Practical Problems in Pediatric Parenteral Drug Administration Pediatric

Practical Problems in Pediatric Parenteral Drug Administration • Pediatric needs vary from adults due to: • smaller dose volumes (smaller doses based on body wt. or body surface area) • reduced fluid requirements (as per organ function + excretion rates)

Subcutaneous Injections Use intermittent: insulin, heparin, MMR vac. continuous: morphine, deferoxamine • 58 inch, < 25 gauge needle • 1. 5 m. L max volume • into the thigh of infants or deltoid area of older children beneath skin and fat but above muscle

Intramuscular Injections Use: • medications that are irritating if given sc • faster absorption and larger volume than sc • if IV route not available for some meds • compliance if patient not taking oral meds • administration of vaccines (i. e. . DPT polio)

Intramuscular Injections • • 23 gauge, 1 inch needle 3 m. L maximum volume (5 m. L in adults) 0. 5 m. L max. volume for deltoid area infants - anterolateral thigh provides largest muscle mass or the rectus femoris (more painful) - avoid medial thigh due to major blood vessels and nerves

Intramuscular Injections • 2 -3 yr - the ventrogluteal area (not into buttock due to sciatic nerve) or deltoid area • older children - the deltoid muscle or the posterolateral aspect of the gluteal area (not into buttock)

Injecting Subcutaneous Medication Via An Insuflon Use: • Many sc medications: i. e. heparin, low molecular weight heparin, DDAVP, filgrastim (G-CSF), and interferon • Used in adults for insulin administration • Not appropriate for all drugs: i. e. growth hormone as induration around insuflon will occur
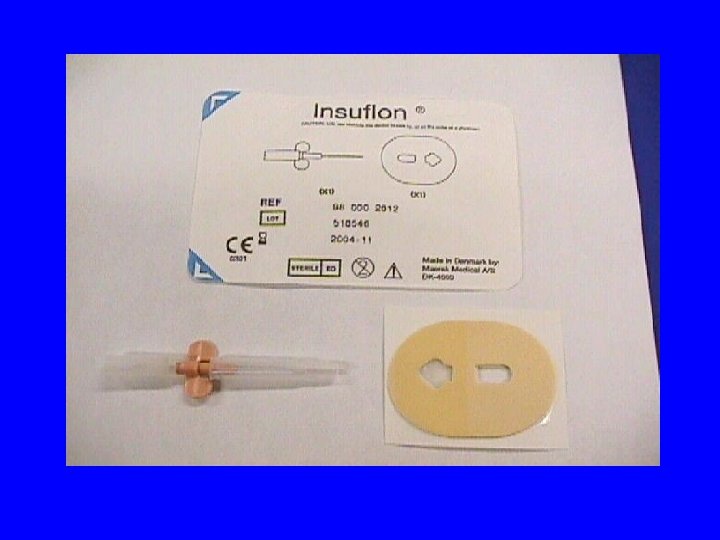

Advantages of SC Administration via an Insuflon • Catheter dead space is very small 0. 0075 m. L • Flush medication into patient with small amount of compatible solution if necessary • 7 days max. indwelling time(3 days for insulin in adults) • rotate insertion sites to avoid tissue damage • allows daily sc administration into canula without daily needle poke

Advantages of Intravenous Injections • Complete and rapid drug absorption with rapid onset of action • Immediate access to cardiovascular system • Useful in neonates with little muscle mass • Less painful route for frequent injections • Administer drugs which cannot be given by another route

Disadvantages of IV Route • Rapid drug/fluid delivery means immediate onset of adverse reactions and inability to withdraw infused solutions • Risk infusion of air, microorganisms, pyrogens and particulate matter • Risk sepsis (infection), phlebitis (venous irritation), extravasation/infiltration (leaking outside of the vein)

