Practical immunology Immunological tools Immunological methods Antibodies as

Practical immunology Immunological tools

Immunological methods: Antibodies as tools • Specific recognition of macromolecules – Cell types: surface molecules (CD markers) – Pathogens – Antibodies • • Antibodies as labels Antibodies as traps Antibodies as competitors Assays: – Detection – Localization – Quantitation

A brief history of antibodies • 1891: The term “Antikörper” coined by Paul Ehrlich – Substance in the blood that confers immunity – "if two substances give rise to two different antikörper, then they themselves must be different” – “Lock-and-Key” theory • 1920’s: Heidelberger and Avery identified antibodies as proteins • 1940’s: Linus Pauling confirms lock-and-key theory • 1948: Immunoprecipitation: use of antibodies for detection • 1956: Glick and Chang-Bursa of Fabricius: Antibodies come from B cells • 1976: Hozumi and Tonegawa, antibody gene rearrangement

Useful characteristics of antibodies • Specificity: Ability to recognize individual epitopes • Cross-reactivity: – Ability to bind to more than one epitope – Shared epitopes between different antigens • Strength of binding: – Affinity: Strength of binding between Fab and epitope – Avidity: Overall strength of binding of serum and antigen
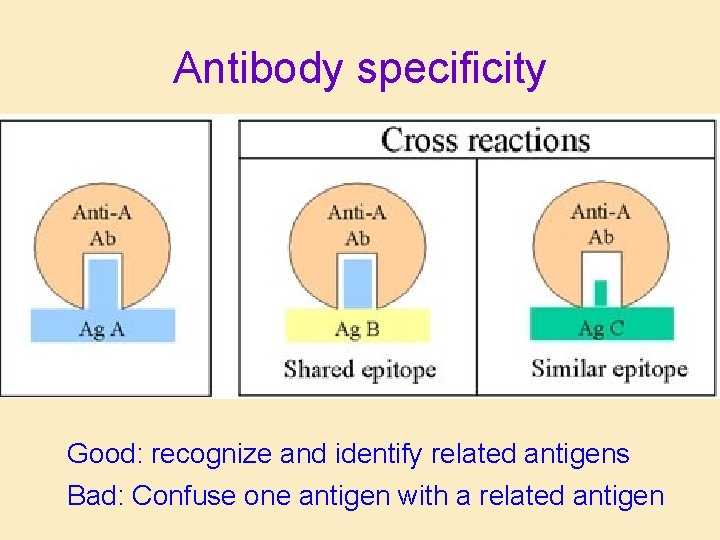
Antibody specificity Good: recognize and identify related antigens Bad: Confuse one antigen with a related antigen
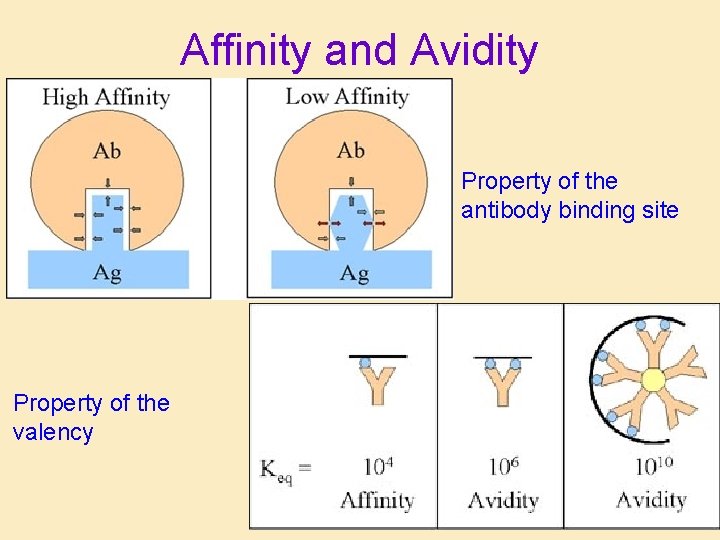
Affinity and Avidity Property of the antibody binding site Property of the valency

Antibody types • Polyclonal antibodies: – Isolated from immune serum – Many different idiotypes – Recognize many different epitopes • Strong avidity • Highly sensitive – Cross reactive: Less specific • Monoclonal antibodies: – – Single idiotype Single epitope Highly specific Lower avidity

