Practical medical mycology Lab 4 Direct examination of

ﺑﺴﻢ ﺍﻟﻠﻪ ﺍﻟﺮﺣﻤﻦ ﺍﻟﺮﺣﻴﻢ Practical medical mycology Lab 4 Direct examination of the specimen and other techniques

The direct examinations are done with both clinical specimens and fungal culture isolates. 1 -Saline wet mount/ Direct mount : used to study yeast morphology microscopically and fungal elements such as hyphae, conidia and budding yeasts and to determine purity of the isolates. 2 -Grams stain : used to view yeast (stain dark purple). Procedure: 1 -Preparation of the smear

2 -Air dry 3 -Heat Fixation 4 -Primary stain : crystal violet (let stand for 1 minute). 5 -Wash with tap water or distilled water using a wash bottle. 6 -Mordant : Gram’s iodine and let stand for 1 minute. 7 -Gently rinse with tap water or distilled water using a wash bottle. The smear will appear as a purple circle on the slide. 8 -Decolorize using 95% ethyl alcohol or acetone. Tilt the slide slightly and apply the alcohol drop by drop for 5 to 10 seconds until the alcohol runs almost clear. Be careful not to over-decolorize.

8 -Immediately rinse with water. 9 -Counter-stain : safranin and let stand for 45 seconds. 10 -Wash with tap water or distilled and view the smear using a lightmicroscope under oil-immersion. 3 -Lacto-phenol cotton blue wet mount : used to stain and preserve fungal elements in culture isolates -Phenol will kill any organisms, while the lactic acid preserves fungal structure -cotton blue stains the chitin in the fungal cell walls.

4. Potassium hydroxide (KOH) wet mount : used to dissolve non-fungal materials (keratin) in skin, nail, and hair samples -makes any fungi more visible. Procedure: 1. Place a drop of (10 -20)% KOH in the center of a clean microscopic slide. 2. Place a fragment of tissue, purulent materials or scraping in KOH drop. 3. Tease the materials well enough with corner of a coverslip to give a thin preparation or break up the materials with a sterile loop. 4. Mount with coverslip.

5. Allow preparation to digest for approximately 10 minutes or longer depending on the tested materials. 6. Gently warm the slide (do not overheat) Gently press on the slide to help disperse tissue materials. 7. Screen under low power objective. 8. Then Use high (40 X) power magnification to verify the presence of fungal elements.

5. India ink : -used to reveal capsules surrounding Cryptococcus neoformans found in CSF. Due to low sensitivity, direct antigen detection assays have replaced the India ink wet mount 6. Calcofluor white stain : -Fluorochrome that stains chitin found in cell walls of fungi -Not absorbed by human tissue -slide viewed with UV microscope fungi will appear white to blue -KOH can be added to clear the specimen of cellular debris.
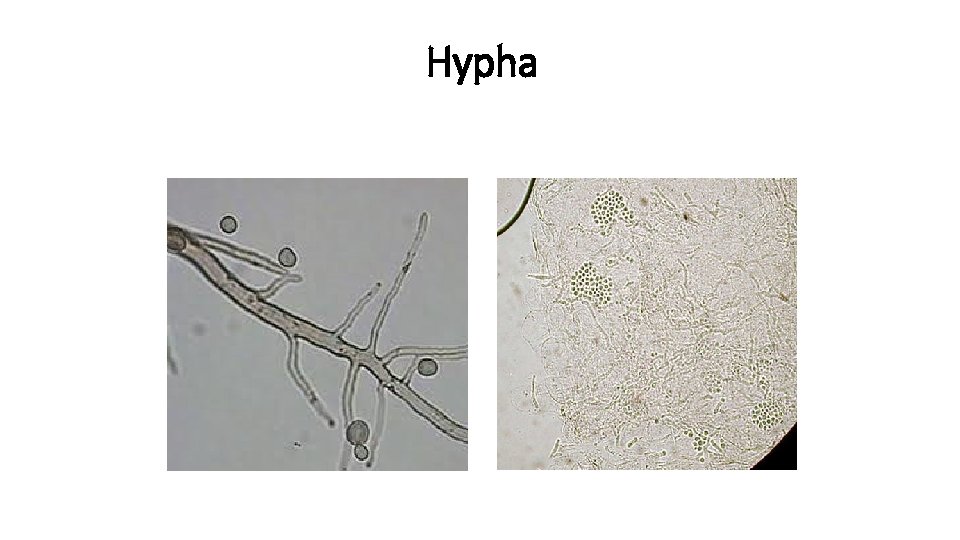
Hypha

