Potential BoronGadolinium Neutron Capture Therapy Program at Fermilab

Potential Boron/Gadolinium Neutron Capture Therapy Program at Fermilab James S. Welsh, MS, MD

Problems and solutions No clinical BNCT in the USA presently l Reactor-based treatments pose some challenges l Fermilab accelerator-based approach might be practical, effective and affordable l

Basic principles l l l BNCT is a binary treatment modality in which neither component alone is lethal or highly toxic… Interaction of a thermal neutron and 10 B creates highly biologically effective radiation in-situ Such radiation is capable of dealing with chemotherapy/radiation-resistant phenotypes irrespective of genetic details l l MGMT promoter methylation, EGFRv. III , mutant isocitrate dehydrogenase isozymes, 2 -hydroxyglutarate, PTEN, ATRX hypoxia/HIF-1 alpha, VEGFR 2, etc, etc Success is predicated on selective localization of boron in tumor cells and adequate flux of neutrons of appropriate energy to the tumor


BNCT History Concept proposed shortly after Chadwick’s discovery l 1934 – Goldhaber in Cambridge, England demonstrated that an isotope (10 B) had a high avidity for thermal neutrons l 1936 – Gordon Locher, biophysicist at Bartol Research Institute in Swarthmore, PA l l suggested that if tumor could be loaded with 10 B and then the region flooded with slow neutrons, a dose-enhancement effect could be achieved (AJR 36: 1; 1936)

The basic three neutron energies Thermal = 0 – 0. 4 e. V l Epithermal = 0. 4 e. V – 10 ke. V l Fast >10 ke. V l

Neutron interactions Neutron decay l Recoiling nuclei (scattering) l Nuclear disintegrations l Neutron capture l
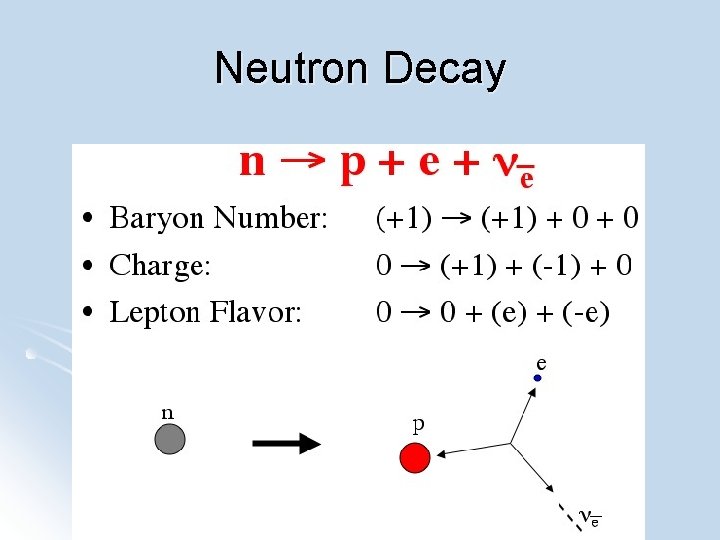
Neutron Decay

Nuclear disintegration l l l Spallation l 12 C + n 3 a l 16 O + n 4 a Fission: l 235 U(n, F) l 10 B(n, a)7 Li Small total fraction of absorbed dose but relatively high biological effect because of the high-LET of the fragments

Neutron interactions l Neutron Capture Cross sections inversely related to energy (Enrico Fermi 1935) E = hn = hc/l l= h/p KE= p 2/2 m l = 2. 87 x 10 -9/(E)0. 5 cm l l Fast neutrons: de. Broglie wavelength ~10 -12 cm l Thermal neutrons: l ~ 1. 8 x 10 -8 l Cross sections increase inversely with energy or… l “Slow, fat and fuzzy neutrons hit easier”

Neutron Capture Cross sections vary heavily with other variables besides de. Broglie wavelength… l Isotope effects l Cd has an absorption cross section of 7000 b at 0. 176 e. V l This is due mostly to 113 Cd which is only 12. 26% abundant but has a cross section of 20, 000 barns l l Other isotopes also have large cross sections for neutron capture (e. g. 10 B, 157 Gd)

Neutron Capture l Neutron capture often leads to Neutron Activation l A = Rk = (Fsact. N) * (1 – e-kt)e-kt’ l R = (Fsact. N)/k * (1 – e-kt)e-kt’ l Basis for neutron activation analysis to determine presence of trace elements

(n, g) reaction l Many nuclei undergo radiative capture and emit a prompt capture gamma e. g. 1 H (n, g) 2 H l Photon Emax of 6 -10 Me. V l in rough agreement with the neutron binding energy l e. g. 157 Gd(n, g)158 Gd releases 7. 9 Me. V l One or several gamma photons can be emitted

(n, a) reaction l l (n, a) typically occurs only with fast neutrons l Boron and Lithium are two exceptions 6 Li(n, a)3 H Li(n, a) H l A means of making tritium 10 B (~20% natural abundance) has a cross-section of 3990 b for thermal neutrons 10 B(n, a)7 Li l Neutron detection instruments - and BNCT!

