Practical Ways to Use Hansen Solubility Parameters AIMCAL

Practical Ways to Use Hansen Solubility Parameters AIMCAL R 2 R Conference October 31, 2018 - Phoenix, AZ Clean Room Coating Solutions Dr. Ken Mc. Carthy Clean Room Coating Solutions 151 Flint Locke Drive Duxbury, MA 02332 617 -549 -4620(C) kenclnrm@prodigy. net

Practical Ways to Use Hansen Solubility Parameters Today we will talk about Hansen Solubility Parameters • Background • What they are • Finding a substitute solvent • Determining HSP for a new material • Replacing a good solvent with two bad solvents • Insoluble materials • Drying considerations

Practical Ways to Use Hansen Solubility Parameters 1. Background Hansen solubility parameters were developed by Charles M. Hansen in his Ph. D thesis in 1967 as a way of predicting if one material will dissolve in another and form a solution. They are based on the idea that like dissolves like, where one molecule is defined as being 'like' another if it bonds to itself in a similar way. Wikipedia

Practical Ways to Use Hansen Solubility Parameters 2. What they are: Specifically, each molecule is given three Hansen parameters, each with the units of MPa 0. 5: • The energy from dispersion forces between molecules (van der Waals, polarizabilty, correlates with refractive index) • The energy from dipolar intermolecular force between molecules • The energy from hydrogen bonds between molecules. Wikipedia

Practical Ways to Use Hansen Solubility Parameters These three parameters can be treated as co-ordinates for a point in three dimensions, also known as the Hansen space. The nearer two molecules are in this threedimensional space, the more likely they are to dissolve into each other. To determine if the parameters of two molecules (usually a solvent and a polymer) are within range, a value called an interaction radius (R 0) is given to the substance being dissolved. This value determines the radius of the sphere of that substance in Hansen space and its center is the three Hansen parameters. Wikipedia
Practical Ways to Use Hansen Solubility Parameters

Practical Ways to Use Hansen Solubility Parameters The Wikipedia description above does not tell us how R 0 is determined. We will discuss that determination a little later. In essence, R 0 is determined by the solvents that are compatible with, “like”, the material of interest.

Practical Ways to Use Hansen Solubility Parameters The distance between a solute and a solvent determines solubility. Wikipedia

Practical Ways to Use Hansen Solubility Parameters Professor Steven Abbott, in conjunction with others, has developed several apps that make the use of Hansen solubility parameters much more accessible to workers in this field. This presentation will explore the power present in these apps and show they can be used to guide development work in making solutions. Abbott. Apps for Solutions and Dispersions Prof Steven Abbott TCNF Ltd Visiting Professor University of Leeds Presentation at the AIMCAL FTC, Oct. , 2014 www. stevenabbott. co. uk Solubility Science: Principles and Practice (free book) Now, we will look at finding a replacement solvent.
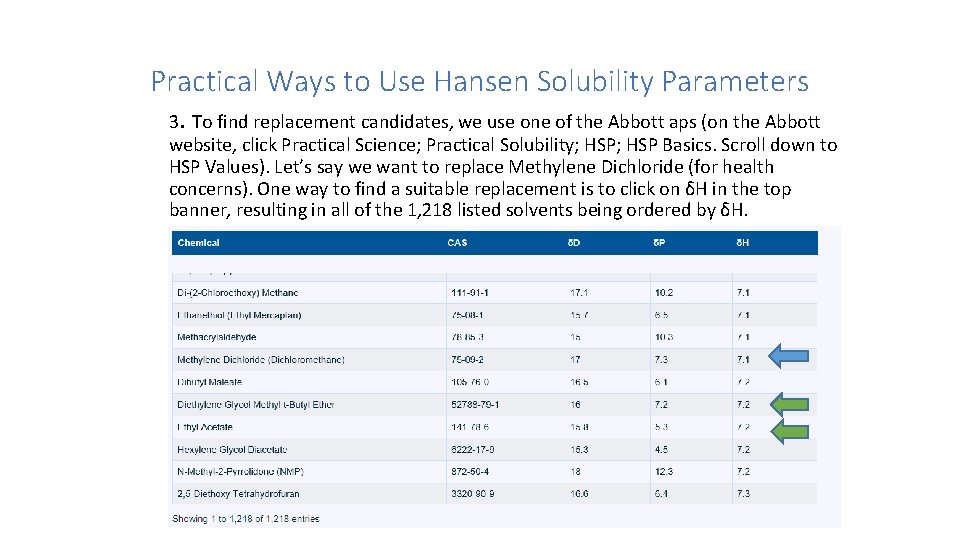
Practical Ways to Use Hansen Solubility Parameters 3. To find replacement candidates, we use one of the Abbott aps (on the Abbott website, click Practical Science; Practical Solubility; HSP Basics. Scroll down to HSP Values). Let’s say we want to replace Methylene Dichloride (for health concerns). One way to find a suitable replacement is to click on δH in the top banner, resulting in all of the 1, 218 listed solvents being ordered by δH.

