Potassium Homeostasis Its disorders By Dr Mohammad ElTahlawi

Potassium Homeostasis & Its disorders By Dr. Mohammad El-Tahlawi

Objectives Potassium homeostasis Hypokalamia Definition Causes Effects Diagnosis Treatment

POTASSIUM Potassium play an important role in: 1 -Electerophysiology of cell membrane for all cells in which polarizationdepolaization cycles are functionally relevant(cardiac and neuromuscular cells). 2 -Carbohydrates and protien synthesis

POTASIUM DISTRIBUTION Extracellular 2% 70 meq Intracellular 98% 3430 meq Plasma 20% 15 meq Na-K ATPase K content = 50 meq/kg In 70 kg Total body K = 3500 meq
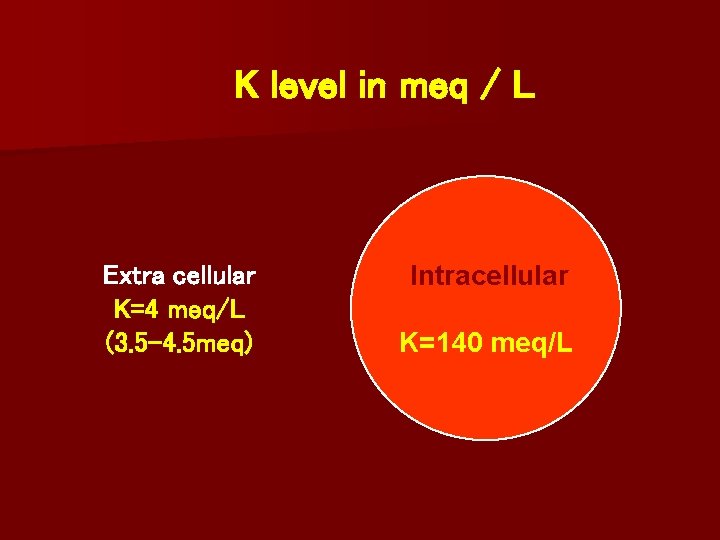
K level in meq / L Extra cellular K=4 meq/L (3. 5 -4. 5 meq) Intracellular K=140 meq/L

Decrease in plasma K from 4 - 3 meq/L intracellular K deficit BY 100 - 200 meq

Decrease in plasma K from 3 - 2 meq/L intracellular K deficit BY 200 - 400 meq

Plasma K concentration Correlates poorly with the total body k deficit
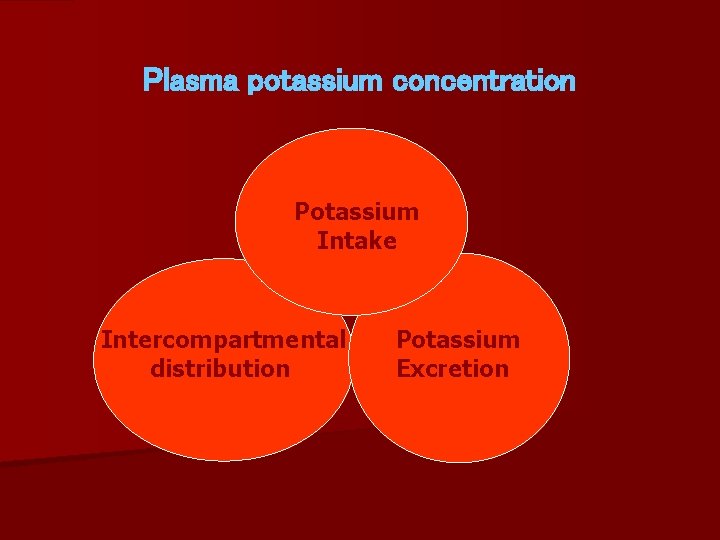
Plasma potassium concentration Potassium Intake Intercompartmental distribution Potassium Excretion

