Practical clinical chemistry Lab 1 QUALITY ASSURANCE Quality

Practical clinical chemistry Lab 1

QUALITY ASSURANCE Quality Assurance: The coordinate process of providing the best possible service to the patient and physician. QA is a complete system which contain quality control. (Right result at right time on right specimen from right patient)

WHAT IS QUALITY CONTROL? • Laboratory quality control is designed to detect, reduce, and correct deficiencies in a laboratory's internal analytical process prior to the release of patient results, in order to improve the quality of the results reported by the laboratory. • Quality control is a measure of precision, or how well the measurement system reproduces the same result over time and under varying operating conditions. • Laboratory quality control material is usually run at the beginning of each shift, after an instrument is serviced, when reagent lots are changed, after calibration, and whenever patient results seem inappropriate.

QC TYPES: Qc can be achieved through: 1 - Internal quality control Is a Procedure for utilizing the results of only one laboratory for quality control purpose 2 - External quality assessment Is a procedure for utilizing, for quality control purposes, the results of several laboratories which analyze the same specimens.

WHEN ERRORS OCCUR? üErrors occur when there is a loss of accuracy and precision. üA primary goal of quality assurance is to reduce and detect errors or to obtain the best possible accuracy and precision.

SOURCE OF ERROR: v. All analytical results are subject to errors arising from a variety of causes but it should kept to a minimum Errors can occur at various stages in the process: 1 - Pre-analytical : occur outside the LAB 2 - Analytical: occur within the lab 3 - Post-analytical: the correct result is generated but is incorrectly recorded in the patient's record

True analytical errors are those which occur during the actual performance of the analysis which can be classified into : 1 - Systematic errors. 2 - Random Errors.

o. Systematic errors: On repeated analysis of the sample we may also find values at wrong level(too high or too low) which can be due to the analytical methods, technical performance, reagents, measuring equipment and the technicians. There are two varieties of systematic errors. Ø Constant error Error In a test system in which the amount of error remains constant throughout the range of the test process. Ø Proportional error Error In a test system in which the amount of error increases the concentration of the substance being measured.


o. Random errors: Errors or mistakes the occur without prediction or regularity. Manifest themselves by variations in the results of repeated analysis of the sample ( either positive or negative errors are usually distributed) üThese variations may caused by several variable factors which include variations in apparatus, temperature, weighing, etc. , ü random error cannot be avoided üTheses random errors can be reduced by more precise methods and more accurate equipment

METHOD VALIDATION: q the ideal analytical method is accurate, precise, sensitive and specific. §Accuracy: The measure of correctness or truth of laboratory test. § precision: Agreement between replicate measurements (reproducibility)


§Sensitivity ¤the ability of a diagnostic test to detect very small amounts of the analyte. ¤Ability to correctly identify individual with disease ¤Ability to exclude false negative

§Specificity ¤detect the desired analyte without interferences. ¤Ability to correctly identify individual without disease ¤Ability to exclude false positive


§Linearity ü Is the range over which the analytical system exhibit a linear ü The highest conc. Can be detected ü The measured value from a series of samples solution is linearly proportional to the actual concentration or content of the analyte in the sample solution.

CONTROL, STANDARD, AND CALIBRATOR üControl A solution that contains the same constituents as those being analyzed in the patient sample ü Standard Substance of constant composition and of sufficient purity to be used for comparison purposes. ü Calibrator Similar to standard but it is not pure “ contain more than one analyte’.

INTERPRETATION OF QUALITY CONTROL CHARTS o. A variety of statistical control techniques have been used in clinical laboratories. o. The levey- jennings control chart has been the most widely used technique. levey- jennings control chart The control results are plotted on the y-axis. This chart shows the expected mean value by the solid line in the center and indicates the control limits or range of acceptable values by the dashed lines. The usual way of interpreting this control chart is to consider the run to be in control when the control values fall within the control limits, and to be out of control when result exceeds the control limits.