General thermal neutron interactions in tissue during BNCT Hydrogen recoil (proton scattering) l Nitrogen neutron capture l Hydrogen neutron capture l Boron neutron capture l

Hydrogen recoil Collisions of nth with hydrogen (protons) l Recoiling protons dissipate energy l Little energy actually transferred this way l But 99% of flux attenuation (of thermal neutrons) is via proton scattering l

Nitrogen neutron capture l Capture of nth and emission of a 500 ke. V proton l l Products have a 10 mm range of dose deposition l l Speculation: Could some of the positive effects of BNCT be due to the increased nitrogen content of aneuploid tumor cells? ? ? N-capture constitutes 25 -50% of non-boron dose l l 14 N(n, p)14 C But little effect on flux attenuation Together with hydrogen neutron capture >99% of non-boron tissue dose

Hydrogen neutron capture l Absorption is followed by a 2. 2 Me. V prompt gamma l l 1 H(n, g)2 H Mean path of gamma photon for dose-deposition is 5 -10 cm 50%-75% of non-boron dose is due to hydrogen capture As with N capture, has little affect on attenuation of beam

Boron neutron capture l 10 B(n, a)7 Li (93%) Average of 2. 31 Me. V kinetic energy between the a (1. 47 Me. V) and 7 Li (0. 84 Me. V) 4 He range ~ 7 mm 7 Li range ~ 4 mm Initial LET is between 200 -300 ke. V/mm l l l 10 B + 1 n 7 Li + 4 He + g

Basic radiobiological principles BNCT products possess high d. E/dx (i. e. LET) l High LET reduced OER l High-LET radiation typically confers a higher RBE for tumor cell killing l More difficult to repair – little to no PLDR or SLDR l Direct action on biomolecules often cannot be mitigated (whereas indirect action might be via free radical scavengers) l
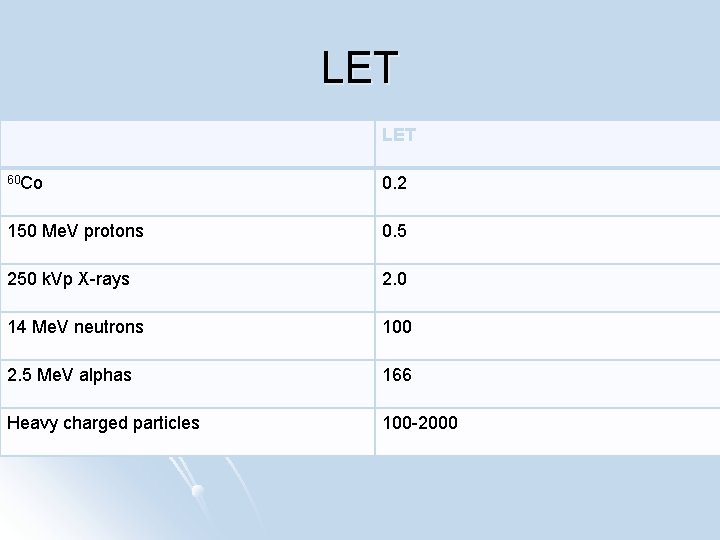
LET 60 Co 0. 2 150 Me. V protons 0. 5 250 k. Vp X-rays 2. 0 14 Me. V neutrons 100 2. 5 Me. V alphas 166 Heavy charged particles 100 -2000
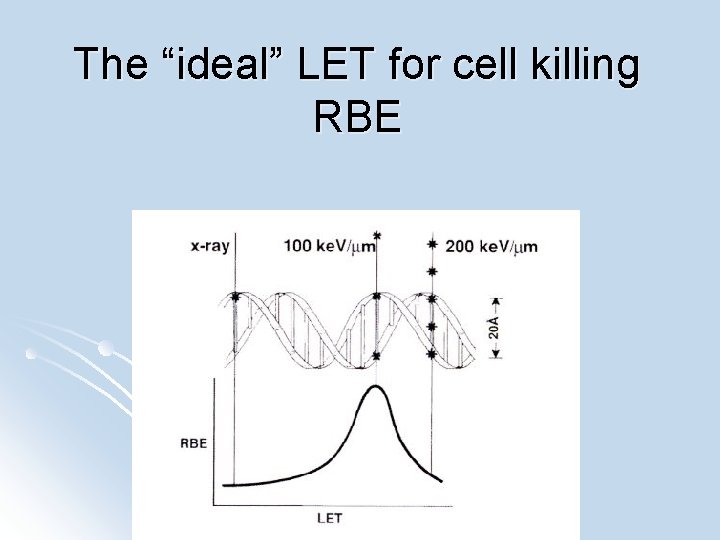
The “ideal” LET for cell killing RBE



