Powerpoint Jeopardy Atom stuff Chemical stuff Bonding stuff
Powerpoint Jeopardy Atom stuff Chemical stuff Bonding stuff Stuff stuff Mr mac stuff 10 10 10 20 20 20 30 30 30 40 40 40 50 50 50 1

A compound made of two elements 2

An ion composed of two of more elements 3

An compound that reacts with a base to produce water and a salt 4

A compound compose of one metal and one nonmetal 5

An polyatomic ion containing oxygen 6

The substance that are started with in a chemical reaction 7

An acid that completely disassociates into H+ ions. . 8

A substance that completely disassociates into –OH ions. 9

A reaction in which electrons are transferred between the components. 10

A substance that gives up -OH ions. 11

General name given to compounds consisting of a metal and nonmetal 12

A reaction in which aqueous solutions produce a solid product. 13

Reaction where a pair of cations and anions exchange partners 14

A rapid reaction with oxygen. 15

A reaction where a compound breaks down into components 16

A reaction where an H+ ion and a -OH ion combine to produce water 17

A reaction where component are combined into one product 18

A small unit of mass equal to 1. 66 x 10 -24 grams 19

A number equal the number of Carbon atoms in 12. 01 grams of carbon 20

The mass in grams of one mole of a compound 21

The simplest whole number ratio of atoms in a compound 22

6. 022 X 1024 23

The percent by mass of a compound or a mixture or of a given element in a compound 24

The exact formula of a molecule given the type of atoms and number of atoms 25

An atom with a positive or negative charge. 26

Binary compound 27

Polyatomic ion 28

acid 29

binary ionic 30

oxyanion 31

reactant 32

strong acid 33

strong base 34

oxidation reduction reaction 35

base 36
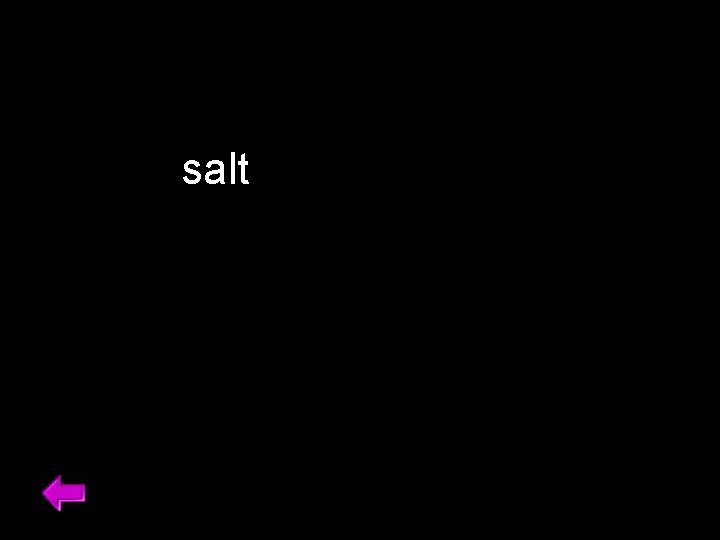
salt 37

Precipitation 38

Double replacement reaction 39
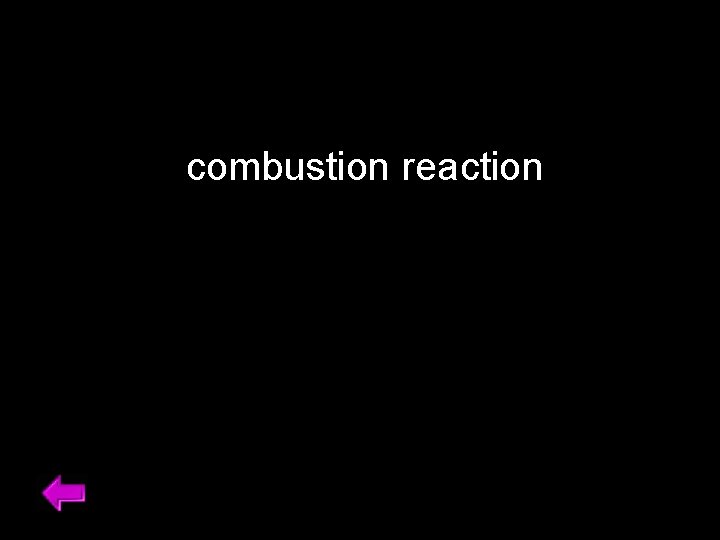
combustion reaction 40

decomposition 41

Acid base reaction 42

Synthesis reaction 43

Atomic mass unit 44

mole 45

Molar mass 46

empirical formula 47

Avogradro number 48
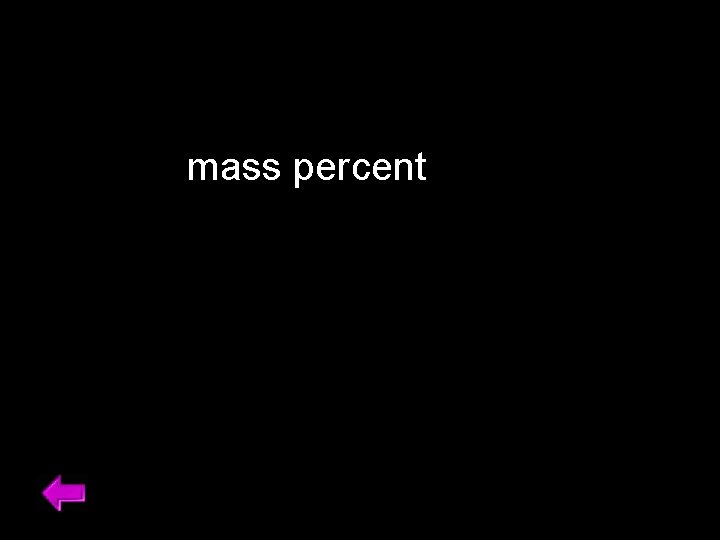
mass percent 49

Molecular formula 50
ion Molecular formula 51
- Slides: 51