Powerpoint Jeopardy Atom stuff Chemical stuff Bonding stuff
Powerpoint Jeopardy Atom stuff Chemical stuff Bonding stuff Stuff stuff Mr mac stuff 10 10 10 20 20 20 30 30 30 40 40 40 50 50 50 1

Subdivision of principle energy levels 2

Statement that only electrons of opposite spin can occupy the same orbital 3

The electrons in the outer most energy level 4

The amount of energy needed to remove an electron from an atom or ion 5

The arrangement of electrons about an atom 6

The inner electrons of an atom that are not in the outer most energy level 7

A force that hold any two atoms together 8

A pure substance that can not be broken down into simpler substances 9

A substance that contains two or more substances in variable concentration. 10

Pairs of electrons Two or more elements combine in definite proportions. 11

A mixture that is the same throughout 12

A positive ion. 13
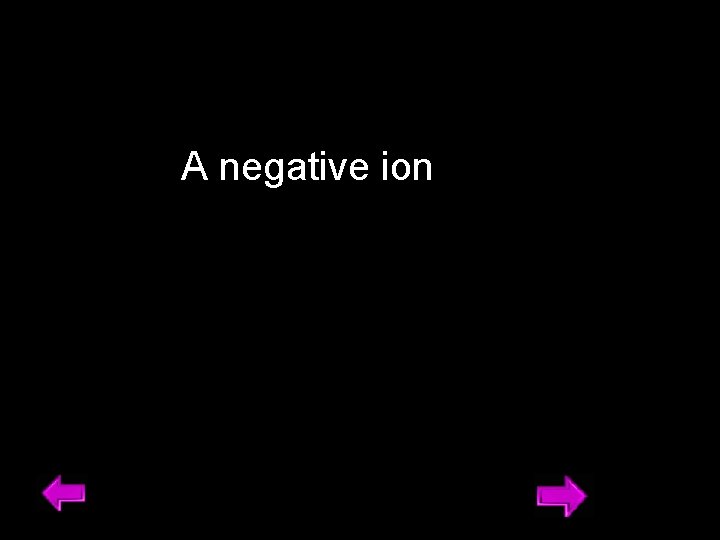
A negative ion 14

A compound created by the transfer of electrons 15

The number of valence electrons in Na 16

The number of valence electrons in sulfur 17

The number of valence electrons in Ne 18

A unit of heat energy based on the amount of heat needed to raise the temperature of one gram of water one degree 19

A number equal the number of Carbon atoms in 12. 01 grams of carbon 20

The mass in grams of one mole of a compound 21

An instrument used to measure heat in a chemical reaction 22

A “particle” of electromagnetic radiation 23

The discrete energy levels within an atom 24

The number of waves that pass a given point every second 25

A representation of the space occupied by electrons in an atom. 26

sublevels 27

Pauli exclusion principle 28

valence 29

Ionization energy 30

Electron configuration 31

Core electrons 32

Bond 33

element 34

mixture 35

compound 36

homogeneous 37

cation 38

anion 39

ionic compound 40

one 41

6 42

eight 43

calorie 44

mole 45

Molar mass 46

calorimeter 47

photon 48

principle energy level 49

frequency Molecular formula 50

orbital Molecular formula 51
- Slides: 51