IV Access Sites • Peripheral Sites: -vein in hand or forearm -scalp vein or foot vein in infant (possible but central IV site preferred in neonates) • Central IV Sites: subclavian vein into superior vena cava -central line inserted peripherally -umbilical vein in neonates
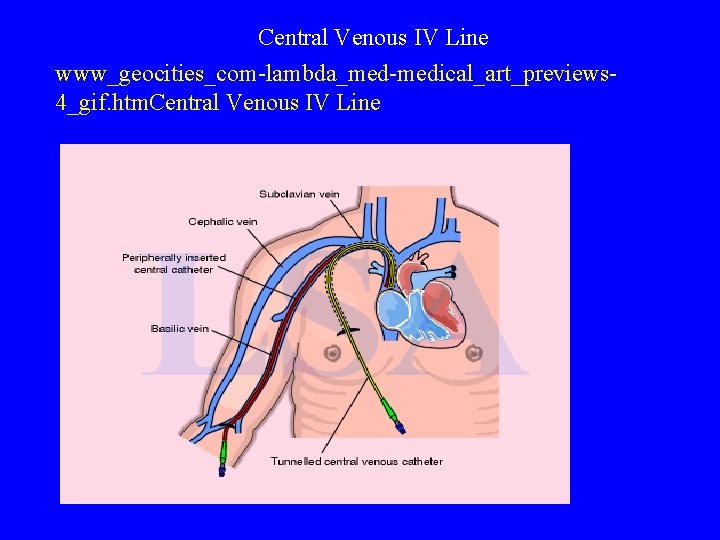
Central Venous IV Line www_geocities_com-lambda_med-medical_art_previews 4_gif. htm. Central Venous IV Line

Factors Influencing IV Drug Delivery • • • small dose volume slow infusion rate dead space volume drug specific gravity infusion device used

Small Dose Volumes • many pharmaceutical manufacturers do not provide suitable concentrations of meds for pediatric administration • require prior dilution in order for dose to be measurable (ie. Dose volume < 0. 5 m. L) • potential for dosing errors secondary to unsuitable drug formulations and dilution procedures

Drug . Available Conc. Diluent Final Conc.

Small Dose Volumes • accurate dose measurement (i. e. with appropriate syringe) • small syringe with integral hubless needle for volumes < 0. 5 m. L • provide 0. 05 m. L drug overfill in syringe to fill needle hub if needle must be attached (include overfill information on label)


When Dose Volume is Less Than Fluid Volume in IV Tubing • Provide medication device with 24 hr drug volume + extra volume equal to tubing loss • change tubing and prime with fresh supply of drug Q 24 h • after 24 h - tubing is discarded for infection control and because the drug in the tubing has reached it’s expiry time • drug must be stable in selected diluent for 24 h at room temperature

Slow IV Infusion Rates • cause an increase in the time required to deliver the dose • Slow/low flow rates (1 -10 mlhr) commonly used with neonates and fluid restricted patients • potential for drug to disperse or layer out within macrobore IV tubing when IV flow rate is slow (use microbore tubing instead) • use of microbore (low volume) tubing (diameter 0. 06 - 0. 14 cm) will minimize this effect

Residual Volume (Dead Space) • > 0. 05 m. L of volume found in hub of needle, stopcocks, Y-type or T-type injection ports or in-line filters • overdosing by 0. 05 m. L may occur if syringe is milked • under dosing by 0. 05 m. L may occur if needle is changed after the exact dose is measured (dosing error =5% for a 1 m. L dose volume)
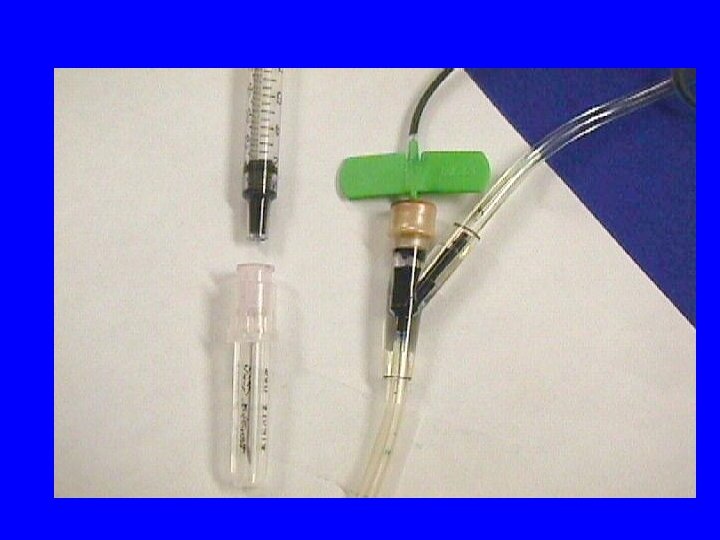
Dead Space