Practical Ways to Use Hansen Solubility Parameters 3. Finding a Replacement Solvent Scroll down to Methylene Dichloride (HSP values are 17, 7. 3, and 7. 1), we find δH is 7. 1. (See table) We search for solvents that are “close” to it. Two solvents are near-by, Diethylene Glycol Methyl t-Butyl Ether (HSP values are 16, 7. 2, and 7. 2) and Ethyl Acetate (HSP values are 15. 8, 5. 3, and 7. 2). Lab dissolution tests can confirm if these solvents are close enough to replace Methylene Dichloride in the solution of interest.

Practical Ways to Use Hansen Solubility Parameters 4. Finding HSP for a New Material (next four slides) We now return to Professor Abbott’s description of spheres (www. hansensolubility. com; HSP Science; HSP for Beginners) and how they are determined. The calculation and representation of the sphere for a given solute requires the HSPi. P software package. Cost for a single license is $1195, with modest volume discounts.

Practical Ways to Use Hansen Solubility Parameters It has been found that perfect HSP matches are not required for complete solubility, however. If one tests many solvents to see whether they are good or bad for the sticky spruce resin, one finds that the good ones clump together in a three dimensional plot that forms a sphere in the three dimensional HSP space with the HSP of the sticky spruce resin in the middle. There are characteristic differences in HSP in all three parameters that are allowed for good compatibility. These determine the radius of the sphere. Good solvents are within the sphere while bad ones are located on the outside. Steven Abbott
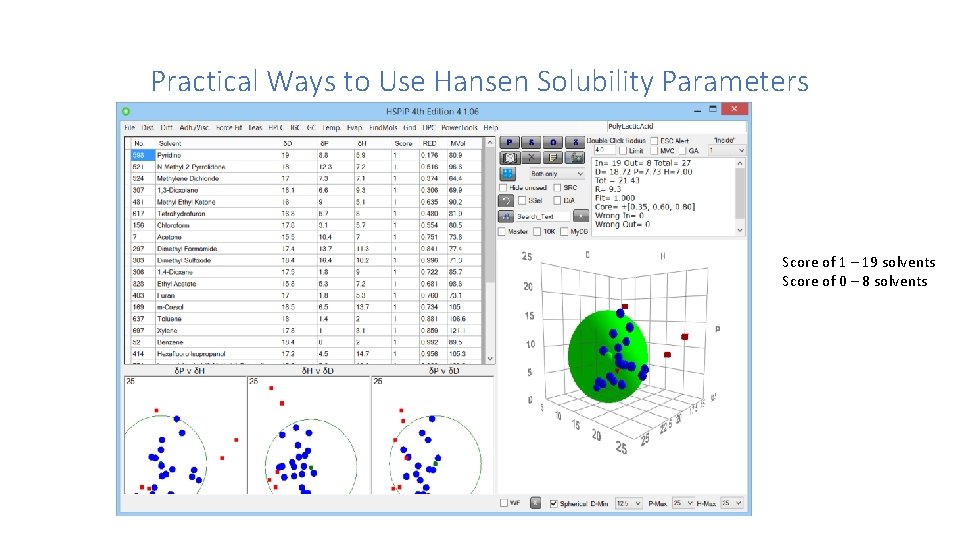
Practical Ways to Use Hansen Solubility Parameters Score of 1 – 19 solvents Score of 0 – 8 solvents

