PostGolgi biosynthetic pathways MDCKcell Resting fibroblast Migrating fibrobl
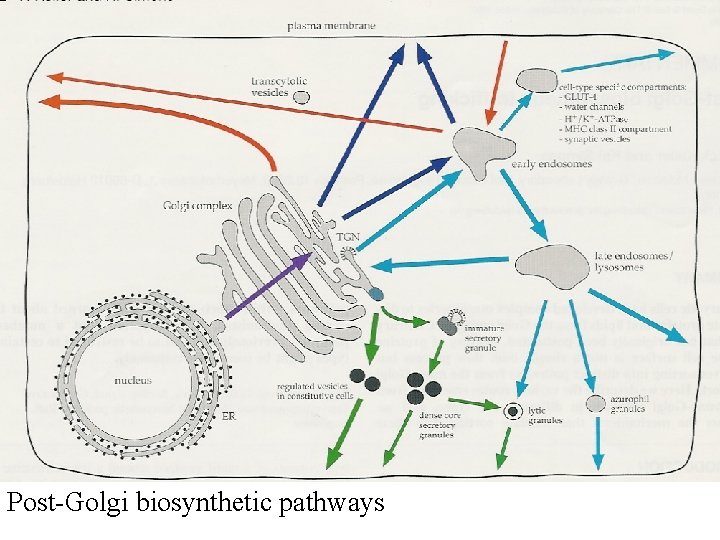
Post-Golgi biosynthetic pathways
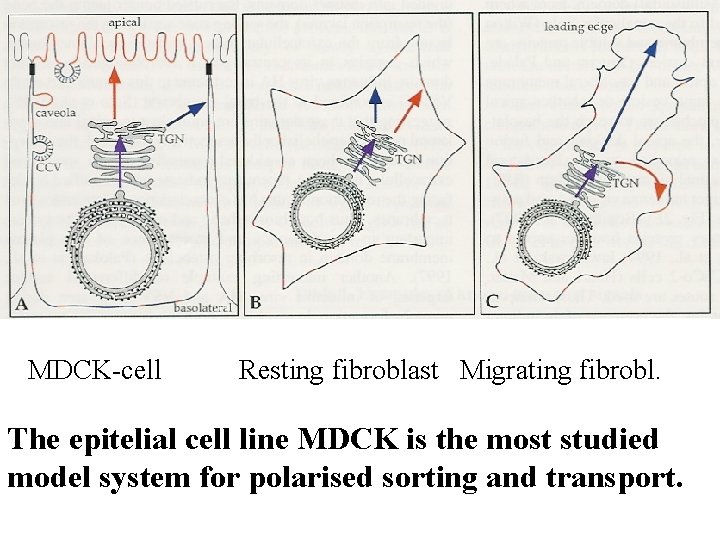
MDCK-cell Resting fibroblast Migrating fibrobl. The epitelial cell line MDCK is the most studied model system for polarised sorting and transport.
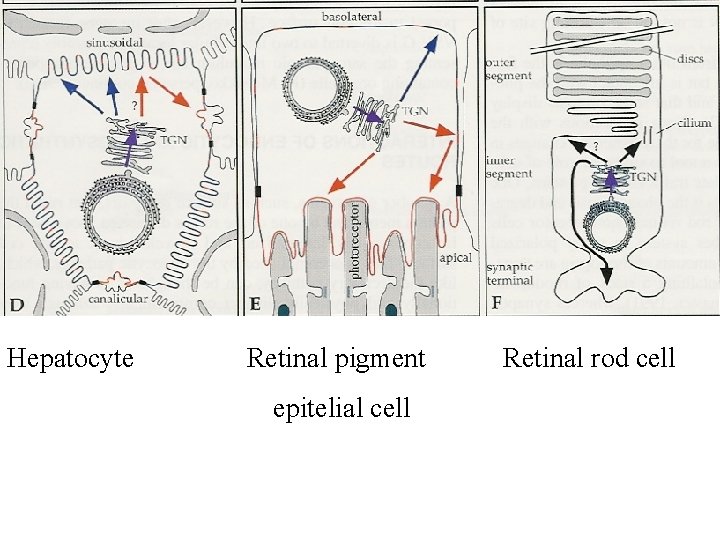
Hepatocyte Retinal pigment epitelial cell Retinal rod cell
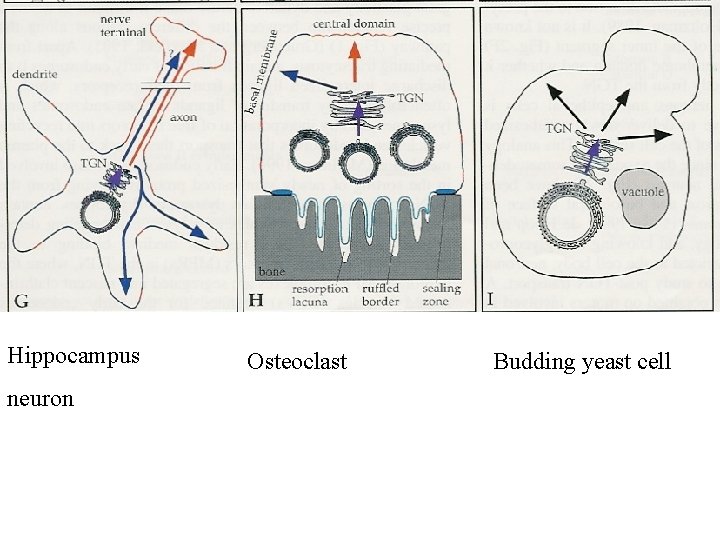
Hippocampus neuron Osteoclast Budding yeast cell
