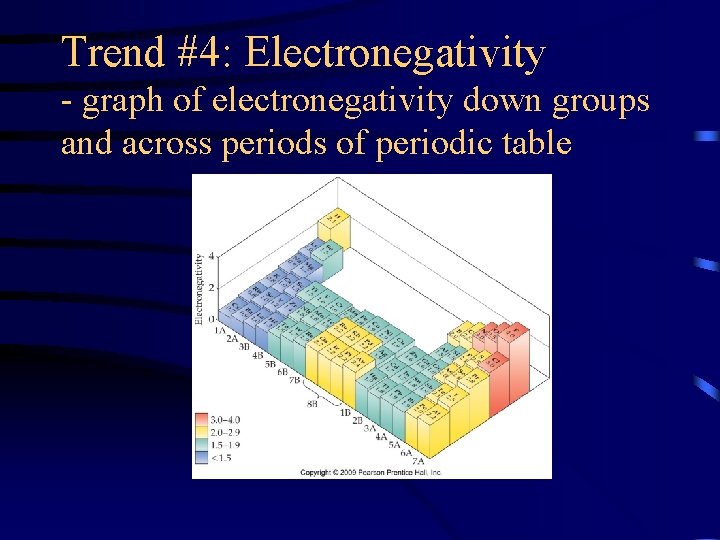
Trend #4: Electronegativity - graph of electronegativity down groups and across periods of periodic table

Trend #4: Electronegativity • EN generally decreases down a group • EN generally increases across a period

Trend #5: Electron Affinity (EA) • EA is the energy change that occurs when an electron is gained by a neutral atom

Trend #5: Electrons Affinity • This number is negative when energy is released and positive when energy is gained

Trends in Electrons Affinity • EA decreases down a group • EA increases across a period

Review • Write the electron configuration for Sulfur in orbital, shorthand, and noble gas notation.

Review • Write the electron configuration for Chromium in orbital, shorthand, and noble gas notation.

Review • Explain why a Mg atom is smaller than both Ca and Na atoms.

Review • Explain why a K atom has a smaller ionization energy than both Ca and Li atoms.

Review • The two ions and each have 18 e-. Which would you expect to have a smaller radius? Justify your answer. + K 2+ Ca

Review • Atomic size • IE • Ionic Size • EN • EA F F F Br Br Br

Review • Atomic size • IE • Ionic Size • EN • EA Cs Cs Cs Ba Ba Ba
- Slides: 35