Practical medical mycology Lab 5 Direct examination of

ﺑﺴﻢ ﺍﻟﻠﻪ ﺍﻟﺮﺣﻤﻦ ﺍﻟﺮﺣﻴﻢ Practical medical mycology Lab 5 Direct examination of the specimen and other techniques ( part 2 )

A. skin or dermatomycosis KOH for superficial involvement, look for: 1. Spaghetti & meat balls (lung aspirate) Malassezia furfur 2. Pseudohyphae and yeasts (vaginal secretions) Candida species. 3. Hyaline septate hyphae ex: Dermatophytes 4. Dematiaceous septate hyphae ex: Tinea nigra.

H & E stain( Hematoxylin and eosin stain) for on Skin biopsy of tongue, look for: 1. Pseudohyphae 2. yeasts Oral Candidiasis
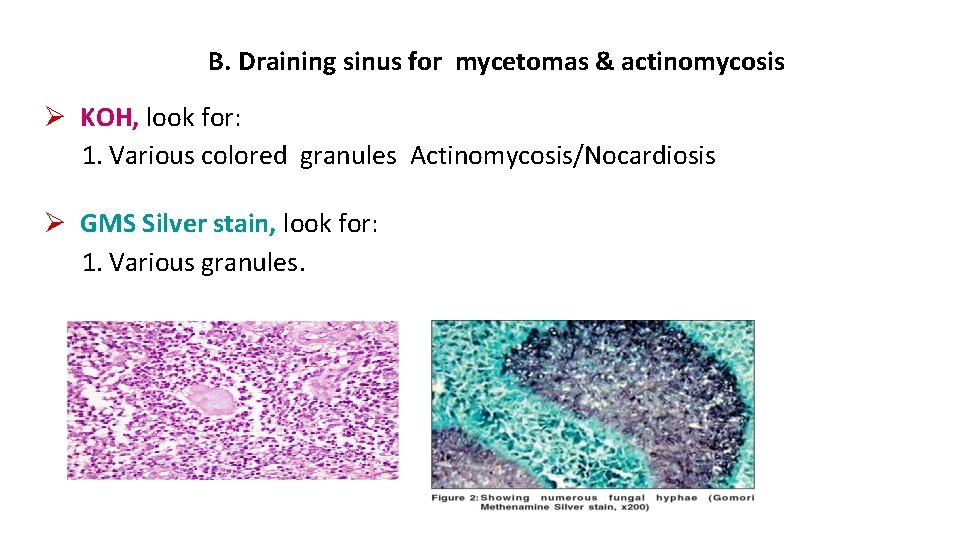
B. Draining sinus for mycetomas & actinomycosis KOH, look for: 1. Various colored granules Actinomycosis/Nocardiosis GMS Silver stain, look for: 1. Various granules.