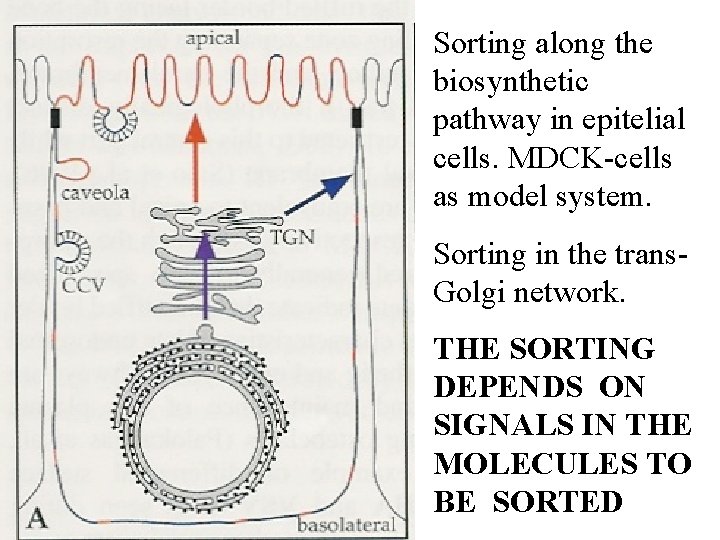
Sorting along the biosynthetic pathway in epitelial cells. MDCK-cells as model system. Sorting in the trans. Golgi network. THE SORTING DEPENDS ON SIGNALS IN THE MOLECULES TO BE SORTED
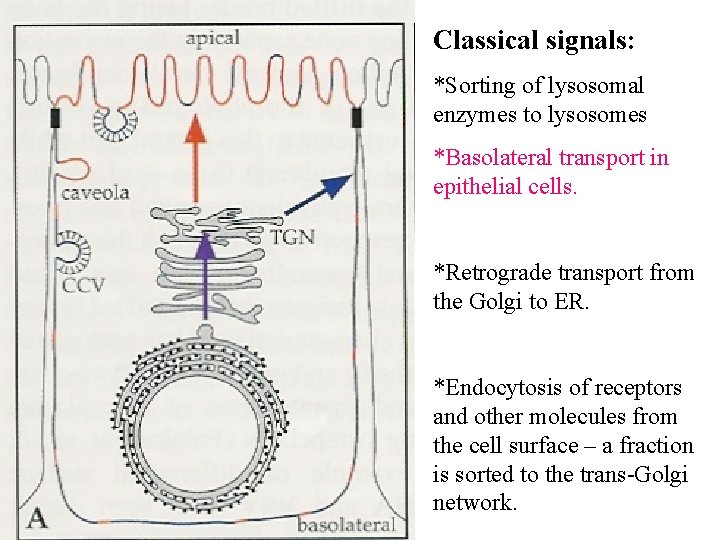
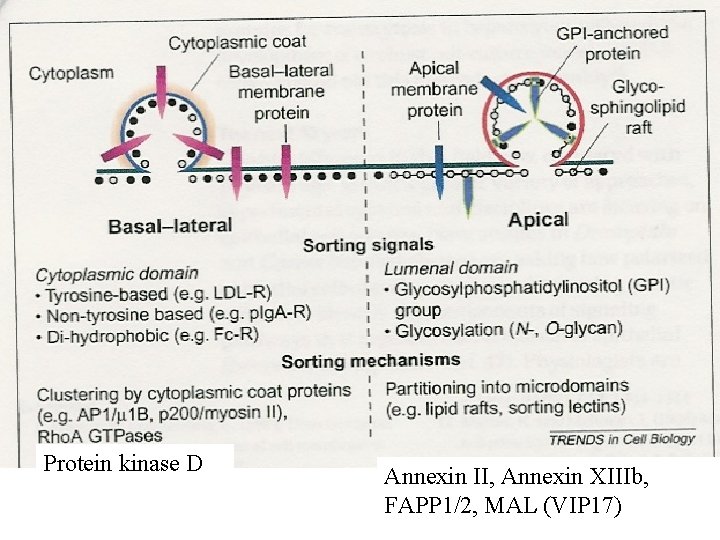
Classical signals: *Sorting of lysosomal enzymes to lysosomes *Basolateral transport in epithelial cells. *Retrograde transport from the Golgi to ER. *Endocytosis of receptors and other molecules from the cell surface – a fraction is sorted to the trans-Golgi network.