Generation of antibodies as reagents • Polyclonal: – Purify antigen – Inject into rabbit or goat (or other species) – Collect serum – Purify immunoglobulin – Specificity depends on purity of antigen • Monoclonal: – Immunize a mouse – Culture spleen cells fused with myeloma cells – Select clones based on antibody specificity – Specificity to a single epitope
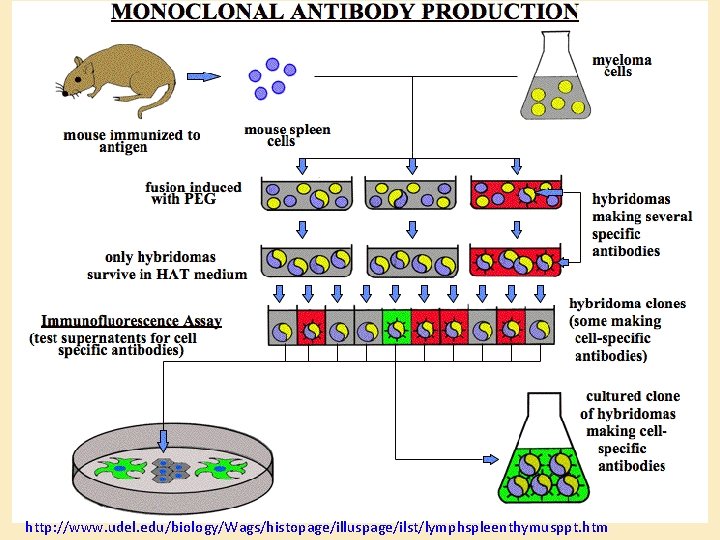
http: //www. udel. edu/biology/Wags/histopage/illuspage/ilst/lymphspleenthymusppt. htm

Immunological assays • Diagnostic: – Assay for antigen – Assay for antibody • Research: – Identify cells – Identify cell products – Isolate cells or macromolecules – Assay cell function

Examples Diagnostic assays Research techniques • Hemaggluination • Hemagglutination inhibition • Enzyme-linked Immunosorbant Assay (ELISA) • Radioimmunoassay (RIA) • Immunofluorescent assay (IFA) • • ELISA Immunohistochemistry Flow cytometry Cell sorting ELIspot Immunoblot Immunoprecipitation Mixed lymphocyte response • Chromium release assay

Types of assays • Precipitation tests: Detection based on precipitation of complexes • Colorimetric tests: – Fluorescent – Enzyme-linked
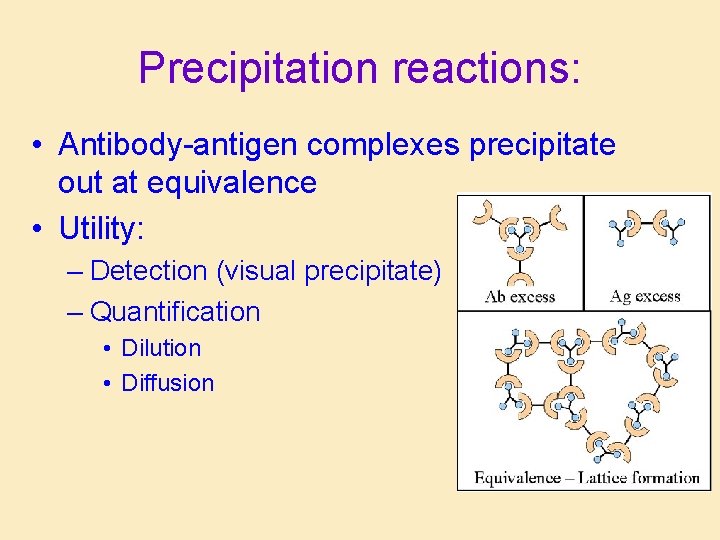
Precipitation reactions: • Antibody-antigen complexes precipitate out at equivalence • Utility: – Detection (visual precipitate) – Quantification • Dilution • Diffusion
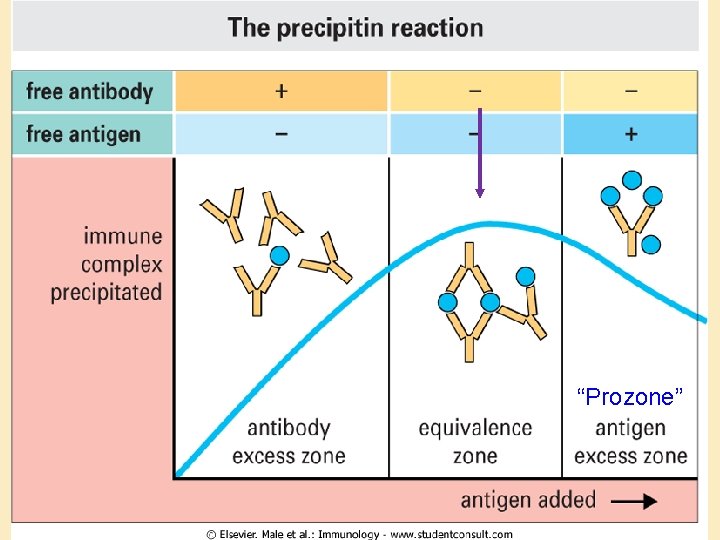
“Prozone”