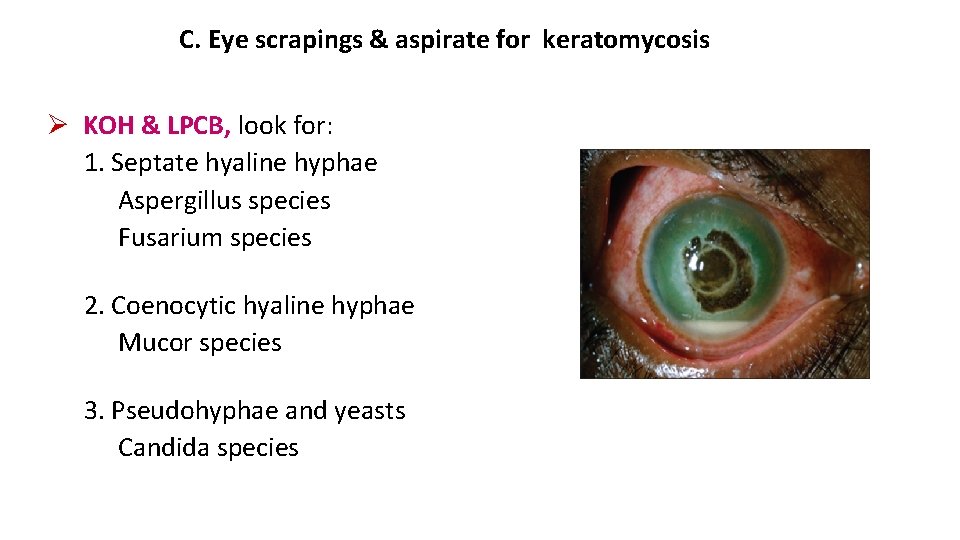
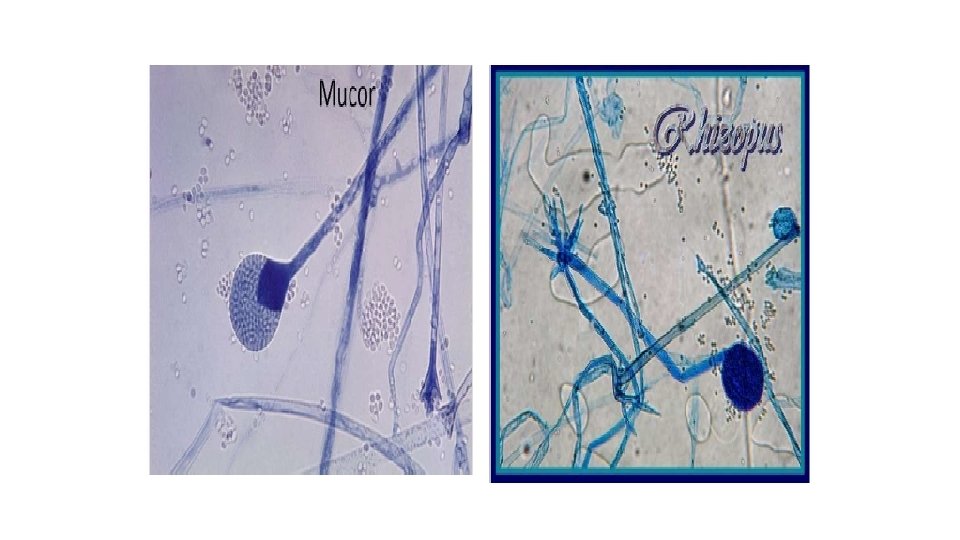
C. Eye scrapings & aspirate for keratomycosis KOH & LPCB, look for: 1. Septate hyaline hyphae Aspergillus species Fusarium species 2. Coenocytic hyaline hyphae Mucor species 3. Pseudohyphae and yeasts Candida species
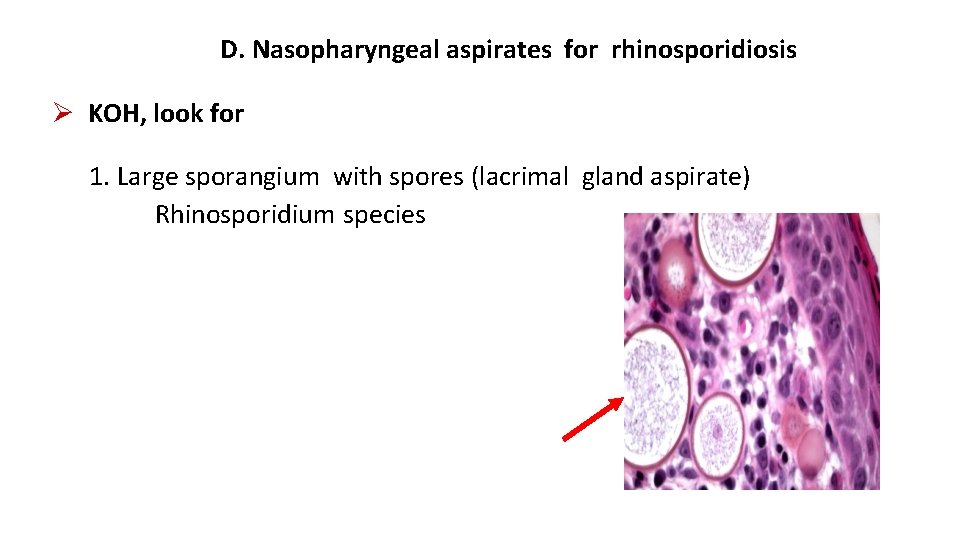
D. Nasopharyngeal aspirates for rhinosporidiosis KOH, look for 1. Large sporangium with spores (lacrimal gland aspirate) Rhinosporidium species
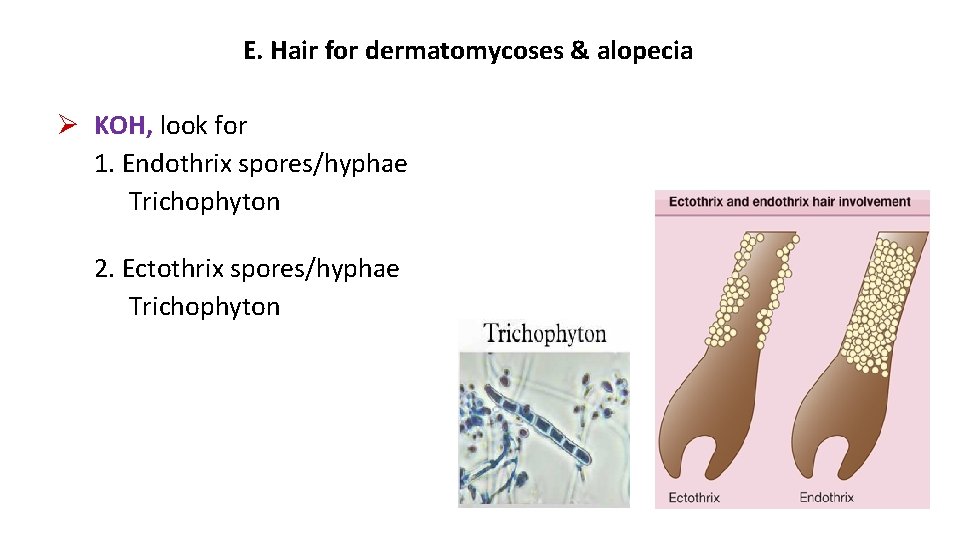
E. Hair for dermatomycoses & alopecia KOH, look for 1. Endothrix spores/hyphae Trichophyton 2. Ectothrix spores/hyphae Trichophyton