Drug Specific Gravity • in slow moving fluid, the specific gravity can cause the drug to settle, float or pool in a bend of IV tubing and not be administered on schedule

IV Infusion Devices • large volume (e. g. Gemini) or small volume (e. g. CADD) infusion pumps • syringe pumps are accurate for small volume delivery • volumetric infusion devices (Buretrol) used for small total fluid requirements and slow rates of administration • choice of device also depends on frequency of dose (ie. intermittent vs continuous infusion), whether the drug or its vehicle requires special containers and administration sets, and the latex allergy status of the patient.

Health Risks Caused By DEHP • Affects developing reproductive tract (testes) of male fetus, male infants and potentially of pre-pubertal males • infants especially premature infants more susceptible to toxic effects • cardiac toxicity - may affect cardiac transplant patients of all ages • patients receiving multiple transfusions for trauma, hemodialysis patients also at risk

LEACHING OF PLASTICIZER (DEHP) • Medications which are lipophilic or are in vehicles containing lipophilic surfactants (e. g. soy oil emulsion, polysorbate 80) leach significant amounts of DEHP from PVC containers & administration sets • Use glass bottles, polyolefin IV bags (P. A. B. IV bags-B Braun Mc Gaw), ethyl vinyl acetate (EVA) IV bags, polyethylene lined PVC administration sets, or polypropylene syringes.

Drug Incompatibility with Infusion Device • adsorption of drug onto plastic or glass infusion device • a significant portion of the dose may be lost to adsorption if very dilute solutions of the drug are infused • flush device and tubing first with drug infusion solution to saturate binding sites prior to starting infusion • choose low sorbing administration set & tubing , titrate the dose to clinical response


Volume Control Set (Buretrol) • Disadvantages: • Advantages: • dose may have to flow • dilute drug to specific through up to 20 m. L volume appropriate for the of tubing dose (maximum volume 150 m. L in the mixing • small volume doses chamber) given at slow infusion rates will have delayed • Y-in the dose close to delivery patient and/or use microbore tubing