Mycelium
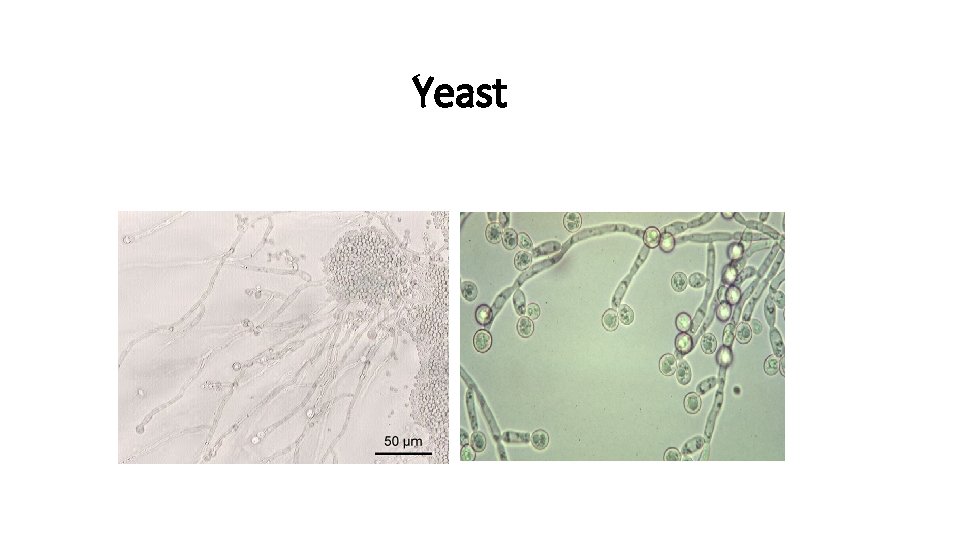
Yeast

7. Periodic Acid - Schiff Stain (PAS) - stains certain polysaccharide in the cell walls of fungi. Fungi stain pink-red with blue nuclei. 8. Gomori. Methenamine Silver Stain - silver nitrate outlines fungi in black due to the silver precipitating on the fungi cell wall. The internal parts of hyphae are deep rose to black, and the background is light green. 9. Papanicolaou Stain - good for initial differentiation of dimorphic fungi. Works well on sputum smears. 10. Giemsa Stain - used for blood and bone marrow specimens. Histoplasma capsulatum is an intra cellular organism.

Serological and skin test Serological tests - demonstrates B-cell (humoral) immunity to a fungus; sera should be drawn in pairs (acute and convalescent). (1) Complement fixation (2) Agglutination tests (3) Precipitin tests (4) Immunofluorescence (5) Immuno-diffusion techniques Skin tests - demonstrates T-cell immunity (cellular) to a fungus.

API 20 C Yeast Identification System • The API 20 C Yeast Identification System consists of 20 cupules containing 19 different carbon source substrates and a negative control. • The system is used according to the manufacturer's instructions, except for modifications made to accommodate the slower, more filamentous growth of some of the dematiaceous fungi. • Modifications included an extended culture incubation time of 8 to 10 days, in contrast to the 2 to 3 days required for yeasts, and an indirect (versus direct for yeasts) inoculum preparation technique, as described below.

Procedure: • Mature (8 - to 10 -day) cultures grown at 30°C on modified Sabouraud glucose agar were used to obtain inoculum suspensions. • The fungal colonies were covered with approximately 1 ml of sterile distilled water. • Suspensions were made by gently probing the colony with the tip of a Pasteur pipette.

• The resulting mixture is withdrawn and transferred to a sterile tube. • The heavy portions of the suspension are allowed to settle, and the upper homogeneous suspension was used as the inoculum. • The molten basal medium supplied by the manufacturer was held at 50°C and seeded with the mycelial-conidial suspensions and then adjusted to a density slightly less than 1 + using a Wickerham card.

• The API 20 C strips are inoculated with a sterile Pasteur pipette by filling each cupule with 0. 2 ml of the seeded medium, as indicated by the manufacturer. • The inoculated strips are wrapped in plastic bags to increase humidity and incubated at 30°C for 8 to 10 days. • Reactions (growth) were read daily for 3 days and on days 8 and 10.

• Cupules showing turbidity (carbohydrate assimilation) significantly heavier than that of the negative control cupule are read as positive. • Positive reactions are compared with that of the glucose cupule, which served as the positive control. • Weak-to-strong reactions are considered positive. • Questionable reactions and absence of growth are considered negative.

- Slides: 18