Precipitin tests: Immunodiffusion • Double immunodiffusion: – Well or trough punched in an agarose gel – Antibody and antigen placed in separate well – Precipitate forms if antibody recognizes antigen • Identification of antigen or antigen mix
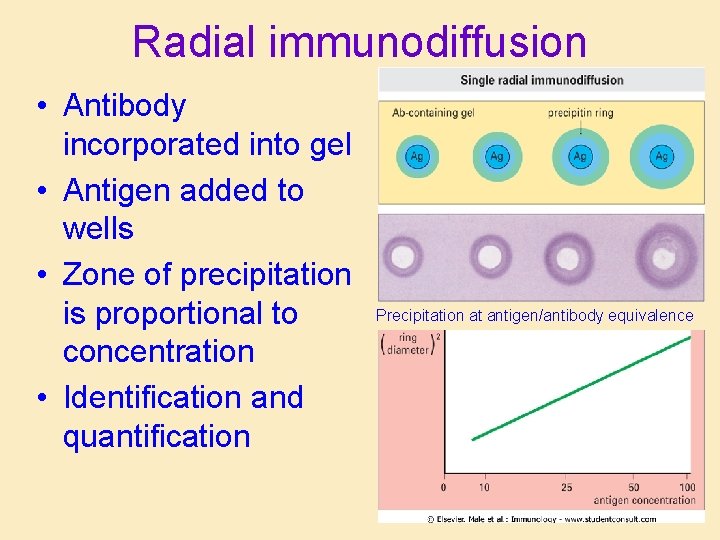
Radial immunodiffusion • Antibody incorporated into gel • Antigen added to wells • Zone of precipitation is proportional to concentration • Identification and quantification Precipitation at antigen/antibody equivalence
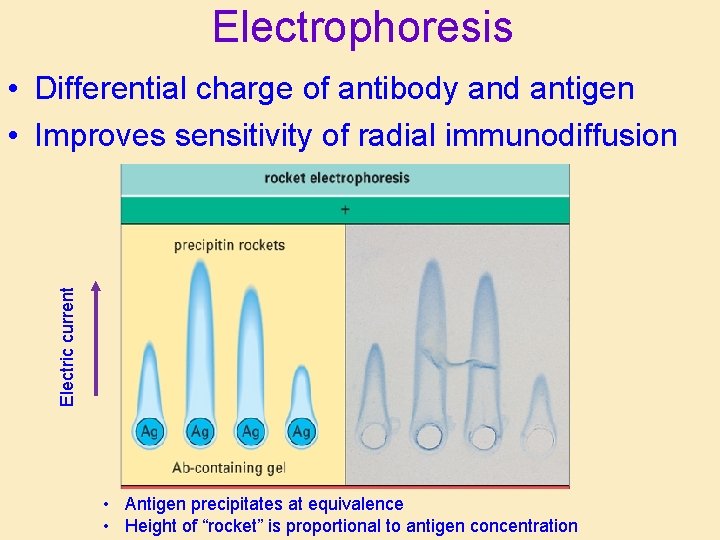
Electrophoresis Electric current • Differential charge of antibody and antigen • Improves sensitivity of radial immunodiffusion • Antigen precipitates at equivalence • Height of “rocket” is proportional to antigen concentration

