PRACTICALS CARBOHYDRATES 1 Benedicts test Add 8 drops

PRACTICALS CARBOHYDRATES 1

Benedict’s test ü Add 8 drops of glucose solution into a test tube containing 5 m. L of Benedict’s solution and boil for 2 minutes over a Bunsen flame/boiling water bath. ü Allow the test to cool at room temperature and observe the precipitate. 2
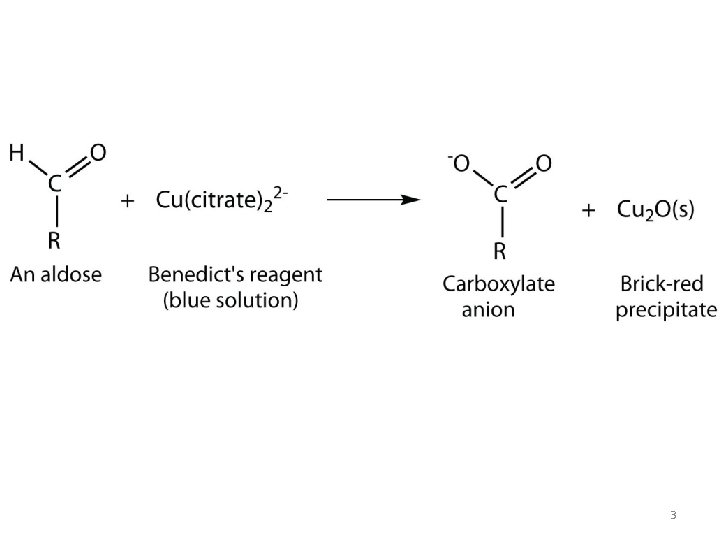
3

Identification of reducing property of sugars ü Repeat the first experiment for glucose, maltose & Sucrose at the same time. ü Observe the color change/precipitate & explain your observation 4

Fehling’s test ü Take 1 m. L of Fehling’s solution into a test tube and 6 drops of glucose solution and keep it in a hot water bath for five minutes. ü Observe the brick red precipitate 5

Molisch’s test Principle All carbohydrates (monosaccharides, disaccharides and polysaccharides) give a positive reaction for Molisch test. It is based on the dehydration of the carbohydrate by Sulphuric acid to produce an aldehyde, Aldehyde condenses with two molecules of α-naphthol, resulting in appearance of a purple ring at the interface 6

Test ü To a 2 m. L of glucose solution add 4 drops of 1% solution of αnaphthol in alcohol and tilt the tube carefully without mixing. ü Add 2 m. L of concentrated sulfuric acid by pouring it down the side of the tube. 7

Barfoed’s test Principle Reducing monosaccharides are oxidized by the copper ion in solution to form a carboxylic acid and a reddish precipitate of copper (I) oxide within three minutes. Reducing disaccharides undergo the same reaction, but do so at a slower rate. 8

Test ü Add 1 m. L of sugar solution to a 3 m. L of freshly prepared Barfoed’s reagent and mix well ü Place in a boiling water bath for 1 minute ü Allow to stand at room temperature for 5 minutes ü Perform this test for both glucose and maltose and compare the time takes for the formation of red precipitate 9

10

Seliwanoff’s Test Principle Seliwanoff’s reagent contains a non-oxidizing acid (HCl) and resorcinol. When a ketose is reacted with this reagent, it becomes dehydrated and a cherry-red complex forms (not a precipitate). 11

ü Add 10 drops of seliwanoff reagent solution into a test tube containing 2 m. L of Fructose solution. ü Mix well and place the test tube in the boiling water bath for 10 minutes. ü Observe the cherry red colour solution. . 12

Test for polysaccharides ü Add 1 drop of iodine solution into 1 m. L of starch solution. ü Observe the blue-black colour solution. ü Repeat the same test for glycogen and observe the colour change ü Explain your observation 13

14

Hydrolysis of Starch/sucrose ü Place 3 m. L of 2% starch in two test tubes and 3 m. L of 2% sucrose solution in two more test tubes. ü To one sample each of sucrose and starch, add 20 drops Of 10% HCl. ü To the other samples of sucrose and starch, add 20 drops of H 2 O. 15

ü Label the test tubes and place in a boiling water bath for 10 minutes. ü Remove the test tubes from water bath and let them cool. ü To the samples containing HCl add 10% Na. OH (about 20 drops) until one drop of the mixture turns litmus paper blue, indicating the HCl has been neutralized. ü Perform the iodine test and Benedict’s test all four samples 16
- Slides: 16