Potato Lab SET UP LAB Each lab table

Potato Lab • SET UP LAB: • Each lab table will prepare petris of 6 salt solutions. • Record mass and length of each potato piece. • Record turgidity of each piece of potato. • The potatoes will then soak for 30 minutes. Turgid (Turgidity) is how “rigid” or hard something feels. This is opposite of flaccid which is how “floppy” something feels. Turgidity is measured by feeling. Rate it from 1 to 5. 5 is the hardest (most rigid). 1 is the softest and most floppy.

KEY VOCAB Hypertonic solution is when lots of solute (e. g. sugar/salt) exists outside the cell so the water moves out of the cell. That tells us there is a HIGH concentration of solute outside the cell compared to inside. (Cell SHRINKS) Isotonic solution is when the solution of water inside is equal to outside the cell Hypotonic solution is when small amount of solute (e. g. sugar) exists outside the cell so the water moves into of the cell. That tells us there is LOW concentration of solute outside the cell compared to inside. (GROWS FATTER/MORE TURGID)

HYPERTONIC SOLUTION HYPOTONIC CELL X = Solute (e. g. Sugar/Salt) O = Solvent (e. g. Water H 2 O) Cell shrinks and becomes FLACCID Outside concentration = 8/18 = 44% Inside concentration = 1/6 = 17%
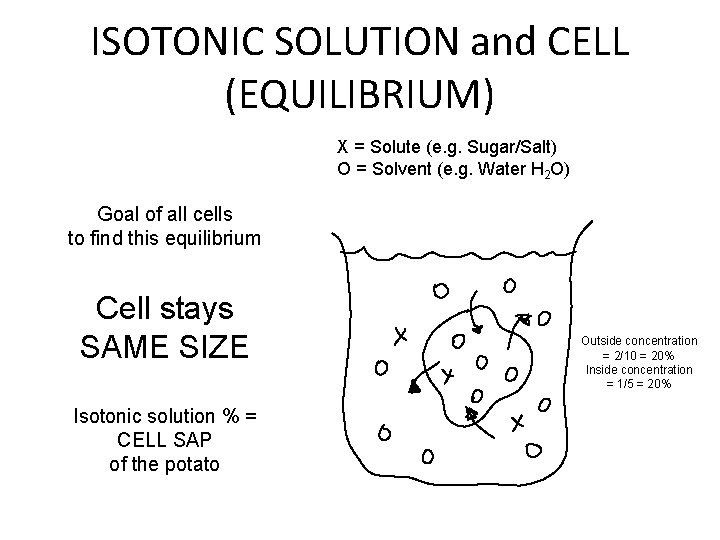
ISOTONIC SOLUTION and CELL (EQUILIBRIUM) X = Solute (e. g. Sugar/Salt) O = Solvent (e. g. Water H 2 O) Goal of all cells to find this equilibrium Cell stays SAME SIZE Isotonic solution % = CELL SAP of the potato Outside concentration = 2/10 = 20% Inside concentration = 1/5 = 20%
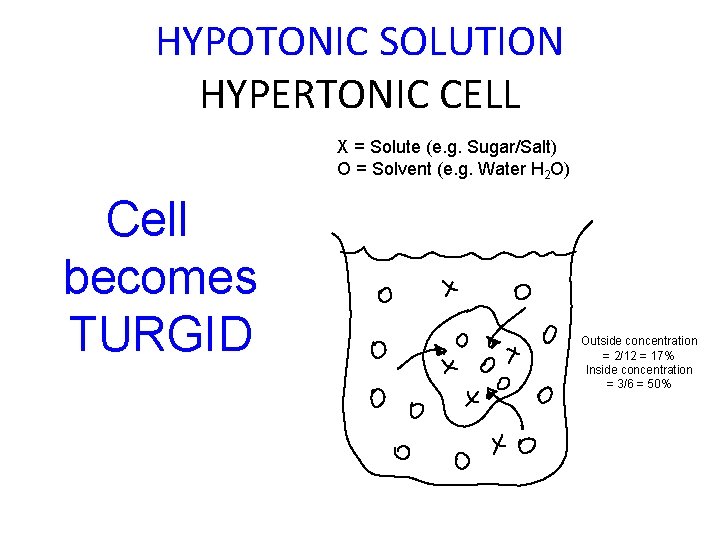
HYPOTONIC SOLUTION HYPERTONIC CELL X = Solute (e. g. Sugar/Salt) O = Solvent (e. g. Water H 2 O) Cell becomes TURGID Outside concentration = 2/12 = 17% Inside concentration = 3/6 = 50%