Dietary K intake = 80 meq/day Excretion = 70 meq/day (urine). = 10 meq/day (GIT).
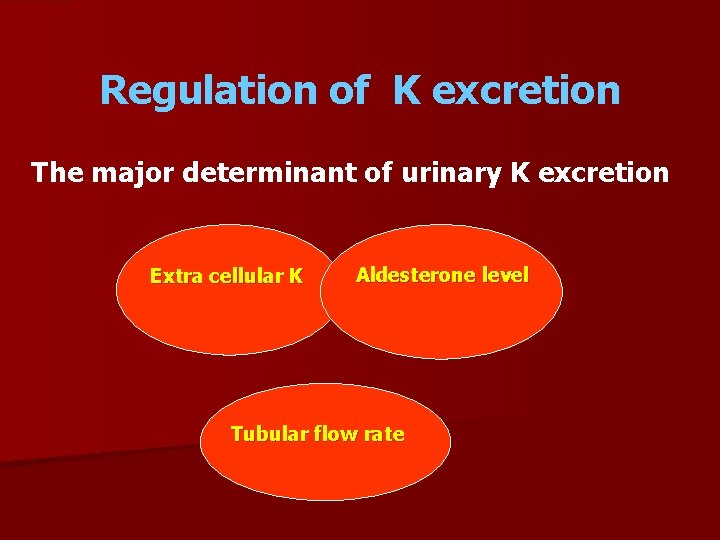
Regulation of K excretion The major determinant of urinary K excretion Extra cellular K Aldesterone level Tubular flow rate

Intercompartmental shift of Potassium 1 - Extracellular p. H. 2 - Circulating insulin level. 3 - Circulating catecholamine activity. 4 - Plasma osmolality. 5 - Hypothermia. 6 - Exercise.
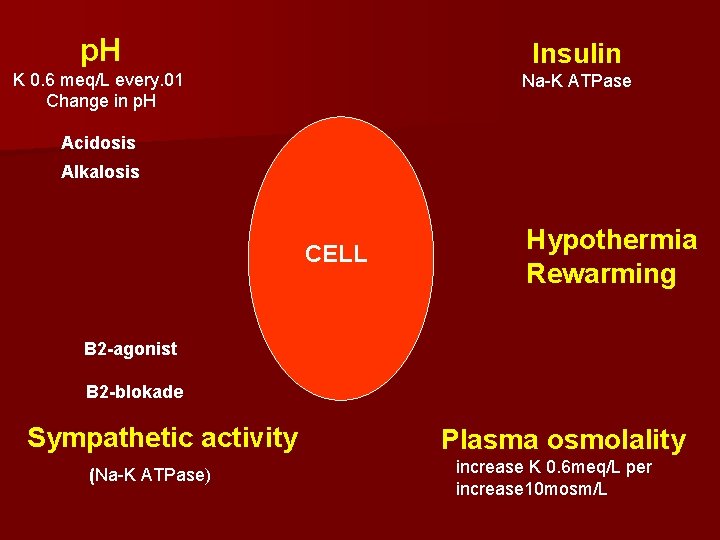
p. H Insulin K 0. 6 meq/L every. 01 Change in p. H Na-K ATPase Acidosis Alkalosis CELL Hypothermia Rewarming B 2 -agonist B 2 -blokade Sympathetic activity (Na-K ATPase) Plasma osmolality increase K 0. 6 meq/L per increase 10 mosm/L

HYPOKALEMIA

HYPOKALAEMIA (K ion less than 3. 5 meq/L) Causes: 1 -Intercompartmental shift of K. 2 -Increase k loss. 3 -Inadequate k intake.

Causes of hypokalamia Intercompartmental shift of K: Alkalosis Insulin administration B 2 adrenergic agonist Hypothermia Treatment of megaloplastic anaemia Periodic paralasis Transfusion of frozen blood

Causes of hypokalamia Increase K losses (Renal or extrarenal) Renal: Diuretics Increase mineralocorticiod activity Renal tubular acidosis Ketoacidosis Hypomagesaemia Urinary diversion with long ileal loop Carbinecillin and Amphotericin B

Causes of hypokalamia Extrarenal: GIT : Diarrhea, Vomiting, Fistula, Laxative abuse, Urinary diversion. Sweet Dialysis Decrease K intake