Quantification by titration • Principle: – Quantitation of antibody or antigen by serial dilution – Single concentration of antigen (or antibody ) – Dilutions of antibody (or antigen) – Titer = the dilution at which precipitation occurs – Reflects but does not equal concentration • Assays: – Hemagglutination – ELISA
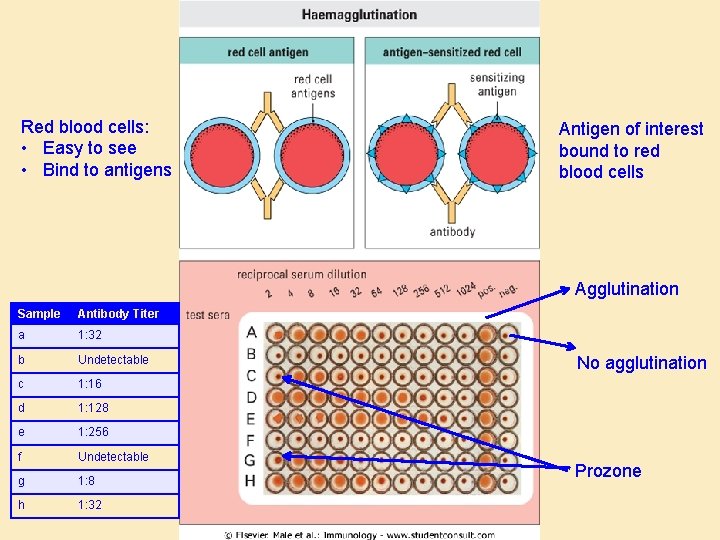
Red blood cells: • Easy to see • Bind to antigens Antigen of interest bound to red blood cells Agglutination Sample Antibody Titer a 1: 32 b Undetectable c 1: 16 d 1: 128 e 1: 256 f Undetectable g 1: 8 h 1: 32 No agglutination Prozone

Colorimetric tests: Enzyme-Linked Immunosorbant Assay • ELISA • Principles: – Specific interaction between antibody and antigen – Solid-phase – Sensitive detection • Description: – – – Antigen bound to a surface Enzyme-bound antibody Colorimetric reaction Semi-quantitative Generate titer
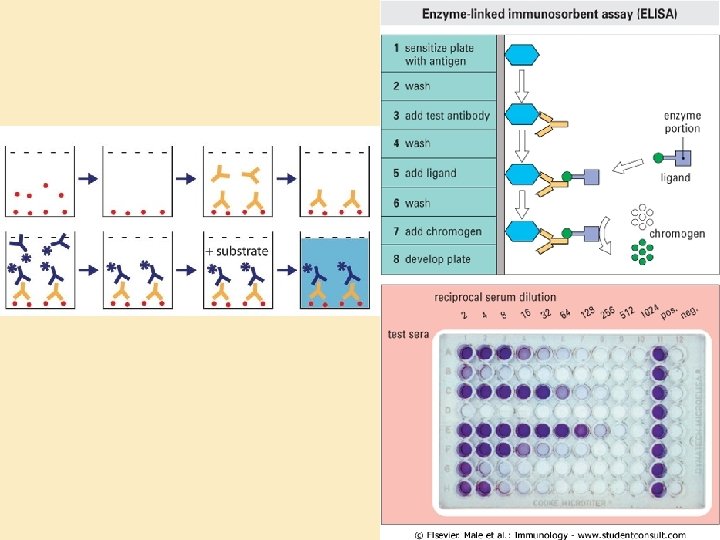