Minibags or Bottles • Advantages: • convenient to dilute the dose in a prefilled IV bag • available in standard sizes 25 m. L, 50 m. L, 100 m. L of D 5 W or Normal saline
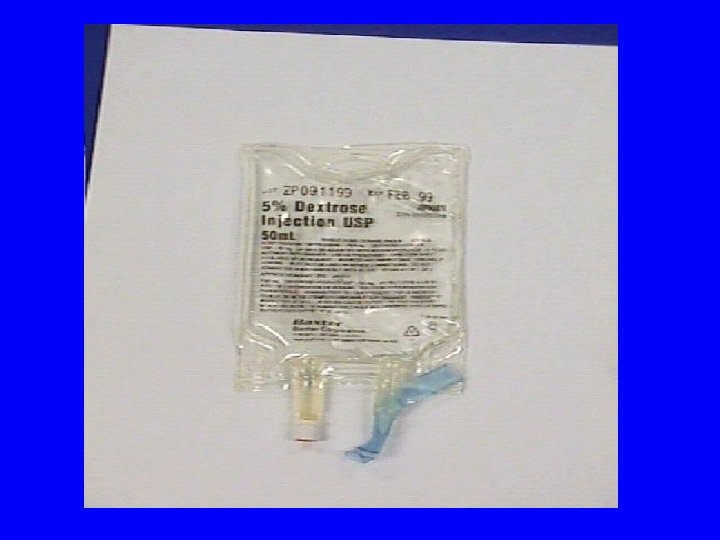
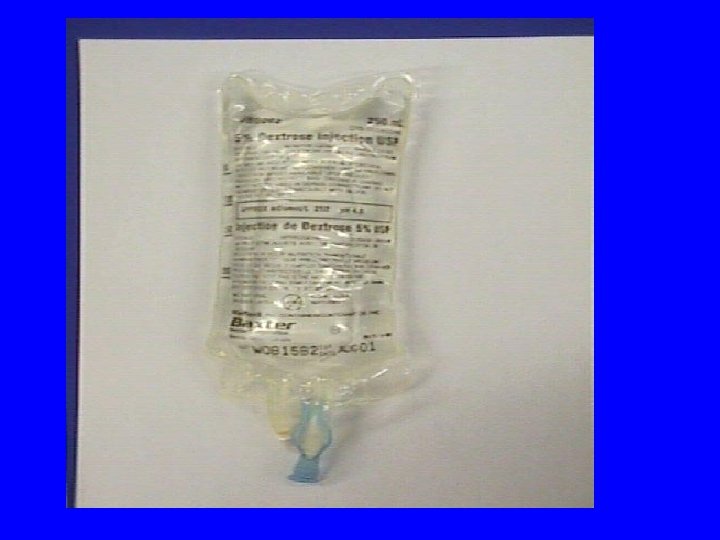

Disadvantages of Minibags or Bottles • may contain more fluid than pt. can tolerate • fixed dilution may provide inappropriate final drug concentration • inaccurate dose delivery due to manufacturer’s overfill unless entire bag is given (25 m. L bag contains 27 -33 m. L or a 24% volume difference)

Disadvantages of Minibags or Bottles • IV set used to administer the dose may retain up to 7 m. L of fluid (23% of dose in 25 m. L bag). This could be flushed into the patient using additional fluid or discarded with a set change or given at the start of the next dose after possible drug degradation. • if attached to primary infusion line, the extra fluid volume causes additional delay in drug reaching the patient

Syringes for Manual IV Injection Advantage: • can inject very small volumes into tubing at injection site near pt. • administration rate controlled by primary IV solution flow rate.

Syringes Manual IV Injection Disadvantages • time for drug delivery depends on drug volume and flow rate of primary IV solution • amount of drug delivered depends on amount lost to dead space at the injection site • requires microbore tubing to decrease delays in delivery • more labour intensive – requires flush


Syringe Pumps Advantages • prepare drug in syringe at required dilution and give via microbore tubing at connection close to patient (reduced fluid volume) • dose given accurately at a rate independent of primary IV solution • used for both intermittent and continuous therapy

Syringe Pumps Disadvantages • only for small volumes < 50 m. L • tubing will retain part of the dose • initial capital investment required

TDM Implications of Factors Influencing IV Drug Delivery • Actual time for dose delivery may be longer than the predicted time due to increases in the distance between the drug injection site and the patient, the total volume to be infused, the specific gravity of the drug, or a slow IV flow rate. • Dose and dose timing errors may occur due to dead space volume errors.

Total Volume Infused and Injection Site • Volume = Dose + Fluid in tubing + Flush (Y-site closest to patient = smallest volume, Y-site above Buretrol = largest volume) • Injection site: to ensure 30 minute drug delivery in pediatric patients, inject IV manually or infuse IV using a syringe pump with microbore tubing at Y-site of IV set closest to patient

Time for Delivery of Chloramphenicol Using Buretrol IV Set with Tubing. Volume 18 m. L • .