Statistical calculation: Ø mean The arithmetic mean is the average. ØStandard deviation Is the distribution range around the mean value. ØPercentage coefficient of variation (%cv) It is the standard deviation expressed in percentage

WESTGARD QC RULES ¤There are six basic rules in the Westgard scheme. ¤These rules are used individually or in combination to evaluate the quality of analytical runs. ¤Most of the quality control rules can be expressed as NL where N represents the number of control observations to be evaluated and L represents the statistical limit for evaluating the control observations. ¤ ¤Thus 13 s represents a control rule which is manifested when one control observation exceeds the ± 3 s control limits
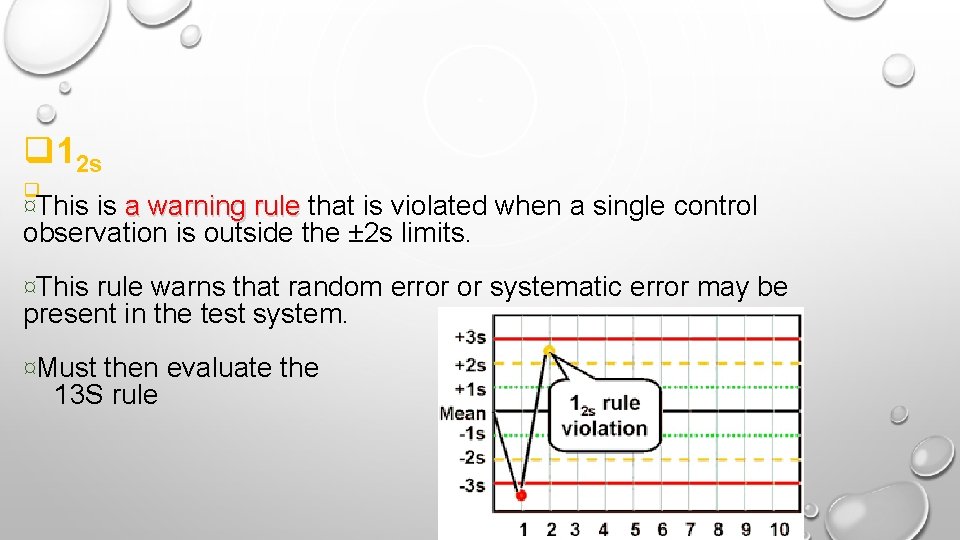
q 12 s q ¤This is a warning rule that is violated when a single control a warning rule observation is outside the ± 2 s limits. ¤This rule warns that random error or systematic error may be present in the test system. ¤Must then evaluate the 13 S rule
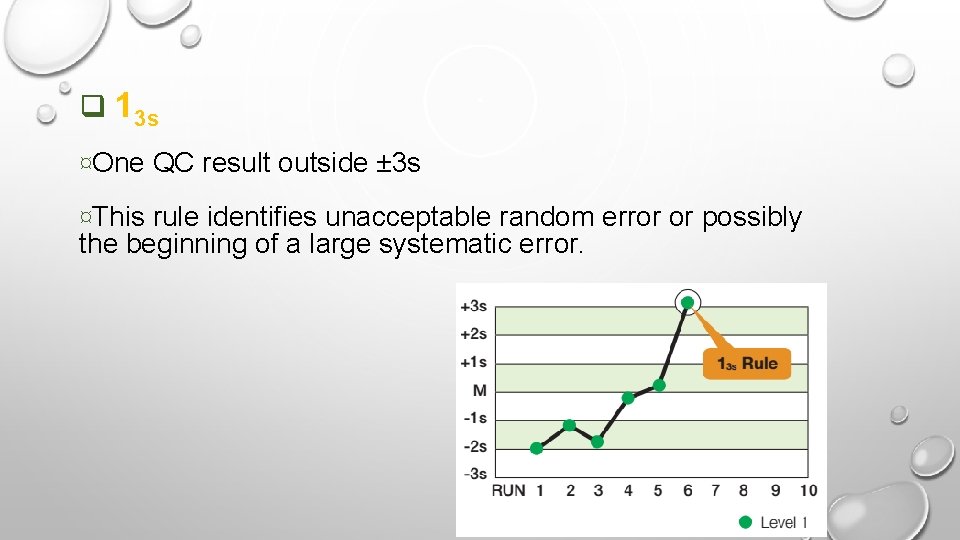
q 13 s ¤One QC result outside ± 3 s ¤This rule identifies unacceptable random error or possibly the beginning of a large systematic error.