Variations on ELISA • • Indirect ELISA Capture (Sandwich) ELISA ELIspot Competitive ELISA Radioimmunoassay Immunoblot (“Western” blot) Diagnostic kits
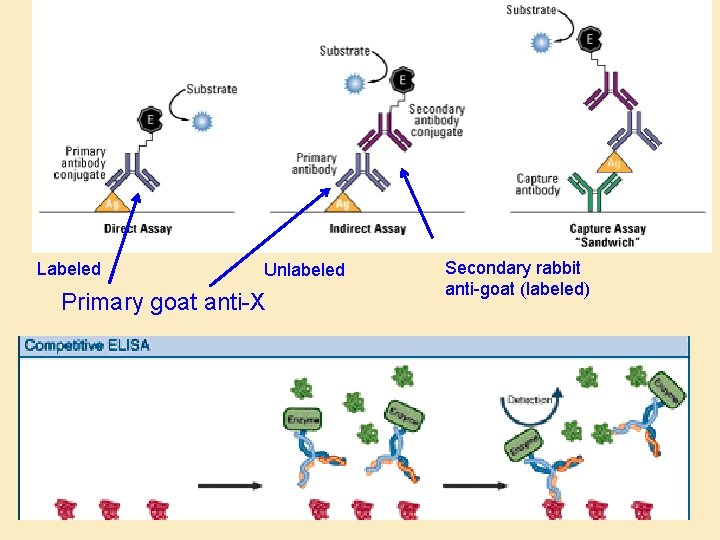
Labeled Unlabeled Primary goat anti-X Secondary rabbit anti-goat (labeled)
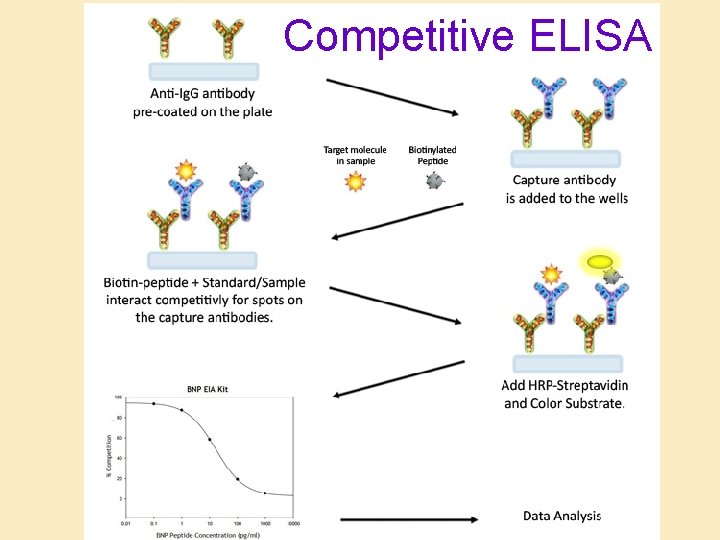
Competitive ELISA

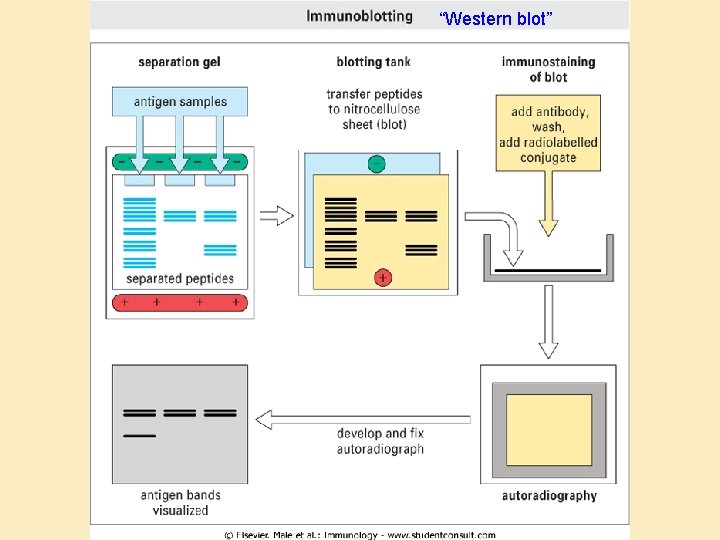
“Western blot”