Practical Ways to Use Hansen Solubility Parameters How many solvents should be tested? Most of the cases presented look at 20 - 30 solvents that cover a good part of “HSP” space. You can download a free spreadsheet with HSP functions from: http: //www. stevenabbott. co. uk/Practical. Solubility/HSP_Calculations. xlsx This spreadsheet can provide R values for solvents, but does not give the all-important sphere graphically.

Practical Ways to Use Hansen Solubility Parameters 5. Replacing a “good” solvent with two “Bad” solvents When Charles Hansen first came up with his HSP scheme, he saw that it made a surprising prediction. That two bad solvents (the blue dots in the image) on opposite sides of the HSP Sphere should produce an excellent solvent (green dot in the middle) when mixed 50: 50. If his HSP ideas were wrong, then mixing two bad solvents would create another bad solvent. When he did the test his ideas were confirmed - you really can make a good solvent from a mixture of bad ones. This is probably the single most useful idea in the whole of HSP. It allows formulators amazing freedom to combine two solvents that are attractive in terms of cost, safety, odor, volatility etc. but which are poor solvents for the specific system. The blend of the two creates an excellent solvent. Steven Abbott

Practical Ways to Use Hansen Solubility Parameters Replacing a “good” solvent with two “Bad” solvents It is important to realize that the HSP of the resultant mixture of solvents is proportional to the volume ratio of the two solvents. So, it is not necessary that the mix be 50: 50. That is particularly the case where the solvents are not precisely equally opposite to the solute’s sphere. The only requirement is that the resultant HSP of the mix ends up within the sphere of the solute.

Practical Ways to Use Hansen Solubility Parameters 6. What about “insoluble” materials? Steven Abbott
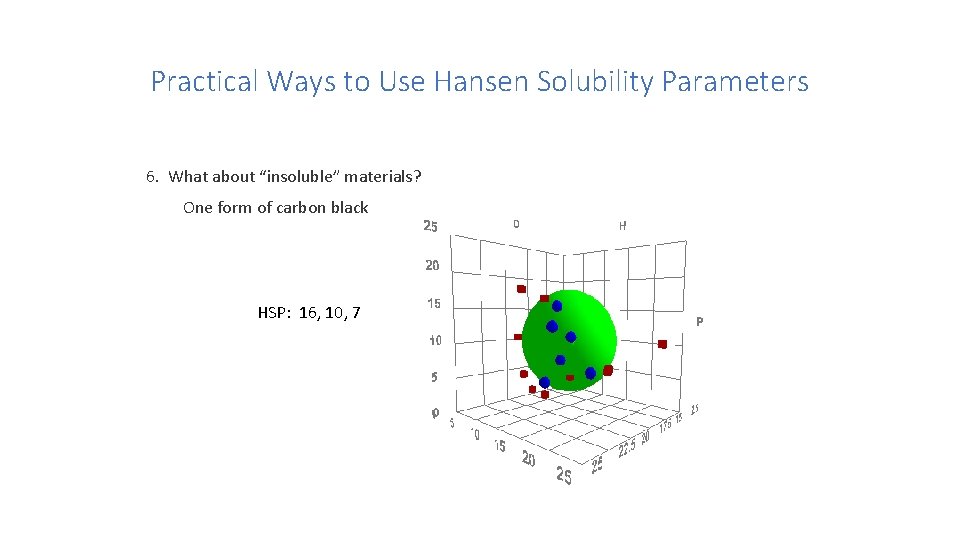
Practical Ways to Use Hansen Solubility Parameters 6. What about “insoluble” materials? One form of carbon black HSP: 16, 10, 7
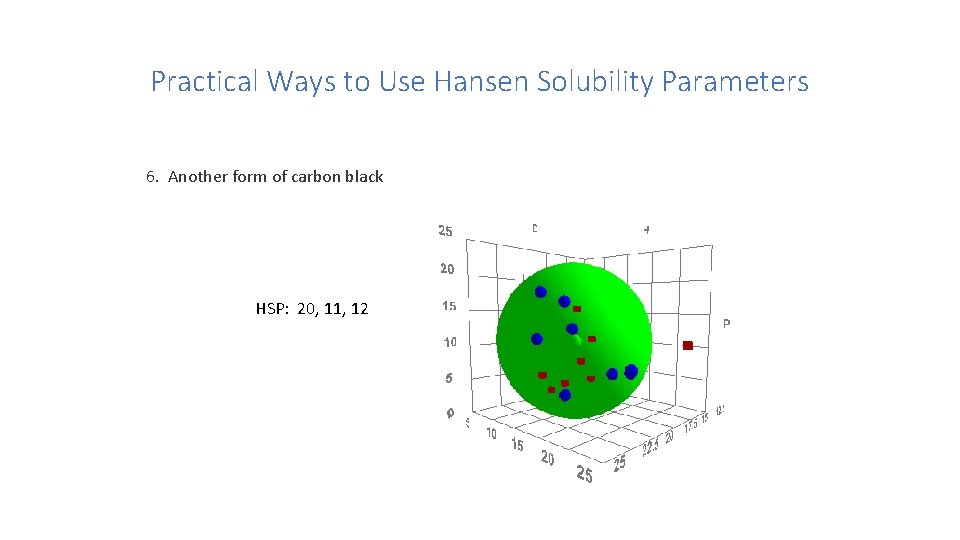
Practical Ways to Use Hansen Solubility Parameters 6. Another form of carbon black HSP: 20, 11, 12