Boron neutron capture l 10 B is 19. 9% naturally abundant Cross section for thermal neutrons (sth) = 3990 barns l Requires ~109 10 B per cell or 35 mg 10 B/g tissue l At this concentration 85% of radiation damage will be due to the BNCT reaction l

Boron neutron capture Normal tissue ideally should be kept under 5 mg/g l Tumor cellular concentrations of 20 -40 ppm have been achieved l l With tumor-normal tissue concentration ratios of 3: 1 to 10: 1

Alternatives to Boron-10 l Lithium-6 l l Helium-3 and xenon-135 l l l Good nuclear properties but difficult chemistry Higher cross sections than 10 B 135 Xe has highest measured at 2, 720, 000 barns! Nobel gases: chemically and biologically useless Additionally 135 Xe is radioactive… Uranium-235, plutonium-241, americium-242 l l l Undergo fission into very high LET products 242 Am has a cross section of 8000 b All are radioactive

Alternatives to Boron-10 l Cadmium-113 and Samarium-149 l Larger cross sections than boron-10 but result in prompt gammas rather than high -LET hadron fission products l But is that a real drawback? ? ?

Alternatives to Boron-10 l l Gadolinium-157 l Paramagnetic effects on protons l Allows visualization and localization with mm resolution l Many approved Gd-containing MRI contrast agents l MRI contrast agents such as gadopentetate dimeglumine (Magnevist®), gadolinium diethylene triamine pentaacetic acid (Gd-DTPA) are routinely used Makes the idea attractive l one could determine ahead of time if the Gd is accumulating in the tumor or not



Gadolinium as a neutron capture agent? l l l Natural abundance of 157 Gd = 15. 6% Thermal neutron cross-section 62 x 10 B sth = 255, 000 barns l Large cross section means self-shielding of deep seated tumors l Might require >200 ppm l But less than 1000 ppm overall to reduce toxicity

Gadolinium-157 l 157 Gd(n, g)158 Gd releases 7. 94 Me. V total kinetic energy – over twice that of 10 B(n, a)7 Li 157 Gd + nth → [158 Gd] → 158 Gd + g + 7. 94 Me. V l How is that energy distributed? ?

Gadolinium-157 l Prompt gammas converted into characteristic x- rays as well as: l Conversion electrons l Auger electrons l Coster-Kronig electrons l Showers with submicron ranges in tissue l Auger and Coster-Kronig e-’s have high LET! l But submicron range suggests Gd. NCT might require intranuclear localization of Gd l Highly toxic to work with

Gadolinium as a neutron capture agent? Kassis 2005 Auger and Coster-Kronig electrons are short-range and high-LET/high RBE GADOLINIUM MUST BE IN NUCLEUS FOR REAL BIOLOGICAL EFFECT – Perhaps immediately adjacent to the DNA molecules

Thermal neutrons Very rapid falloff; HVL = 2 cm l Negligible skin sparing l Useful BNCT treatment depth <4 cm depending on [10 B] l Therefore tumor must be superficial AND… l Skin and skull reflected during BNCT treatment(!) l Intraoperative radiation therapy l

Thermal neutrons Wide fields can improve PDD l BNCT treatment depth ~6 cm at 15 -22 cm diameter fields l But still requires an intraoperative approach l

Heavy water Ordinary hydrogen captures more neutrons than deuterium does l Many neutrons lost by H-capture before being able to induce 10 B(n, a)7 Li l Replacing body water with D 2 O can enhance thermal neutron penetration by up to 20 -30% l Heavy water concentrations up to 30% are not toxic l