Effects of hypokalemia Most of the patients are asymptomatic until K level below 3 meq/L. Cariovascular effects are most prominent

Effects of hypokalamia Cardiovascular ECG changes Dysrhythmia Myocardial dysfunction Myocardial fibrosis Orthostatic hypotension Increase digitalis toxicity
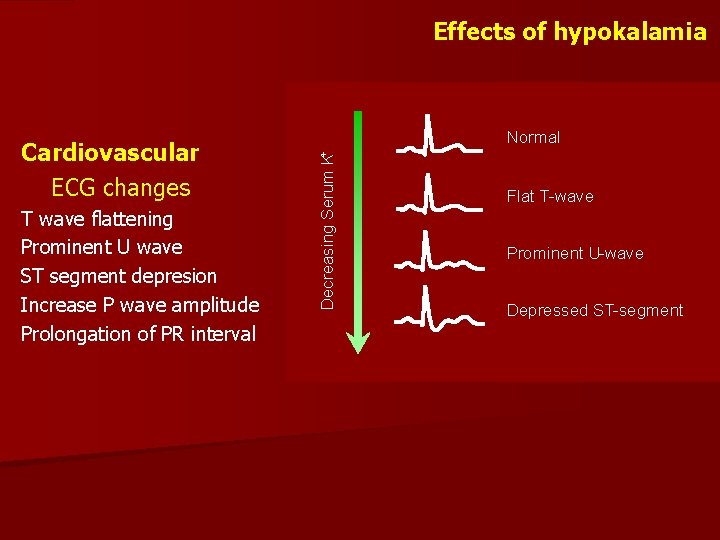
Effects of hypokalamia T wave flattening Prominent U wave ST segment depresion Increase P wave amplitude Prolongation of PR interval Decreasing Serum K+ Cardiovascular ECG changes Normal Flat T-wave Prominent U-wave Depressed ST-segment

Effects of hypokalamia Neuromuscular Skletal ms. Weakness up to respiratory failure. Tetany Rhabdomyolysis Ileus , Urine retention Renal Polyuria Increase amonium production Increase HCO 3 reabsorption Increase Na retension Increased renin secretion→ increase Ang. II→ thirst

Effects of hypokalamia Metabolic Decrease insulin secretion Decrease growth hormone secretion Decrease aldesterone secretion Hormonal Negative nitrogen balance Encephalopathy in liver disease

Approach to diagnosis
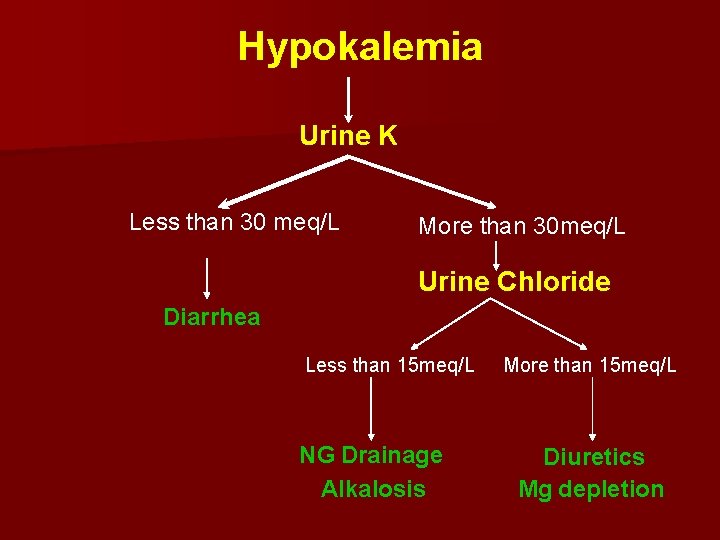
Hypokalemia Urine K Less than 30 meq/L More than 30 meq/L Urine Chloride Diarrhea Less than 15 meq/L NG Drainage Alkalosis More than 15 meq/L Diuretics Mg depletion

Treatment of hypokalemia The goal of therapy: Is to remove the patient from immediate danger and not necessarily to correct the entire K deficit. Firstly concern : Any condition that promotes transcellular K shift.