Practical Ways to Use Hansen Solubility Parameters 6. What about “insoluble” materials? Comparison of the two forms of carbon black Steven Abbott

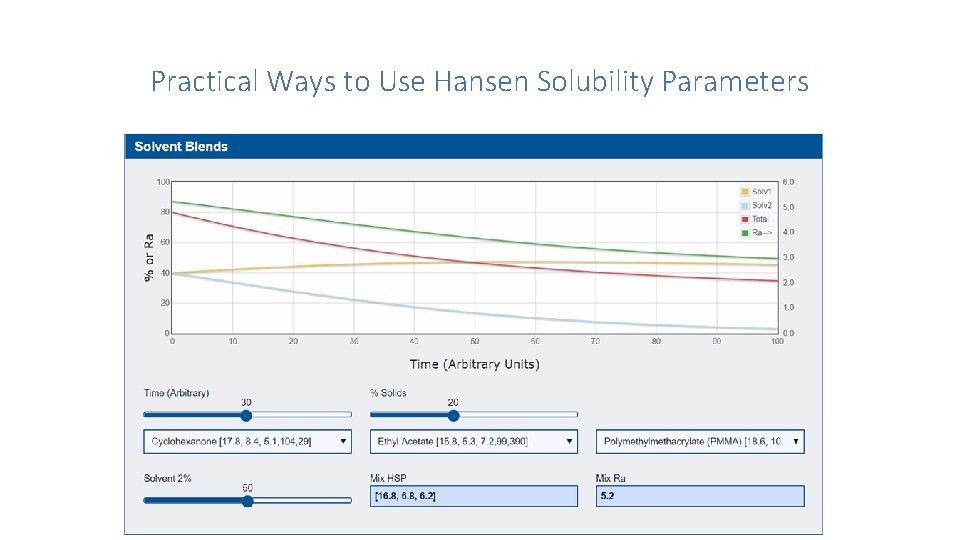
Practical Ways to Use Hansen Solubility Parameters 7. Drying Considerations In this section, Professor Abbott describes how to handle cases where two solvents are being used as a substitute for a given solvent, but also, during the drying process, there is a need to precipitate the solute at a particular time, i. e. , for microencapsulation. Here, understanding where the system is in Hansen space is vital to making the correct choice in adjusting the solvent mix to get the precipitation at the right moment. It is also important to consider the evaporation rates of the two solvents. There is an Abbot App for evaporation rates, shown on P. 80 of Solubility Science: Principles and Practice, the free e-book available on Abbott’s web site. The direct link to the app is: https: //www. stevenabbott. co. uk/practical-solubility/solvent-blends. php
Practical Ways to Use Hansen Solubility Parameters
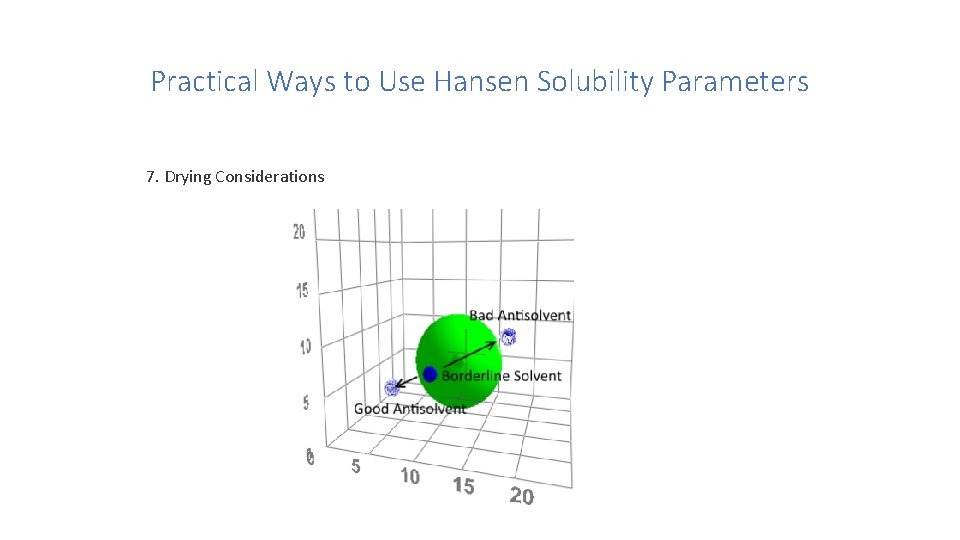
Practical Ways to Use Hansen Solubility Parameters 7. Drying Considerations

Practical Ways to Use Hansen Solubility Parameters 7. Drying Considerations The idea that the addition of a bad solvent can make a better solvent has an negative consequence for those who wish to use anti-solvents to