q 22 s ¤This rule identifies systematic error only. ¤The criteria for this rule are: • Two consecutive QC results Greater than 2 s on the same side of the mean Or Both controls in the same run exceed ± 2 SD ¤Patient results cannot be reported(rejection rule). ¤Requires corrective action


q. R 4 s ¤ This rule identifies random error only ¤ One control exceeds the mean by – 2 sd, and the other control exceeds the mean by +2 sd ¤ The difference between the two results will therefore exceed 4 sd. ¤ This rule is only to be used within a run not across runs.
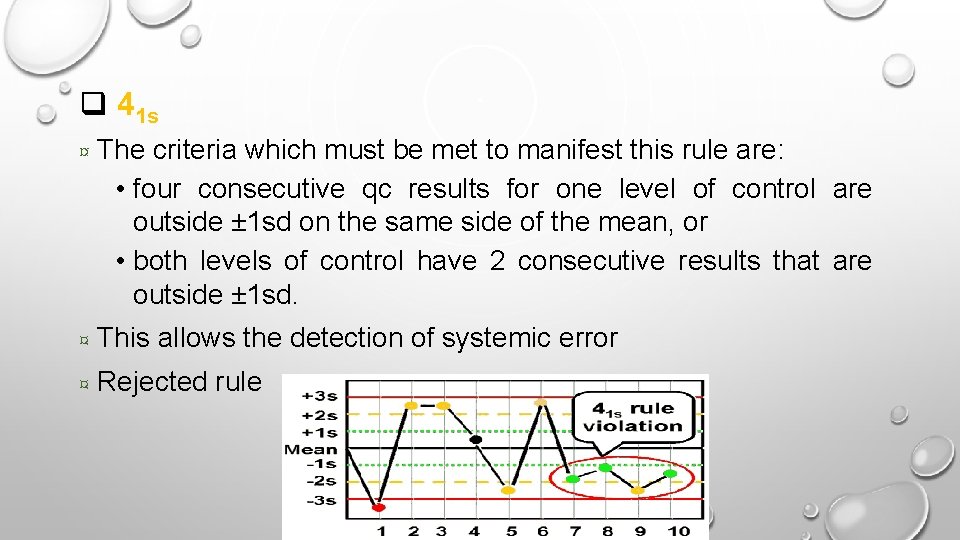
q 41 s ¤ The criteria which must be met to manifest this rule are: • four consecutive qc results for one level of control are outside ± 1 sd on the same side of the mean, or • both levels of control have 2 consecutive results that are outside ± 1 sd. ¤ This allows the detection of systemic error ¤ Rejected rule


q 10 x ¤ These rules are violated when there are: ¤ Ten consecutive qc results for one level of control are on one side of the mean, or ¤ Both levels of control have five consecutive results that are on the same side of the mean (no requirement for SD size). ¤ This allows the detection of systemic error. ¤ Rejected rule


