Pr ECOG 0102 A Randomized DoubleBlind Phase II

Pr. ECOG 0102: A Randomized, Double-Blind Phase II Trial of Fulvestrant plus Everolimus or Placebo in Post-Menopausal Women with Hormone-Receptor Positive, HER 2 -Negative Metastatic Breast Cancer Resistant to Aromatase Inhibitor Therapy Noah S Kornblum, MD 1, Judith Manola, MS 2, Paula Klein, MD 3, Bhuvaneswari Ramaswamy, MD 4, Adam Brufsky, MD Ph. D 5, Phillip J Stella, MD 6, Brian Burnette, MD 7, Melinda Telli, MD 8, Della F Makower, MD 1, Joseph Leach, MD 9, Cristina I Truica, MD 10, Antonio C Wolff, MD 11, Gamini S Soori, MD 12, Barbara Haley, MD 13, Arun Nagarajan, MD 14, Timothy R Wassenaar, MD 15, Lori Goldstein, MD 16, Kathy D Miller, MD 17, and Joseph A Sparano, MD 1 Institutions: 1 Montefiore-Einstein Center for Cancer Care, Bronx, New York, United States, 10461; 2 Dana-Farber Cancer Institute, Boston, MA, United States, 02284 -9168; 3 Mount Sinai Beth Israel Comprehensive Cancer Center, New York, United States, 10011; 4 Ohio State University Comprehensive Cancer Center, Columbus, OH, United States, 43212; 5 University of Pittsburgh, PA, United States, 15213; 6 Saint Joseph Mercy (Michigan Cancer Consortium), Ann Arbor, MI, United States, 48106; 7 Saint Vincent Hospital, Green Bay, WI, United States, 54301; 8 Stanford University Medical Center, Stanford, CA, United States, 95304; 9 Metro-Minnesota Community Oncology Research Consortium, Saint Louis Park, MN, United States, 55416; 10 Penn State Hershey Cancer Institute, Hershey, PA, United States, 17033; 11 Johns Hopkins Sidney Kimmel Comprehensive Cancer Center, Baltimore, MD, United States, 21287; 12 Missouri Valley Cancer Consortium, Omaha, NE, United States, 68106; 13 UT Southwestern Medical Center, Dallas, TX, United States, 75390; 14 CAMC Health System, Charleston, WV, United States, 25304; 15 Pro Health Care, Waukesha, WI, United States, 53188, 16 Fox Chase Cancer Center, Philidelphia, PA, United States, 19111 and 17 Indiana University School of Medicine, Indianapolis, IN, United States, 46202 -5689. This presentation is the intellectual property of the author. Contact nkornblu@montefiore. org for permission to reprint and/or distribute.

San Antonio Breast Cancer Symposium, December 6 -10, 2016 Background: Resistance to Aromatase Inhibitor (AI) Therapy is Inevitable § Strategies to address resistance are emerging § Targeting PI 3 K-AKT-m. TOR pathway 1 § Addition of the m. TOR inhibitor Everolimus to the steroidal AI exemestane improved median PFS (3. 2 vs. 7. 8 mo. , p<0. 0001) in the phase III BOLERO-2 trial § Use of a selective estrogen receptor down regulator (SERD) § More complete blockade of ER signaling than other anti-estrogens § Hypothesis: The combination of everoliumus/fulvestrant would be more effective than fulvestrant alone in AI resistant disease 1 Ma, C, et al. Nature Reviews Cancer 15, 261– 275 (2015) ; 2 Baselga, J. et al, New Engl. J. Med. 366, 520– 529 (2012) ; 3 Yardley, D. et al, Adv Ther (2013) 30: 870– 884; 4 Piccart, M, et al. Ann Oncol 25, 12, 2357 -2362 (2014) ; 5 Bachelot, T. et al. J Clin Oncol. 30, 2718– 2724 (2012); 6 Chia, S. , et al. J Clin Oncol 26: 1664 -70 (2009). ; 7 Di Leo, A et al. J Clin Oncol. 28, 4594 -4600 (2010) This presentation is the intellectual property of the author. Contact nkornblu@montefiore. org for permission to reprint and/or distribute.