important precipitate a solute at a chosen moment, e. g. for microencapsulation. If, as in the diagram, we suppose that we have a borderline good solvent then we can precipitate the solute with either of the two bad solvents. The "good" anti-solvent would be excellent because just a small amount will take the system outside the sphere. The "bad" anti-solvent would be a disaster because by adding it, the solubility will actually increase (heading to the centre of the sphere) before decreasing and finally precipitating the solute.

Practical Ways to Use Hansen Solubility Parameters Today we have talked about Hansen Solubility Parameters • Background • What they are • Finding a substitute solvent • Determining HSP for a new material • Replacing a good solvent with two bad solvents • Insoluble materials • Drying considerations

Practical Ways to Use Hansen Solubility Parameters Summary In summary for this presentation, Hansen Solubility Parameters and the Abbott application apps give to formulators and process engineers a lot of valuable insights into how materials interact and can be used in a variety of applications.

Thank You Questions? Practical Ways to Use Hansen Solubility Parameters AIMCAL R 2 R Conference October 31, 2018 - Phoenix, AZ Clean Room Coating Solutions Dr. Ken Mc. Carthy Clean Room Coating Solutions 151 Flint Locke Drive Duxbury, MA 02332 617 -549 -4620(C) kenclnrm@prodigy. net
- Slides: 28