Immunochemistry and Immunofluorescence • Solid-phase antigen: – In tissue section – Cytology • Detect presence and distribution of: – Surface marker – Intracellular product – Extracellular product – Metabolic product
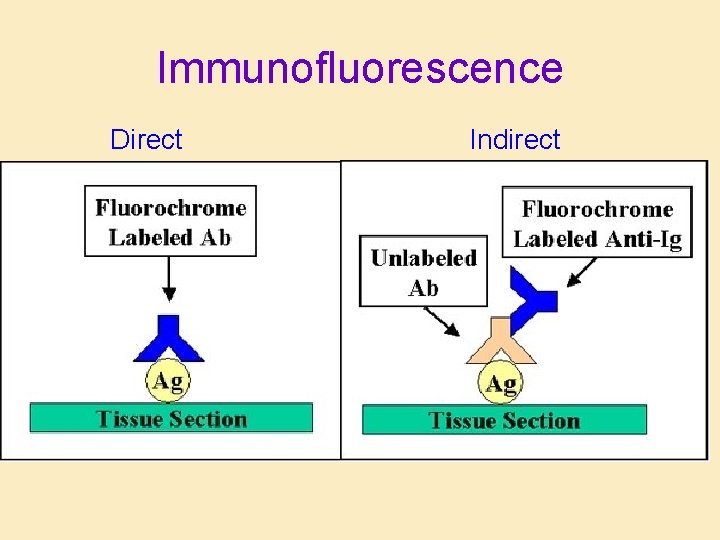
Immunofluorescence Direct Indirect
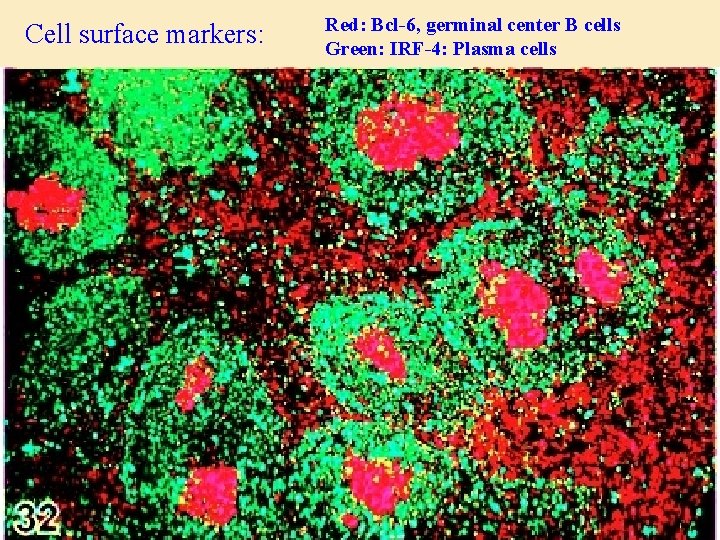
Cell surface markers: Red: Bcl-6, germinal center B cells Green: IRF-4: Plasma cells

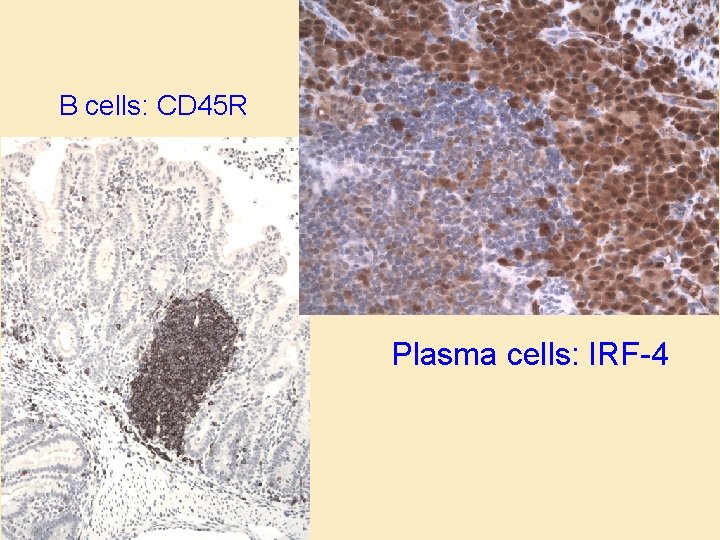
B cells: CD 45 R Plasma cells: IRF-4
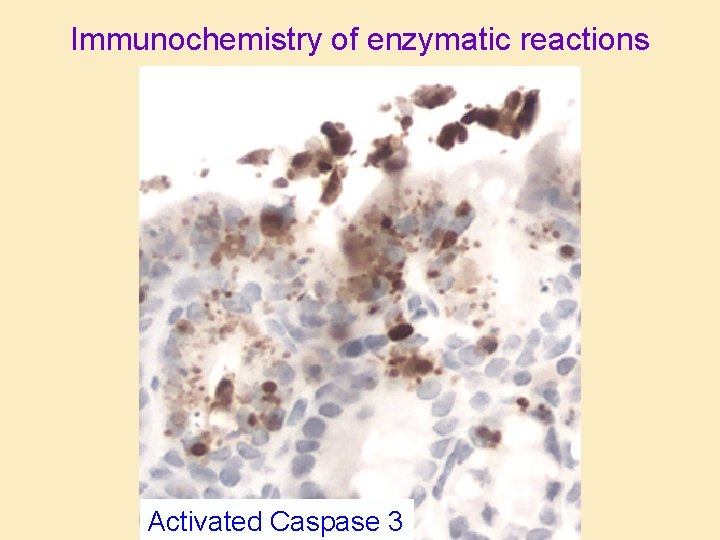
Immunochemistry of enzymatic reactions Activated Caspase 3
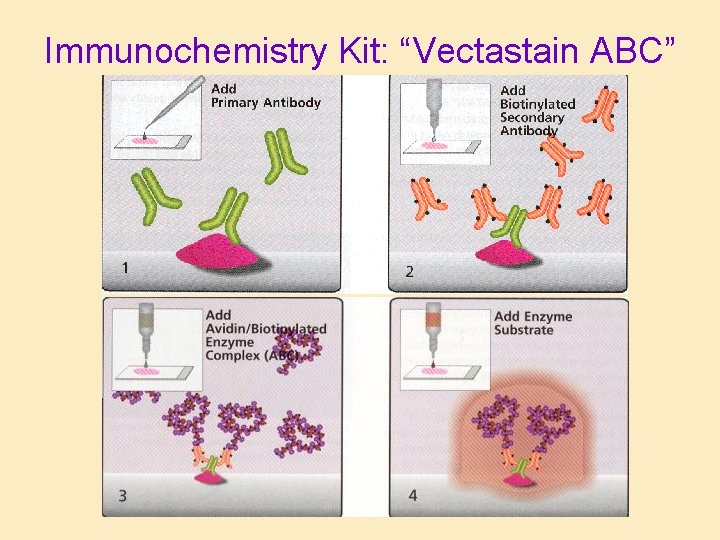
Immunochemistry Kit: “Vectastain ABC”
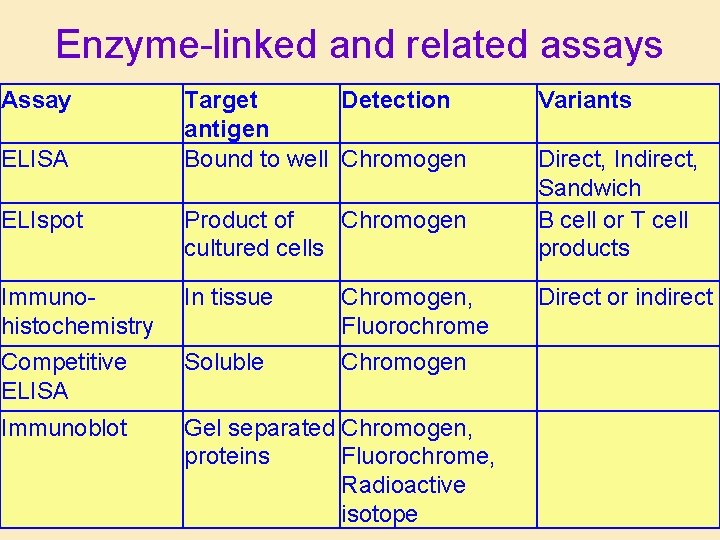
Enzyme-linked and related assays Assay ELISA Target Detection antigen Bound to well Chromogen Variants ELIspot Product of Chromogen cultured cells Direct, Indirect, Sandwich B cell or T cell products Immunohistochemistry In tissue Chromogen, Fluorochrome Direct or indirect Competitive ELISA Soluble Chromogen Immunoblot Gel separated Chromogen, proteins Fluorochrome, Radioactive isotope