E. Hair for piedra KOH, look for: 1. Hard, brown, compact nodules (Black piedra) Piedraia hortae 2. Soft, off-white, concretions/nodules (White piedra) Trichosporon beigeli
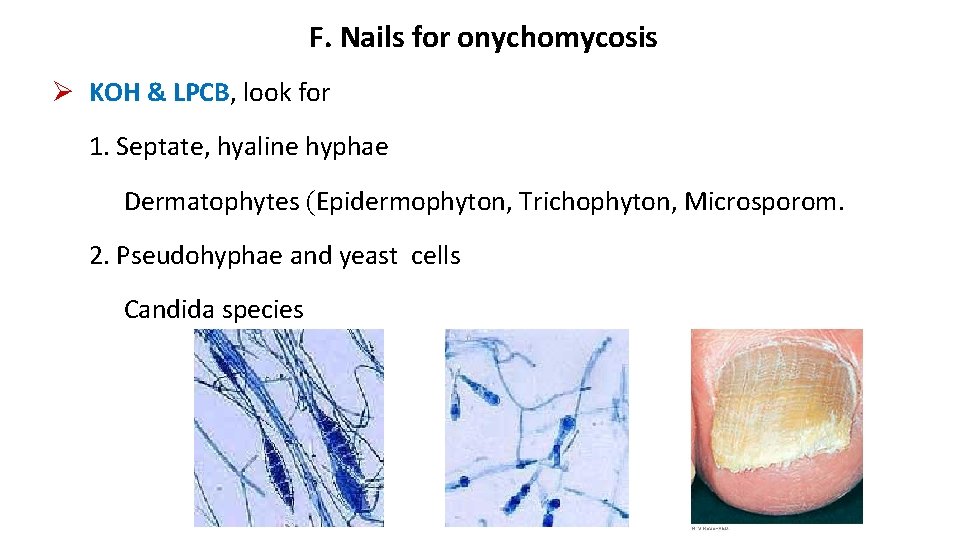
F. Nails for onychomycosis KOH & LPCB, look for 1. Septate, hyaline hyphae Dermatophytes (Epidermophyton, Trichophyton, Microsporom. 2. Pseudohyphae and yeast cells Candida species
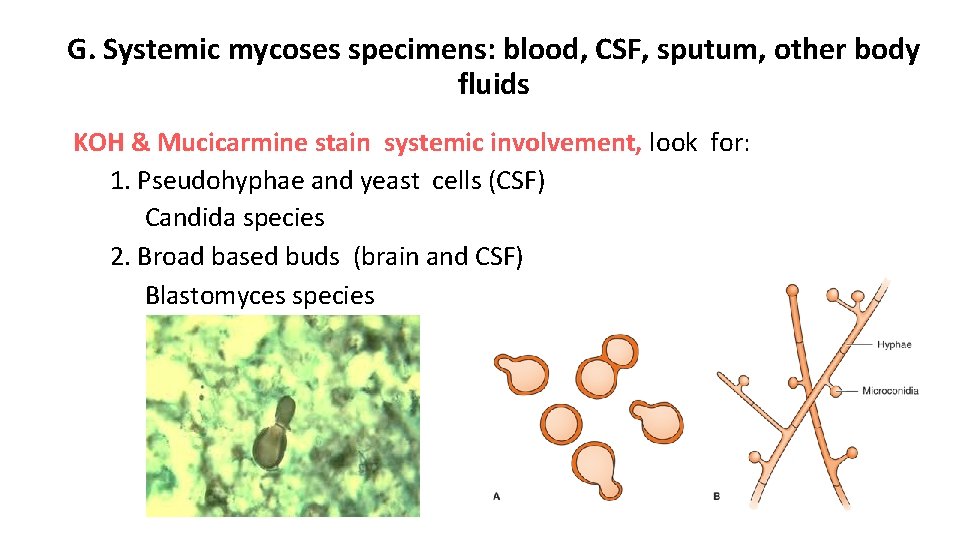
G. Systemic mycoses specimens: blood, CSF, sputum, other body fluids KOH & Mucicarmine stain systemic involvement, look for: 1. Pseudohyphae and yeast cells (CSF) Candida species 2. Broad based buds (brain and CSF) Blastomyces species
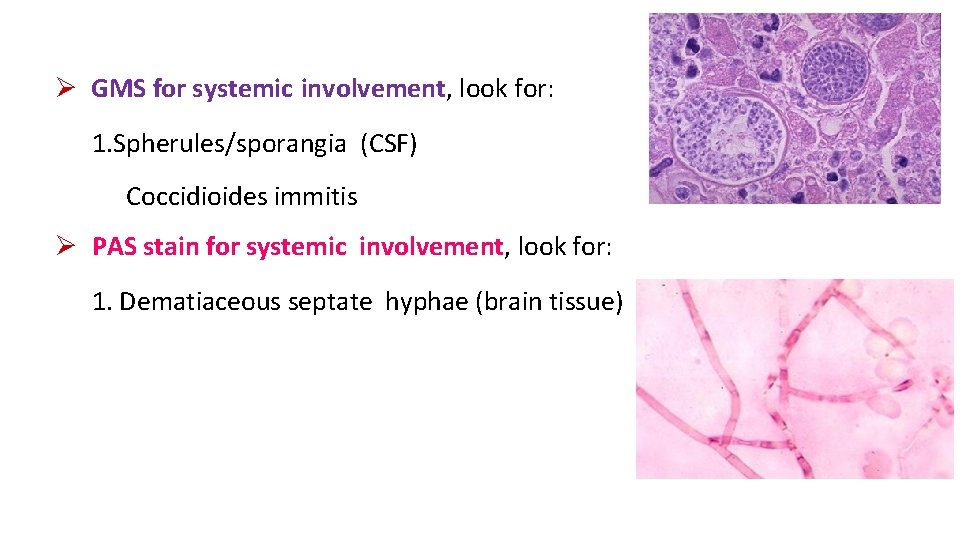