Epithermal neutrons l l l l Better penetration Lose energy via scattering (moderation) to thermal energies HVL 4 -5 cm beyond build-up peak 10 B does NOT capture epithermal neutrons Thus there is superficial tissue dose sparing An “injection” of thermal neutrons at depth BOTTOM LINE: No surgical reflection needed! Allows fractionation

Caveats l l l With thermal neutrons recoil protons contribute negligibly to total dose With epithermal (and fast) neutrons, recoil protons contribute significantly Moderation process yields fast recoil protons l l l and if energy if high enough, carbon and oxygen nuclear reactions Thus high-energy neutrons may be suboptimal if they yield unacceptably high background dose 0. 5 – 30 ke. V neutrons may be best for nonoperative BNCT

Generally desirable features Epithermal neutron beam l Flux >2 x 109 cm-2 s-1 l Treatment < 1 hr, preferably <30 minutes l <2. 8 x 10 -12 Gy cm-2 fast neutrons plus gamma photons contamination l

Producing appropriate neutrons Nuclear reactors l D-T generators l Californium-252 l Cyclotrons and linacs – Stripping reaction l Cyclotrons and linacs – Knock-out process l

Research reactors l l 100 k. Wth – 40 MWth 235 U fission fast neutrons l moderated by tens of cm of D 2 O then l filtered through cms of Pb or Bi to remove gamma rays Beam “hardness” (i. e. thermal: non-thermal neutrons) quantified by Au: cd ratio l Higher Au: Cd means more thermal enhancement l

MITR-II Dedicated medical treatment room l Vertically oriented neutron beam (coming from above) l Thermal and epithermal fission converter neutron beams l Contamination: l l <1. 2 x 10 -13 Gy cm-2 fast neutrons l <3. 2 x 10 -13 Gy cm-2 Gamma radiation l 16 cm diameter field 5 x 109 ns-1 cm-2

Nuclear reactors Similar set ups were in existence at Brookhaven National Lab l High Flux Reactor in Petten, Netherlands l Fi. R 1 in Finland l R 2 -00 research reactor in Studsvik, Sweden l


D-T generators l 2 H + 3 H 4 He + 1 n + 17. 6 Me. V l Low E deuterons (100 -300 ke. V) l 14. 2 Me. V goes to neutron (monoenergetic) l E too low for fast neutron clinical applications l Isotropic: Thus low yield in any one direction l Low dose-rate (~15 c. Gy/min) l attempts to increase dose-rate by increasing d current burns up expensive tritium) l DD generators are similar to DT generator but doesn’t deal with radioactive tritium

Isotopic sources: Cf-252 (96. 9%) l 252 Cf fission + 1 n (3. 1%) Average of 3. 7 neutrons per spontaneous fission Energy range of 0 to 13 Me. V l l l l 252 Cf 248 Cm + 4 a Mean value of 2. 3 Me. V <E> ~1 Me. V T 1/2 = 2. 64 years Beam quality equal to reactor-based approaches

Isotopic sources of neutrons l l l 1 µg emits 2. 3 million n/s Requires ~100 µg for adequate flux for BNCT Approximately the entire annual production of Oak Ridge and the Research Institute of Atomic Reactors in Dimitrovgrad, Russia In principle could allow radioisotope driven 235 U subcritical multiplying assemblies Reduces needed 252 Cf 10 -fold Economics remain to be seen…

Producing neutrons l Cyclotron/linac stripping reactions l High-E deuterons (15 -50 Me. V) strike a Be or Li target and are “stripped” of their protons yielding a neutron beam l First suggested by J. Robert Oppenheimer l Single peak in spectrum l Average energy 40% of incoming deuteron
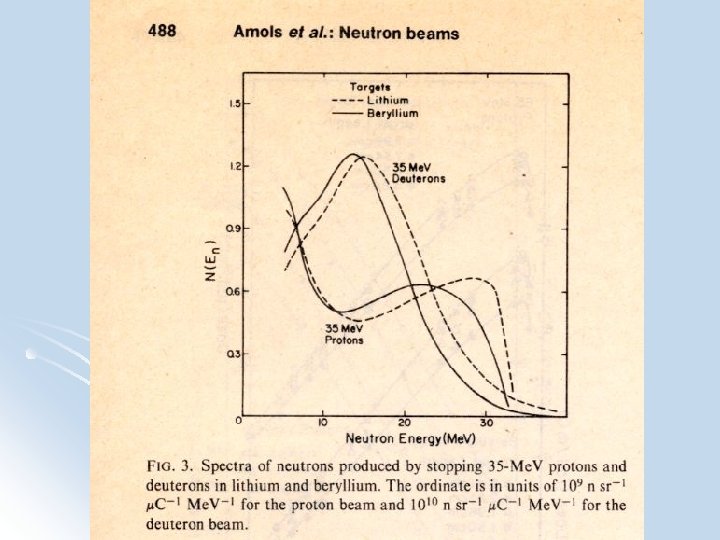