Helpful Websites for Osmosis: Diffusion of Water http: //www 2. nl. edu/jste/osmosis. htm http: //www. tvdsb. on. ca/westmin/science/sbi 3 a 1/Cells /Osmosis. htm

WHILE WE WAIT write your title (centered) write your problem or purpose: Problem: How does the independent var affect the dependent variable? list the Variables: Independent: Dependent: Controlled Variables (you need at least 3) include your data from the lab Data Table: (title) Make data table in excel or draw by hand.
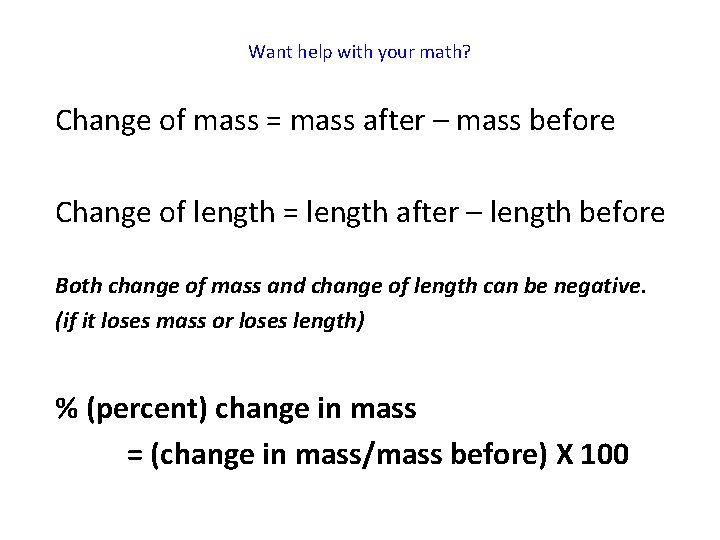
Want help with your math? Change of mass = mass after – mass before Change of length = length after – length before Both change of mass and change of length can be negative. (if it loses mass or loses length) % (percent) change in mass = (change in mass/mass before) X 100
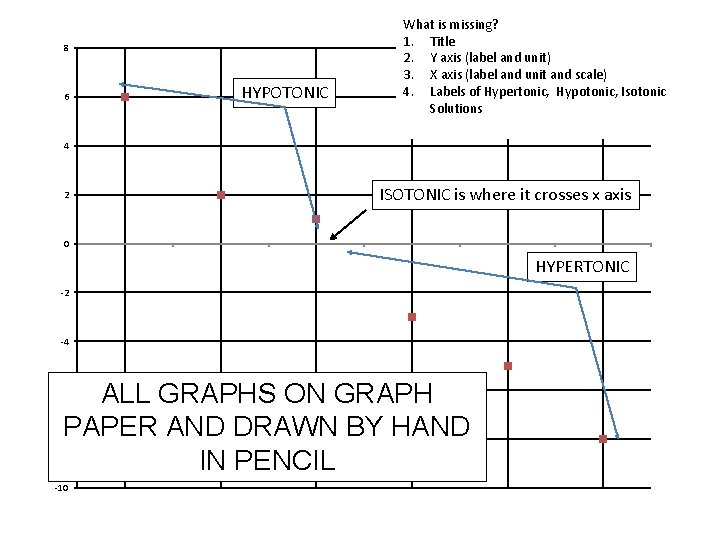
8 6 HYPOTONIC What is missing? 1. Title 2. Y axis (label and unit) 3. X axis (label and unit and scale) 4. Labels of Hypertonic, Hypotonic, Isotonic Solutions 4 2 ISOTONIC is where it crosses x axis 0 HYPERTONIC -2 -4 ALL GRAPHS ON GRAPH PAPER AND DRAWN BY HAND IN PENCIL -6 -8 -10

Next you should make 2 graphs: Graph 1: (title) graph change in mass (g) vs. Na. Cl solution (%) Graph 2: (title) graph change in length (cm) vs. Na. Cl solution (%)

Conclusion: • From my data and my line of best fit, I have determined that the concentration of Na. Cl in the cell sap is ______%. • Also from my data, I determined that the Na. Cl solution (outside the potato cells) is hypertonic from _____% to ______%. I know this because the cell got _____(bigger/smaller) in this range and I’m assuming it’s because of osmosis, when water went ______(into/out of) the cell. Also from my data, I determined that the Na. Cl solution (outside the potato cells) is hypotonic from _____% to ______%. I know this because the cell got _____(bigger/smaller) in this range and I’m assuming it’s because of osmosis, when water went ______(into/out of) the cell.

Evaluation: • Make chart (boxed and numbered) • 3 LIMITATIONS & 3 corresponding IMPROVEMENTS

Lab is Due Monday Oct 15 this powerpoint is posted on my website: lmmurray. weebly. com
- Slides: 13