GMS for systemic involvement, look for: 1. Spherules/sporangia (CSF) Coccidioides immitis PAS stain for systemic involvement, look for: 1. Dematiaceous septate hyphae (brain tissue)

Wright’s/Giemsa stain (Diff quick) & LPCB for systemic involvement, look for: 1. Small, intracellular budding yeast (CSF) Histoplasma species. 2. Small, intracellular yeast dividing by fission (CSF) Penicillin species.
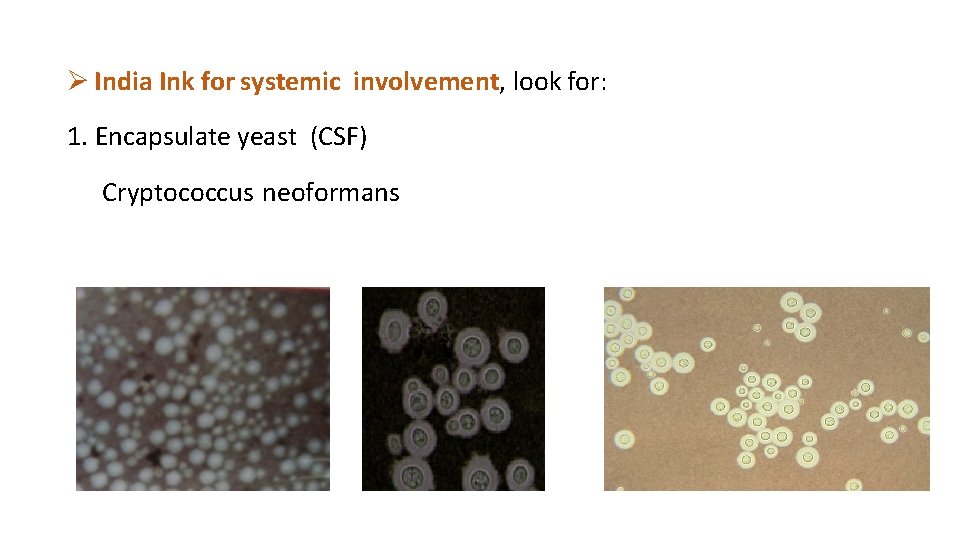
India Ink for systemic involvement, look for: 1. Encapsulate yeast (CSF) Cryptococcus neoformans
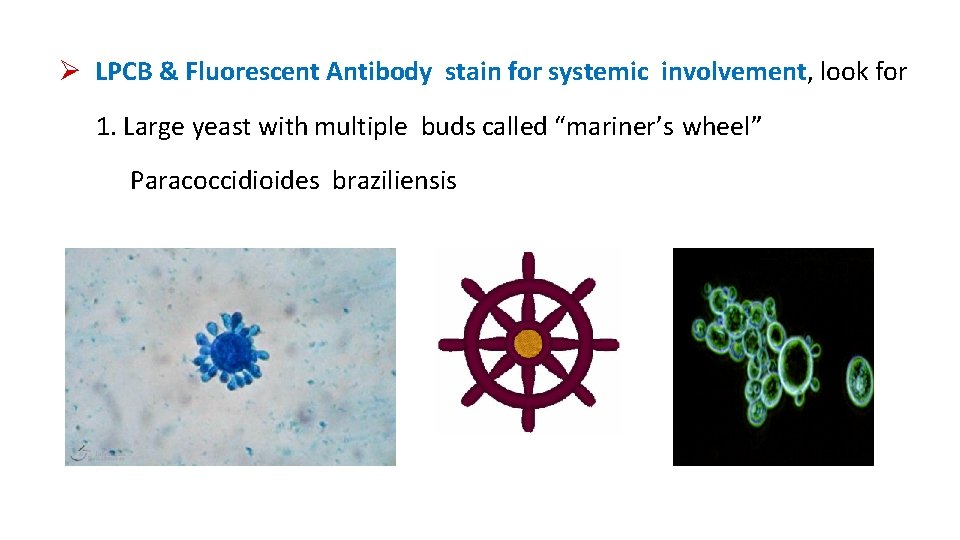
LPCB & Fluorescent Antibody stain for systemic involvement, look for 1. Large yeast with multiple buds called “mariner’s wheel” Paracoccidioides braziliensis
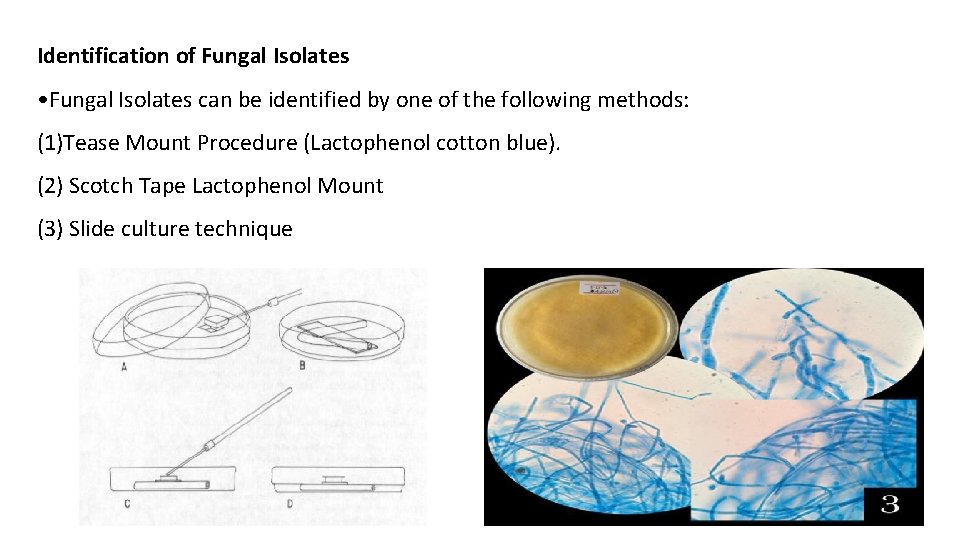