Potassium replacement Oral replacement with Kc. L solution is generally safe(60 -80 meq/d) IV replacement : (Remember ) Serious cardiac manifestation. Peripheral line not exceed 8 meq/h. More than 8 meq/h, centeral line is indicated. Dextrose containing solution should be avoided. ECG monitoring is mandatory in high rate infusion.

Potassium replacement Solutions Potassium chloride and potassium phosphate Kcl: is available in 2 meq/m. L (5 ml) is of choice with metabolic alkalosis as it corrects chloride shifts. Osmolality = 4000 mosm/kg. H 2 O K phosphate: is of choice with coexisting hypophatemia (e. g DKA)

Potassium replacement Deficit =(3. 5 - acutal serum K ) x 0. 4 BW Maintenence = 1 meq / kg BW / day

Potassium replacement Infusion rate (pripheral line) Not exceed 8 meq / h Infusion rate (centeral line) Standard method = 20 meq Kc. L in 100 ml saline/h Maximum rate (serum k less than 1. 5 meq/L) We need peripheral line = 40 meq kc. L / h = ( ½ BW meq/h)

Practical approach If K level <2 m. Eq/L, deficit= 0. 4 x wt(normal – measured K) we can give up to 0. 5 m. Eq/kg/hr. 2. If K level reaches 2. 5 m. Eq/L, slowly corrects K by giving 10 m. Eq/hr. 3. Add the daily intake (1 m. Eq/kg) 1.

n n 1. 2. 3. n It is advisable to give K salts into large but not central vein. Potassium products: IV preparations Oral: 15 ml= 40 m. Eq (if conc. Of KCl in sol. is 10%) Natural sources: -Orange: one orange=300 mg K one litre juice=2. 8 gm K -Bananas: one piece= 750 mg K K therapy in pediatrics: 1 -3 m. Eq/kg/every 1 m. Eq decrease in K level with max. 3 m. Eq/kg/day

Response to the treatment At first The serum K may be slow to rise particularly if K losses are ongoing Full replacement usually takes few days. If there is refractory hypokalemia check magnessium level


CONCLUSION Potassium has important role to vital body function. Plasma K concentration is a function of relationship between entry, the intercompartemental distribution and excretion of K. Hypokalemia : serum K less thd 3. 5 meq/L Cause : Decrease intake, Losses and Intercompartemental shift. Effects : Cardiovascular, Neuromuscular, renal, Hormonal and metabolic. Diagnosis. Treatment : Goals, replacement and response

Hyperkalemia
![Hyperkalemia Plasma [K+] > 5. 0 Hyperkalemia may be the result of disturbances in Hyperkalemia Plasma [K+] > 5. 0 Hyperkalemia may be the result of disturbances in](http://slidetodoc.com/presentation_image_h/7c8a9a0dba229287ad71a6be50c0dd4a/image-37.jpg)
Hyperkalemia Plasma [K+] > 5. 0 Hyperkalemia may be the result of disturbances in external balance (total body K+ excess) or in internal balance (shift of K+ from intracellular to extracellular compartments)
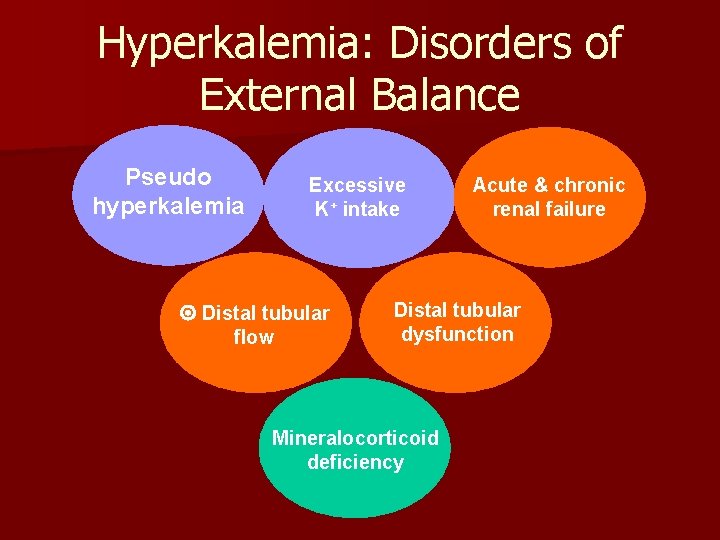
Hyperkalemia: Disorders of External Balance Pseudo hyperkalemia Excessive K+ intake Distal tubular flow Acute & chronic renal failure Distal tubular dysfunction Mineralocorticoid deficiency