Producing neutrons l Knock-out process l Incoming p “knocks out” a n l Broad neutron E spectrum with a higher energy peak than stripping rnxs l Low E neutrons must be filtered out to harden beam for fast neutron clinical use l Resultant depth doses comparable to 6 MV linac


Accelerator approaches l l In principle cyclotrons or linacs could deliver even higher quality beams than ISRNB, cheaper, hospital based, less security concerns and inherently safer than reactors 7 Li(p, n)7 Be Lower energy less moderation needed higher n flux But ~10 m. A current required which is beyond hospital-based cyclotrons

Accelerator approaches l l Science research Labs (Somerville, MA) compact tandem cascade accelerator design 2. 5 Me. V p+ Li Goal: 4 m. A beam current Can be moderated & filtered to an epithermal beam

Accelerator approaches l l l 7 Li(p, n)7 Be has higher flux but has serious limitations Li has a low MP Poor thermal conductivity And the produced 7 Be is radioactive… Thus a trade-off on flux is needed: l l l 9 Be(p, n)9 B – high yield, higher MP, non-radioactive Suzuki et al: 2 m. A 30 Me. V p+ Be AS-BNCT(KURRI) > KUR-BNCT for deep seated tumors

Accelerator approaches l l l Tanaka et al: 1 m. A 30 Me. V p+ Be designed for Kyoto Treatment times less than 30 min and <12. 5 Gy. Eq to normal brain tissue Other options include 9 Be(d, n)10 B stripping rxn 12 C(d, n)13 N 13 C(d, n)14 N – may be best on paper as far as yield of appropriate E neutrons

Clinical results l USA: Clinical trials at BNL and MIT 1994 l Epithermal beams; Boron biodistribution studies; 3 D MC treatment planning 22 pts on Phase I/II trials; 20 with GBM BPAf via central line l l l l l 250 mg/kg x 1 hr (n= 10) 300 mg/kg x 1. 5 h (n = 2) 300 mg/kg x 2 h (n = 10) Med age = 45 (24 -78 yrs) No major side effects - alopecia and scalp dermatitis 76% radiographic regression followed by stability Med OS = 13 mo

Clinical results l Finland: Clinical trials at Fi. R 1 l 18 pts 1999 -2001 Epithermal beams BPAf 290 – 400 mg/kg All had surgery; no chemo; no external beam Avg PTV dose = 30 – 61 Gy(W) Avg nl brain dose = 3 – 6 Gy(W) Single fraction with opposed laterals Well tolerated - no deaths in first 6 months (6 mo OS =100%) 1 yr OS = 61% l l l l

Clinical results l Sweden: Clinical trials at R 2 -00 Studsvik l 30 GBM pts age 26 to 69 yrs in a Phase II trial 26 had surgical debulking Epithermal beams BPAf 6 hr infusion of 900 mg/kg Neutron irradiation began 2 hrs after end of infusion All had surgery; no chemo; no external beam Avg GTV dose = 15. 4 – 4. 3 Gy(W) Avg nl brain dose = 3. 2 – 6. 1 Gy(W) 4 had GI effects probably secondary to BPAf Med OS = 14. 2 mo Salvage temozolamide led to 17. 7 mo l l l l l

Clinical results Japan: l Dr Hiroshi Hatanaka (Tokyo) l Began in 1968 at Hitachi Nuclear Reactor l BSH (Na. B 12 H 11 SH) l l l l 183 total pts Nakagawa et al (1997): 149 pts; 64 with GBM Thermal neutrons beams 7% GBM pts and 56% AA pts survived > 2 yrs Med OS: GBM = 640 days; AA = 1811 days Range: GBM: 39 – 8138 days(!); AA: 17 - 6641 days

Clinical results l l l l l Nakagawa et al (2003): 105 pts treated 1978 – 97 10 with mixed beams, rest thermal neutrons beams All surgically debulked 2 wks before BNCT 100 mg/kg BSH starting 12 -13 hrs before BNCT Balloon placed into surgical cavity to maintain size during BNCT Intraoperative approach Min tumor dose = 15 Gy Min target dose = 18 Gy Max vascular dose <15 Gy Total gamma radiation dose <10 Gy