Identification of Fungal Isolates • Fungal Isolates can be identified by one of the following methods: (1)Tease Mount Procedure (Lactophenol cotton blue). (2) Scotch Tape Lactophenol Mount (3) Slide culture technique

Tease mount For microscopic examination , fragment of fungal culture is teased out using two teasing needles and placed on a glass slide in drop of LCB stain To study the morphology of hyphae, spores and other structures, tease mounts are prepared in lactophenol cotton blue (LCB) which contains lactic acid, phenol, glycerol and cotton blue Microscopic characteristics that should be observed are the following: 1. Sepatate versus sparsely septate hyphae 2. Spores or conidia

Preparation of Lactophenol cotton blue (LPCB) slide mounts • Place a drop of 70%alcohol on a clean microscope slide. • Material from cultures of filamentous fungi should be removed using a stiff inoculating wire. • Remove a small amount of the culture. For fungal cultures, Immerse the fungal material in the drop of 70%alcohol • Before the alcohol dries out add one or at most two drops of the stain lactophenol cotton blue to the slide • Cover by cover slip& examine under the microscope • Make the initial examination using a low power objective lens. The thinner parts of the preparation, generally around the edges of the mounted material, will yield the best images. • Switch to a higher power 40 X objective for more detailed examination of spores and other structures.