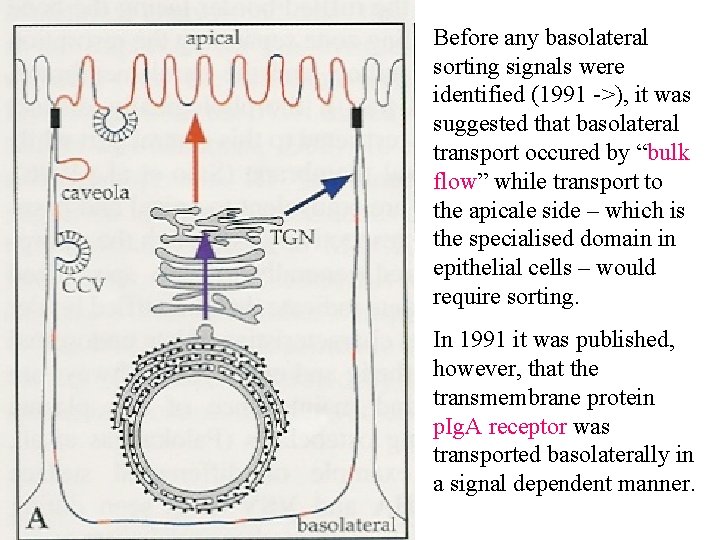
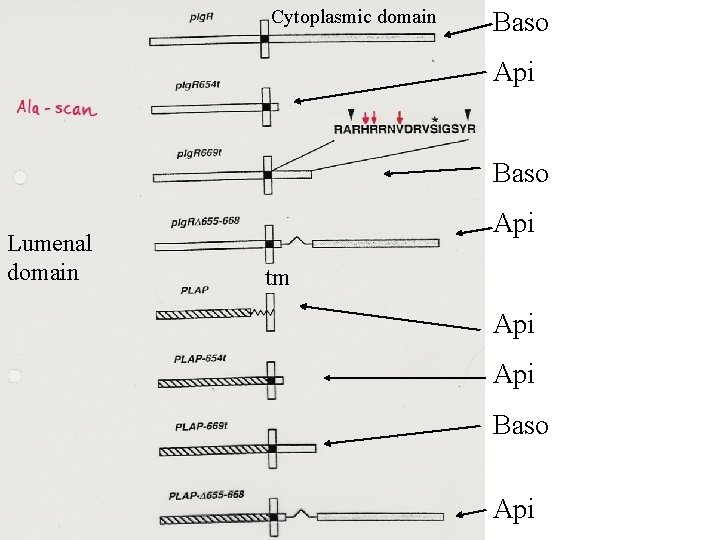
Before any basolateral sorting signals were identified (1991 ->), it was suggested that basolateral transport occured by “bulk flow” while transport to the apicale side – which is the specialised domain in epithelial cells – would require sorting. In 1991 it was published, however, that the transmembrane protein p. Ig. A receptor was transported basolaterally in a signal dependent manner.