Clinical results Nakagawa et al (2003) Results: l 5 of 10 GBM pts died l Of these five l l 2 showed local recurrence l 3 showed disease elsewhere in brain (dissemination) but no local recurrence on autopsy l Five of the ten were still alive in 2003 (at least five years later, maybe 24 yrs? )

Clinical results Kawabuta et al (2008) l 21 GBM pts 2002 – 07 l Epithermal beams; BSH plus BPA l 10 BNCT only: Med OS = 14. 1 mo l 11 pts with fractionated EBRT boost of 20 to 30 Gy : Med OS = 23. 5 months l

Clinical results Epithermal neutrons l Non-surgical approach l BSH plus BPA l 30 Gy EBRT boost to deepest part l Temozolamide on recurrence l Med OS = 25 months l



Fermilab Neutrons Produced by proton on Be knock-out process l 9 Be(p, n)9 B l 66 Me. V p Be l l p+(66)Be(49) Max E ~ 66 Me. V l Mean E = 25 Me. V l

The Spoiler




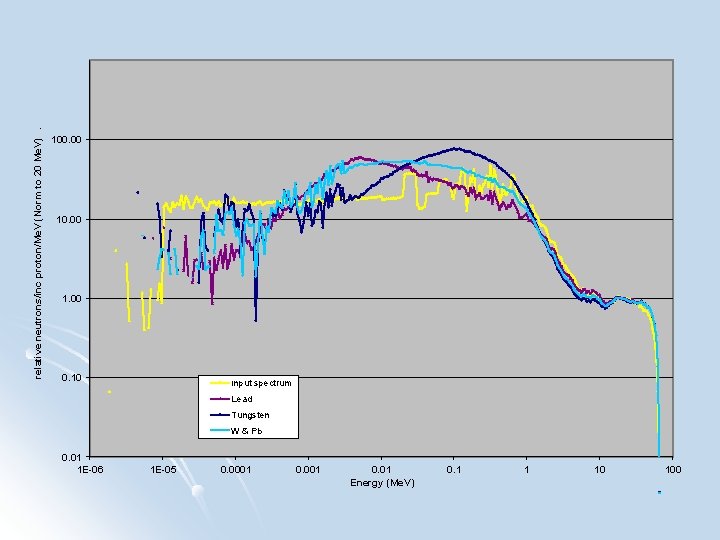
relative neutrons/inc proton/Me. V (Norm to 20 Me. V). 100. 00 1. 00 0. 10 input spectrum Lead Tungsten W & Pb 0. 01 1 E-06 1 E-05 0. 0001 0. 01 Energy (Me. V) 0. 1 1 10 100
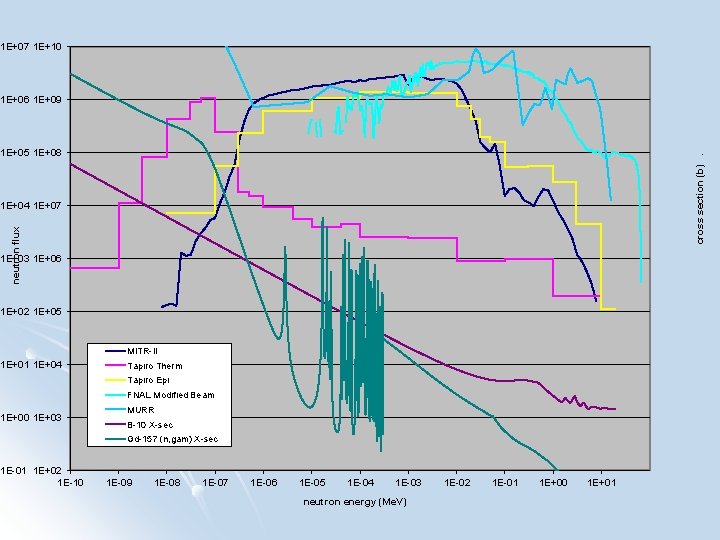
neutron energy (Me. V) cross section (b). neutron flux

Conclusions l l Even in its current iteration, BNCT appears equivalent to current standard treatment (maximal surgical resection, IG-IMRT plus concurrent temozolamide and adjuvant temozolamide) for GBM l but in a single session Modernized BNCT (epithermal neutron beams, new nontoxic tumor-localizing boron compounds, 3 D treatment planning, in-vivo boron quantitative methods) l Possibly fractionated with integrated temozolamide or external beam radiation therapy Could be a very valuable treatment modality for GBM Could be possible at FNAL
- Slides: 76