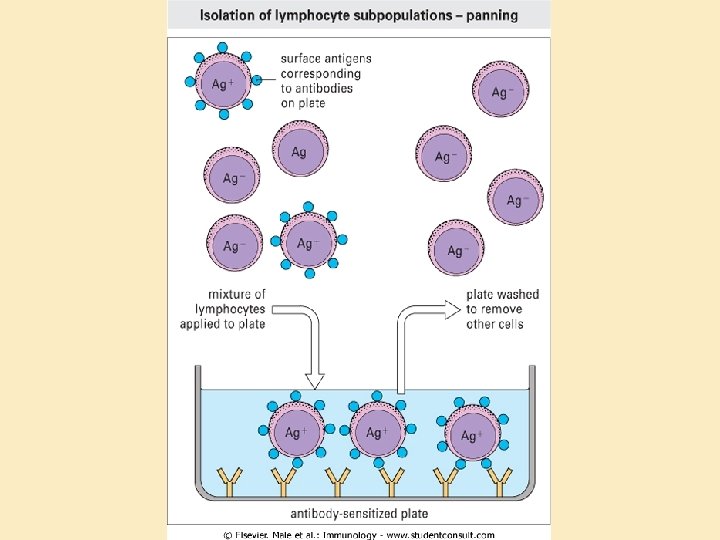
Antibodies as traps • Fluorescence-activated cell sorting (FACS): – Quantitation – Isolation • Plate-isolation of cell populations (“panning”) • Magnetic bead isolation of cells and macromolecules
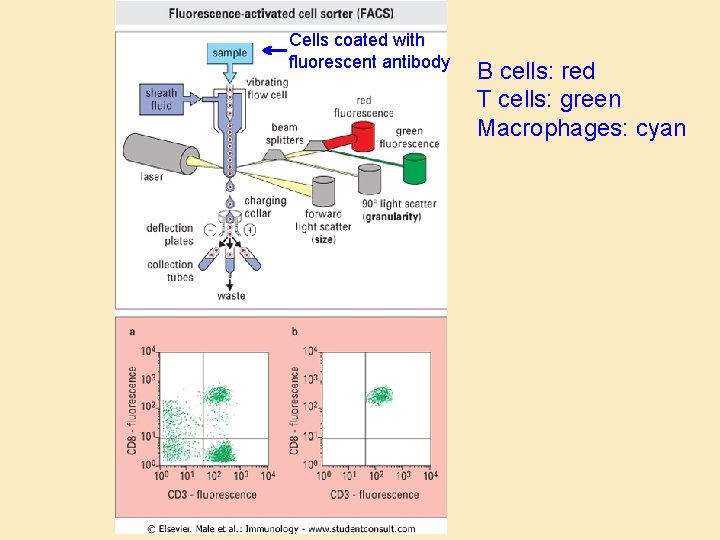
Cells coated with fluorescent antibody B cells: red T cells: green Macrophages: cyan