Predicted time of chloramphenicol delivery with a Buretrol set • Predicted time to delivery of drug dose (minutes) • = Med. volume + volume of set x 60 min IV Rate (m. Lhour) i. e. . 10 m. L + 18 m. L x 60 min = 336 minutes 5 m. L/hr

Time for Drug Delivery Via Buretrol IV Set • Delivery of a drug added to a Buretrol is affected by slow IV flow rates and too small IV flush volumes. • Medication does not flow toward the patient like a plug, pushing the maintenance IV fluid into the patient without mixing. • Viscosity and specific gravity of medications and maintenance IV fluids vary so mixing will occur within the IV tubing

• The amount of mixing depends on the diameter of the IV tubing and IV flow rate • When using a Buretrol set, increase IV flow rate and flush volume to compensate for the mixing occurring within the IV tubing • Use a flush volume equal to approximately 1. 5 to 2 times the volume of the IV set from the bottom of the Buretrol to the patient • Administer dose plus flushes over required dose administration time

• QUESTION: A 2 kg, 1 month old, neonate is receiving Digoxin 1. 5 mcg/kg/dose IV q 12 h. The physician wishes to increase the dose by 10%. • Is the commercial preparation available in a suitable concentration to allow accurate measurement of a 10% change in the dose? • Which drug delivery devices may be used? • What are sources of error in dose delivery & what effect may this have on the reported Digoxin level?

Solution • Digoxin dose and proposed dose change are appropriate • Digoxin is available as 50 mcg/m. L Inj • Cannot measure a 10% dose change with the commercially available product • Dilute Digoxin with NS to 10 mcg/m. L so 10% dose change is measurable • Device - 1 cc syringe, manual IV injection into Y-site nearest to patient

• Sources of error: syringe dead space volume (0. 05 m. L) and low IV flow rate (1 -10 mlhr in neonate) • 3. 3 mcg dose = 0. 33 m. L of 10 mcg/m. L • 0. 05 m. L x 10 mcg/m. L= 0. 5 mcg (dead space volume is 15% of dose ) • Digoxin level either 15% higher or lower than predicted if measuring inaccurate. Inappropriate timing of Digoxin levels and uninterpretable results if drug delivery delayed.

References • 1. Comprehensive Pediatric Nursing. New York: Mc. Graw-Hill; 1986: Appendix 1. • 2. Leff RD, Roberts RJ. Principles and techniques of IV administration. In Practical aspects of intravenous drug administration. ASHP 1992(2): 4 -41. • 3. Nahata M. Methods of intravenous drug infusion in pediatric patients. The Am J Intravenous Therapy & Clin N 1984: May: 6 -7.

• 4. Hunt, Max L. Training Manual for Intravenous Admixture Personnel Fifth Edition. Baxter Healthcare Corporation & Precept Press, USA, 1995. • 5. Roberts RJ. Intravenous administration of medication in pediatric patients: problems and solutions. Ped Clin N Amer 1981; 28: 23 -34. • 6. The Hospital for Sick Children. Policies and Drug Information for Nurses Manual. Parenteral therapy - not intravenous. Toronto; 2000: 4. 01 -4. 03.

• 7. Rice, Stephen P. , A Review of Parenteral Admixtures Requiring Select Containers and Administration Sets, International Journal of Pharmaceutical Compounding, Vol 6, No 2, MarchApril 2002. • 8. Turco, Salvatore J. , Editor. The Sourcebook for IV Therapy IVAC Corporation, San Diego, Ca, 1985.

• 9. http: //www. hc-sc. gc. ca/hpbdgps/therapeut/zfiles/english/advcomm/eap/deh p/eap-dehp-final-report-2002 -jan-11_e. html Health Canada Expert Advisory Panel On DEHP In Medical Devices, Final Report 220 January 11, Health Canada, 2002. • 10. http: //www. fda. gov/cdrh/ost/dehp-pvc. pdf Safety Assessment of Di(2 -ethylhexyl)phthalate (DEHP) Released from PVC Medical Devices Centre for Devices and Radiological Health, U. S. Food and Drug Administration, Rockville, MD, 2001.
- Slides: 53