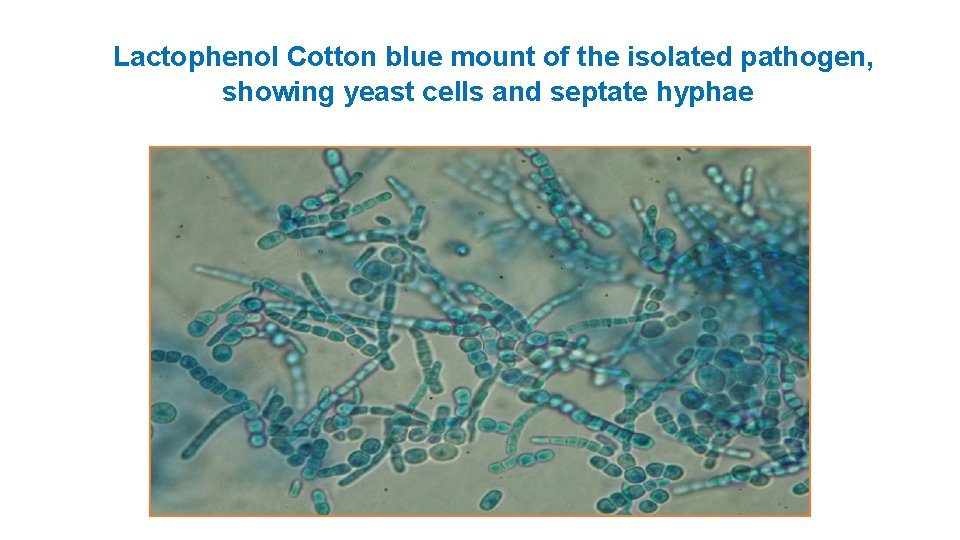
Lactophenol Cotton blue mount of the isolated pathogen, showing yeast cells and septate hyphae
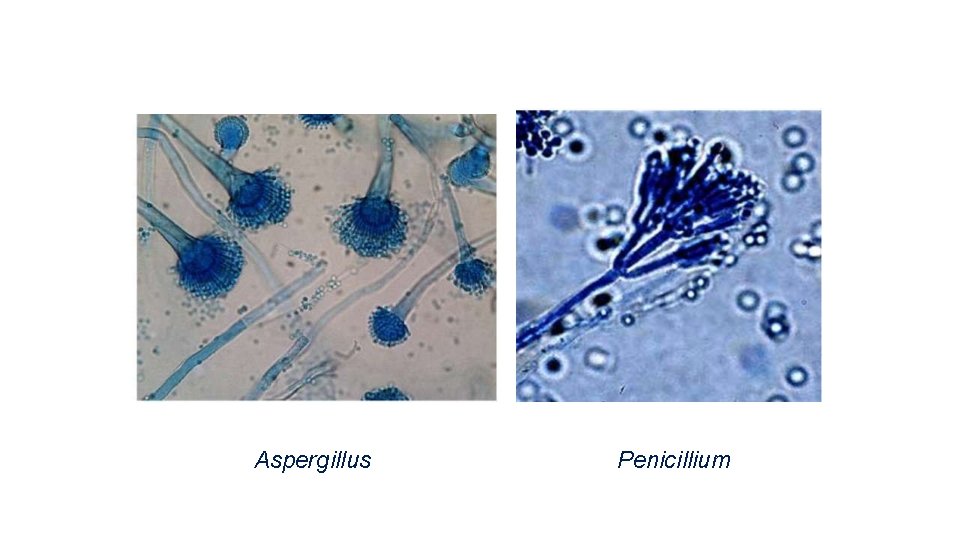
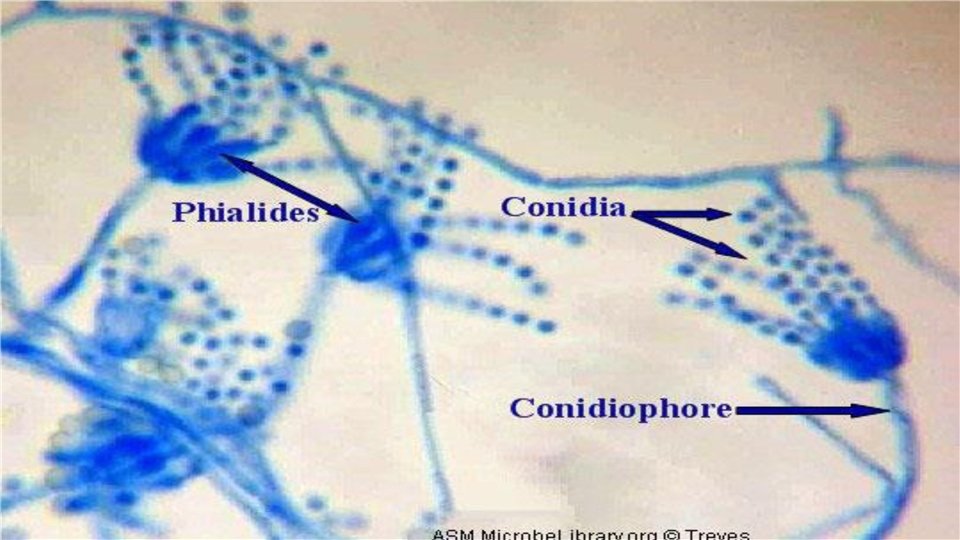
Aspergillus Penicillium


2 -Scotch Tape Lactophenol Mount • The Scotch tape mount is an easy and fast procedure that is used for the identification of filamentous fungi since most structures will be intact for observation thank to the gummed side of the tape. • As with the lactophenol mount, the organism will be immersed in the solution, rendering the organism safe for handling outside of the biological safety hood.

• Limitations include: - the tape will dissolve eventually so that it is not to be used for permanent mounts. - The procedure can only be performed on molds growing from plates.

Procedure (1)Cut a strip of Scotch transparent tape and place ends between thumb and index finger, gummed side out. (2) Making a loop by closing fingers, open plate with opposite hand press tape against the colony to identify. (3) Place a drop of lactophenol on a labeled slide. (4) Press tape against slide with lactophenol. (5) Smooth the tape back on the slide by opening fingers and using gauze. (6) Place another drop of lactophenol on top of the tape. (7) Place a large 20 x 40 mm coverslip on top of slide. (8) Examine the slide under the microscope.

Slide Culture A- Slide Culture Preparation • Aseptically, with a pair of forceps, place a sheet of sterile filter paper in a Petri dish. • Place a sterile U-shaped glass rod on the filter paper. (Rod can be sterilized by flaming, if held by forceps. ) • Pour enough sterile water (about 4 ml) on filter paper to completely moisten it. • With forceps, place a sterile slide on the U-shaped rod. • Gently flame a scalpel to sterilize, and cut a 5 mm square block of the medium from the plate of Sabouraud’s agar or Emmons’ medium. • Pick up the block of agar by inserting the scalpel and carefully transfer this block aseptically to the center of the slide.

• Inoculate four sides of the agar square with spores or mycelial fragments of the fungus to be examined. Be sure to flame and cool the loop prior to picking up spores. • Aseptically, place a sterile cover glass on the upper surface of the agar cube. • Place the cover on the Petri dish and incubate at room temperature for 48 hours. • After 48 hours, examine the slide under low power. If growth has occurred there will be growth of hyphae and production of spores. If growth is inadequate and spores are not evident, allow the mold to grow for another 24– 48 hours before making the stained slides.

- Slides: 28