Lymphocyte assays • Lymphocytes as tools: – Easy to isolate – Survive well in culture • Assays: – ELIspot: B and T helper cell function – Lymphocyte blastogenesis (stimulation) test: T helper cells – Chromium release assay: Cytotoxicity
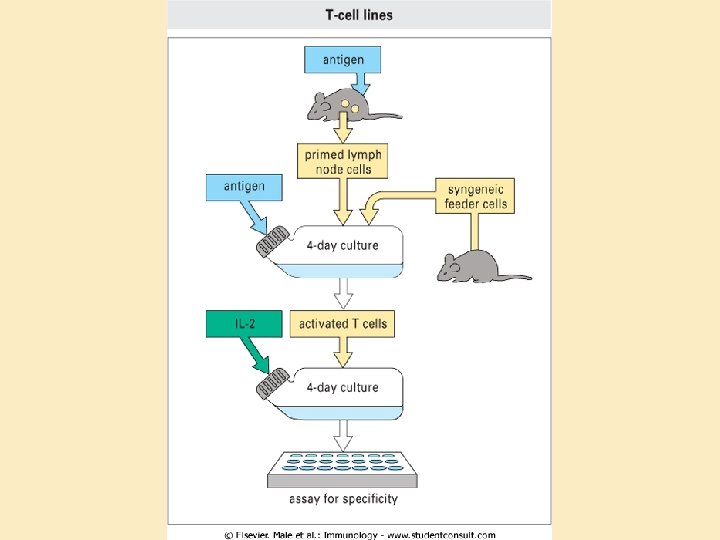
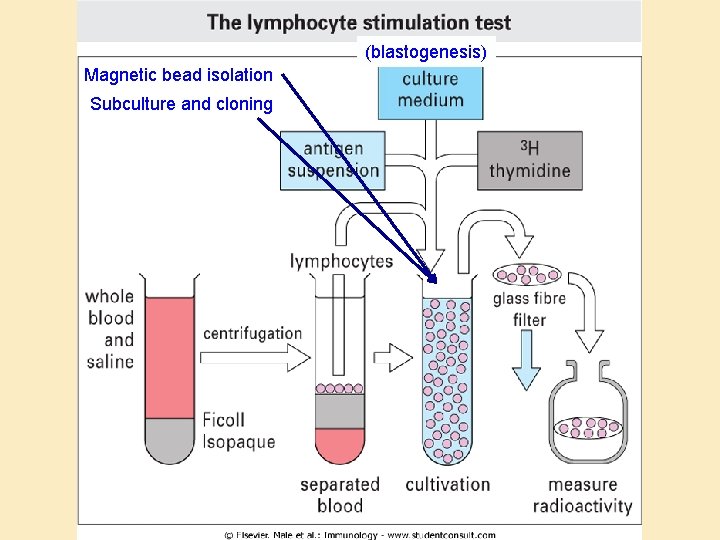
(blastogenesis) Magnetic bead isolation Subculture and cloning


That’s all, folks!!
- Slides: 44