Growth of MDCK epitelial cells on filters. Transfer to glass-dishes with 90 ml of medium for establishment of confluent cell layers.
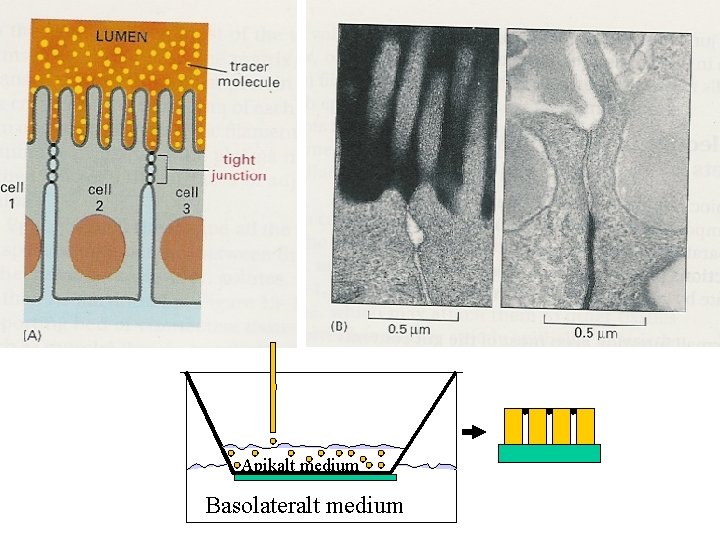
Apikalt medium Basolateralt medium

PROTEIN SORTING STUDIES IN THE SECRETORY PATHWAY OF EPITHELIAL CELLS A protein transported equally well to the apical or basolateral surfaces is regarded as a protein without a sorting signal. Secretory proteins are the ones most easily studied, because these may be identified after harvest of the two opposite media of filter-grown cells. Apical or basolateral membrane proteins may be extracted after differetial biotinylation of the two opposite sides and/or recognition by specific antibodies. Sorting signals are autonomous – they work after ”transplantation” to normally non-sorted molecules.
Cytoplasmic domain Baso Api Baso Lumenal domain Api tm Api Baso Api