San Antonio Breast Cancer Symposium, December 6 -10, 2016 Methods: Key Eligibility Criteria § § Post-menopausal women HR-positive, HER 2 -negative (ASCO-CAP) Inoperable locally advanced or metastatic breast cancer AI resistant disease: § § § • Relapse while receiving adjuvant AI therapy • Progression after one or more AIs for metastatic disease ECOG PS 0 -1 Normal organ function < 1 prior chemotherapy regimen for metastasis Measurable and/or non-measurable disease (RECIST 1. 1) 2 doses of fulvestrant permitted within 28 d prior to randomization This presentation is the intellectual property of the author. Contact nkornblu@montefiore. org for permission to reprint and/or distribute.
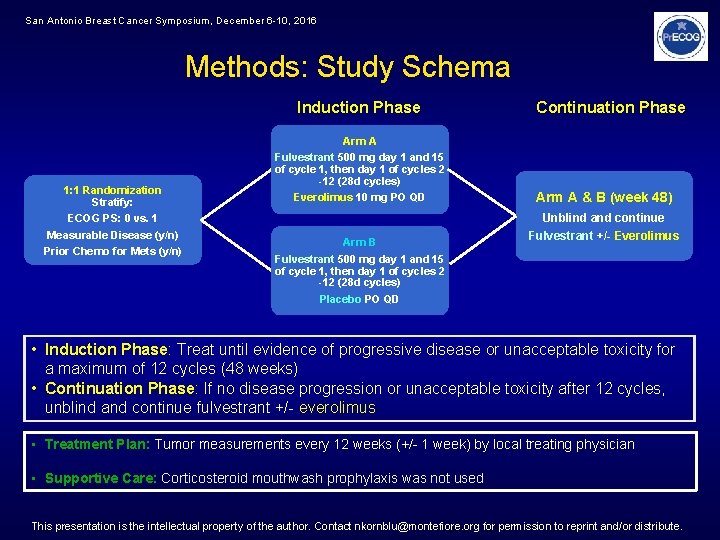
San Antonio Breast Cancer Symposium, December 6 -10, 2016 Methods: Study Schema Induction Phase 1: 1 Randomization Stratify: ECOG PS: 0 vs. 1 Measurable Disease (y/n) Prior Chemo for Mets (y/n) Arm A Fulvestrant 500 mg day 1 and 15 of cycle 1, then day 1 of cycles 2 -12 (28 d cycles) Everolimus 10 mg PO QD Arm B Fulvestrant 500 mg day 1 and 15 of cycle 1, then day 1 of cycles 2 -12 (28 d cycles) Placebo PO QD Continuation Phase Arm A & B (week 48) Unblind and continue Fulvestrant +/- Everolimus • Induction Phase: Treat until evidence of progressive disease or unacceptable toxicity for a maximum of 12 cycles (48 weeks) • Continuation Phase: If no disease progression or unacceptable toxicity after 12 cycles, unblind and continue fulvestrant +/- everolimus • Treatment Plan: Tumor measurements every 12 weeks (+/- 1 week) by local treating physician • Supportive Care: Corticosteroid mouthwash prophylaxis was not used This presentation is the intellectual property of the author. Contact nkornblu@montefiore. org for permission to reprint and/or distribute.

San Antonio Breast Cancer Symposium, December 6 -10, 2016 Methods: Statistical Design § Primary endpoint: progression-free survival (by investigator assessment) § 130 total patients (120 eligible assuming 10% ineligible) § 90% power § 1 -sided Type I error 10% § 70% improvement in median PFS - 5. 4 mo. (CONFIRM)1 to 9. 2 mo. § Stratified log-rank test § ITT analysis § Full information = 98 events (death or progression) 1 Di Leo, A et al. J Clin Oncol. 28, 4594 -4600 (2010) This presentation is the intellectual property of the author. Contact nkornblu@montefiore. org for permission to reprint and/or distribute.

San Antonio Breast Cancer Symposium, December 6 -10, 2016 Progression Free Survival (by investigator assessment – primary study endpoint) This presentation is the intellectual property of the author. Contact nkornblu@montefiore. org for permission to reprint and/or distribute.

San Antonio Breast Cancer Symposium, December 6 -10, 2016 Conclusions § Addition of everolimus to fulvestrant improved PFS § median PFS 5. 1 vs. 10. 4 months § HR 0. 60, p=0. 02 § Associated with more toxicity, including § Grade 3 adverse events: 48% (F/E) vs. 14% (F/P) § Most common grade 3 A. E. s occurring in > 5% included stomatitis (9%), pneumonitis (6%), fatigue (5%), & hyperglycemia (6%) § Saftey profile consistent with everolimus in BOLERO-21 § Prophylactic corticosteroid mouthwash was not used, which has been shown to reduce risk of grade 1 -2 stomatitis from about 65% to 20%2 § Provides additional evidence that adding everolimus to anti-estrogen therapy in AI resistant disease improves clinical outcomes 1 Baselga, J. et al, New Engl. J. Med. 366, 520– 529 (2012) 2 Rugo, H, et al. J Clin Oncol 34, 2016 (suppl; abstr 525) This presentation is the intellectual property of the author. Contact nkornblu@montefiore. org for permission to reprint and/or distribute.
- Slides: 7