n Pseudohyperkalemia n Movement of K+ out of cells during or after blood drawing §Hemolysis §Fist clenching (local exercise effect) §Marked leukocytosis

Hyperkalemia: Disorders of External Balance Excessive Potassium Intake § Oral or Parenteral Intake § K pencillin in high doses § Stored blood

Hyperkalemia: Disorders of External Balance Decreased Renal Excretion Acute and Chronic Renal Failure Decreased Distal Tubular Flow § Volume depletion § Decreased effective arterial blood volume (CHF, cirrhosis) § Drugs altering glomerular hemodynamics with a decrease in GFR (NSAIDs, ACE inhibitors, ARBs) Mineralocorticoid Deficiency § Combined glucocorticoid and mineralocorticoid (adrenal insufficiency) § Hyporeninemic hypoaldosteronism (diabetes mellitus) § Drug-induced (ACE inhibitors, ARBs) Distal Tubular Dysfunction § Disorders causing impaired renal tubular function with hyporesponsiveness to aldosterone (interstitial nephritis) § Potassium-sparing diuretics (amiloride, triamterene, spironolactone)

Hyperkalemia: Disorders of Internal Balance – Insulin deficiency – 2 -Adrenergic blockade – Hypertonicity – Acidemia – Cell lysis

Clinical Manifestations of Hyperkalemia Clinical manifestations result primarily from the depolarization of resting cell membrane potential in myocytes and neurons Prolonged depolarization decreases membrane Na+ permeability through the inactivation of voltage-sensitive Na+ channels producing a reduction in membrane excitability Cardiac toxicity

EKG Manifestations of Hyperkalemia Increasing Serum K+ Normal Peaked T-wave Wide QRS Complex Shortened QT Interval Prolonged PR Interval Further Widening of QRS Complex Absent P-Wave Sine-Wave Morphology (e. g. Ventricular Tachycardia)

Medical Treatment of Hyperkalemia Membrane Stabilization – IV calcium Internal Redistribution – IV insulin (+ glucose) – -adrenergic agonist (albuterol inhaled) Enhanced Elimination – Kayexalate (sodium polystyrene sulfonate) ion exchange resin – Loop diuretic – Hemodialysis

Practical approach n Mild cases: K<6. 5 m. Eq/L→causal management n Moderate cases: K=6. 5 -8 m. Eq/L: -glucose infusion. -glucose insulin infusion. -Na. HCO 3 n Severe cases: K>8 m. Eq/L→calcium injection

Emergency measures: -Dextrose 10%: 200 -500 ml over 30 min. 500 -1000 ml over the next few hours. -Dextrose/insulin infusion Insulin: 0. 1 U/kg then 1 U/kg/hr (add minimum 2 -3 glucose/U insulin). Onset of effect is 1 -5 min. -Na. HCO 3: 150 m. Eq over several minutes ? increased p. H causes K shift into cells. n

Definitive measures: n Key oxalate (Na polysterene) -Oral: 15 -30 g 2 -4 times/day + sorbitol 20 -25% (50 ml/15 gm resin) The resin induces diarrhea and leads to K loss. -Retention enema: 50 gm in 200 ml sorbitol 25%. Every gm resin combines with 1 m. Eq K in GIT. n Dialysis : in cases of RF. n


Potassium Disorders n Normal homeostasis n Hypokalemia – Etiologic factors – Algorithm for diagnosis n Hyperkalemia – Etiologic factors – Algorithm for diagnosis
- Slides: 50