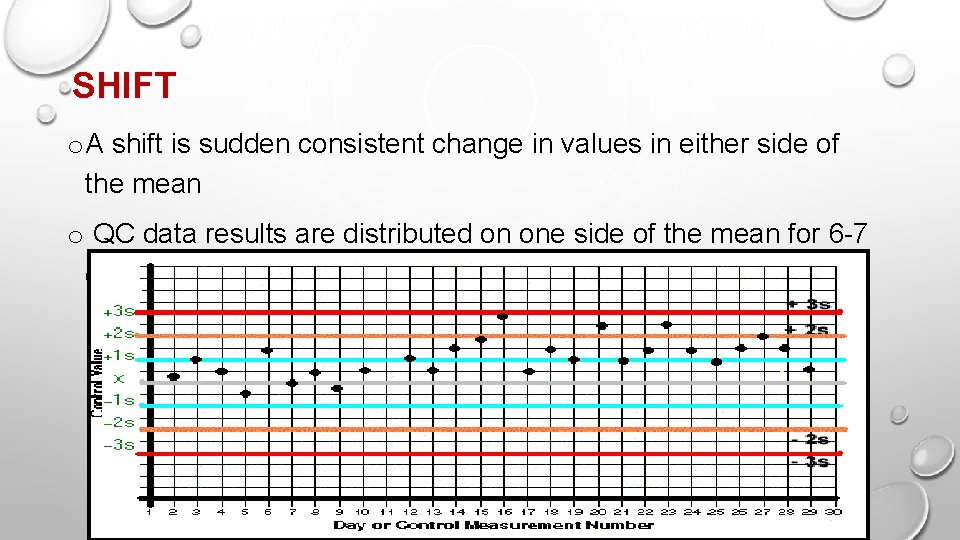
SHIFT o A shift is sudden consistent change in values in either side of the mean o QC data results are distributed on one side of the mean for 6 -7 consecutive days
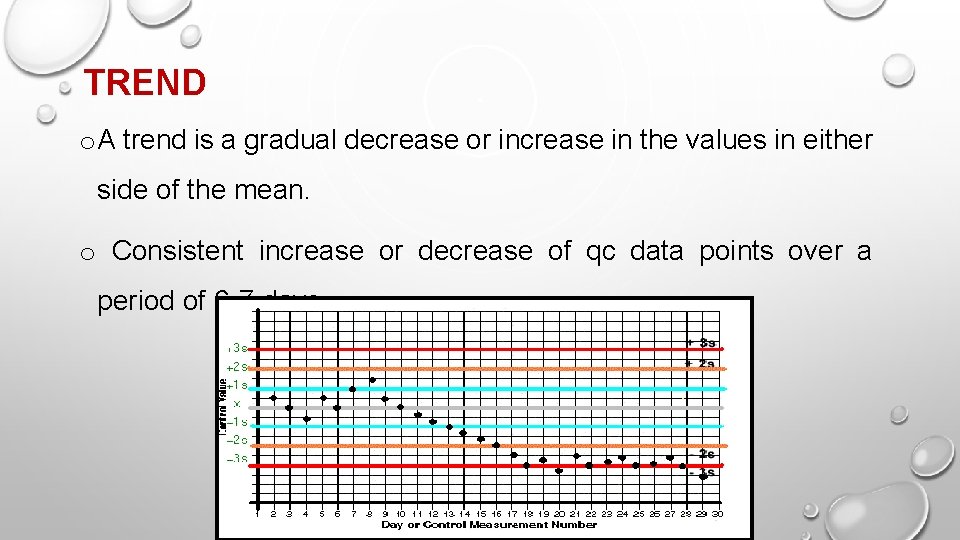
TREND o A trend is a gradual decrease or increase in the values in either side of the mean. o Consistent increase or decrease of qc data points over a period of 6 -7 days.
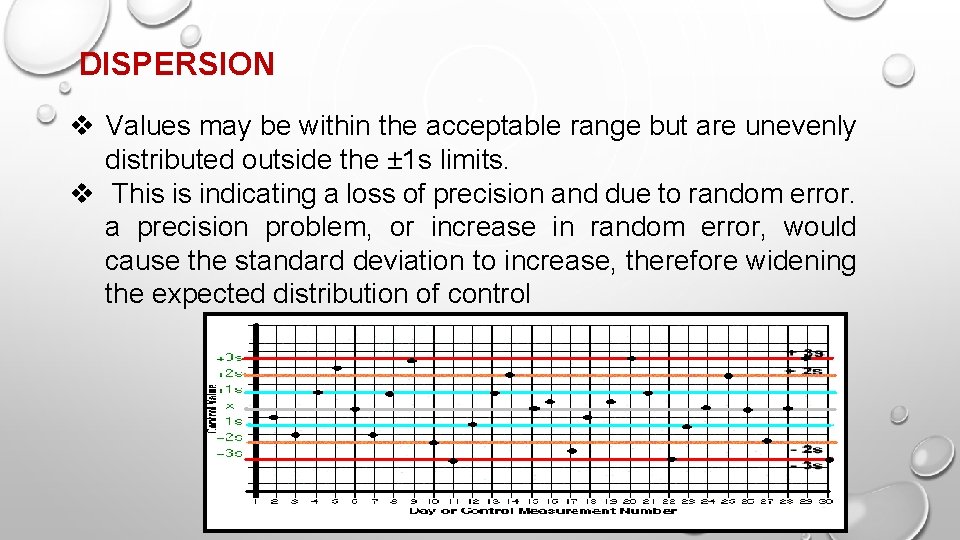
DISPERSION v Values may be within the acceptable range but are unevenly distributed outside the ± 1 s limits. v This is indicating a loss of precision and due to random error. a precision problem, or increase in random error, would cause the standard deviation to increase, therefore widening the expected distribution of control

- Slides: 35