Lumenal domain Transmembrane domain Golgi membrane /endosomal membrane Cytoplasmic domain
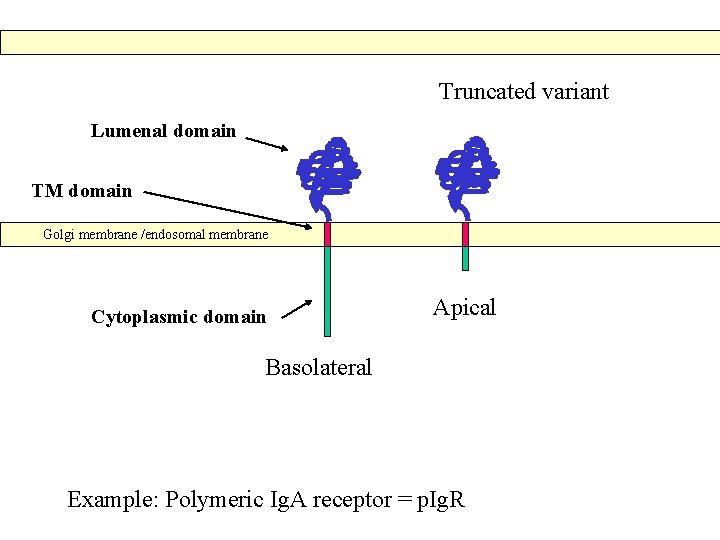
Truncated variant Lumenal domain TM domain Golgi membrane /endosomal membrane Cytoplasmic domain Apical Basolateral Example: Polymeric Ig. A receptor = p. Ig. R
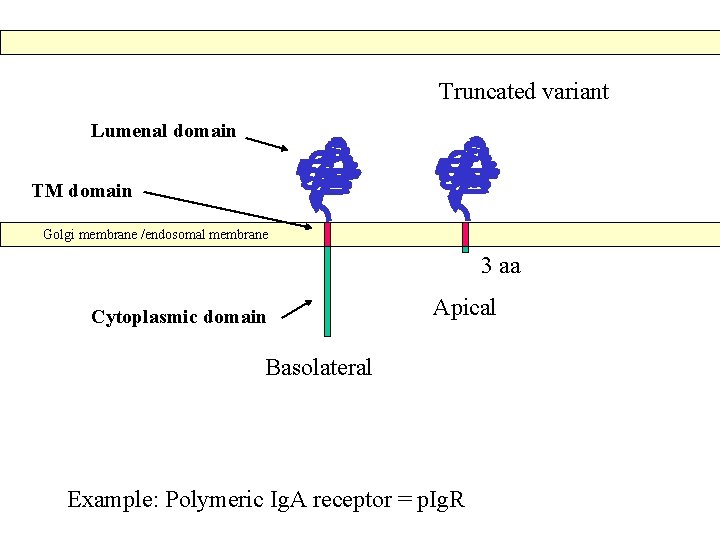
Truncated variant Lumenal domain TM domain Golgi membrane /endosomal membrane 3 aa Cytoplasmic domain Apical Basolateral Example: Polymeric Ig. A receptor = p. Ig. R
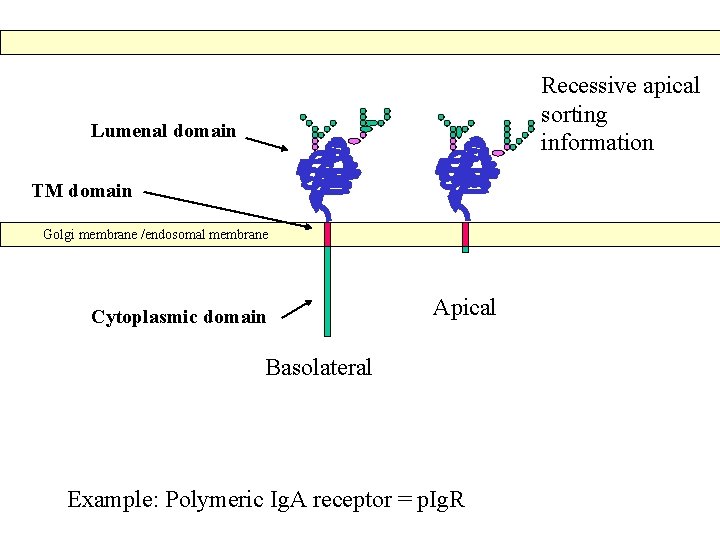
Recessive apical sorting information Lumenal domain TM domain Golgi membrane /endosomal membrane Cytoplasmic domain Apical Basolateral Example: Polymeric Ig. A receptor = p. Ig. R
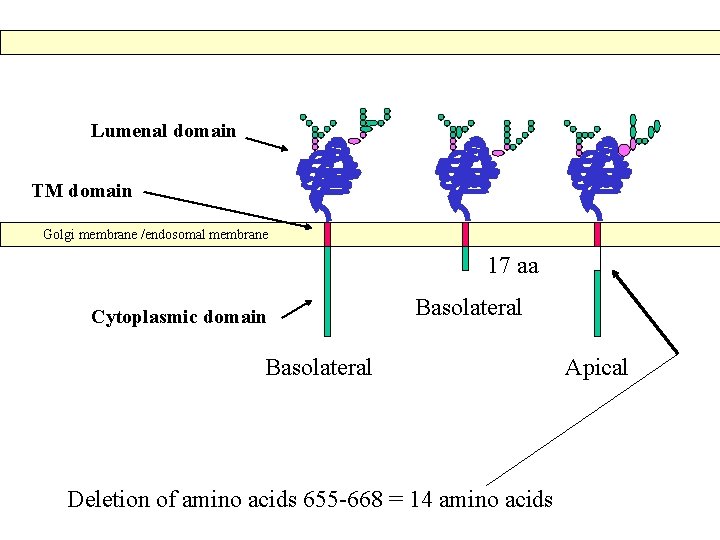
Lumenal domain TM domain Golgi membrane /endosomal membrane 17 aa Cytoplasmic domain Basolateral Deletion of amino acids 655 -668 = 14 amino acids Apical
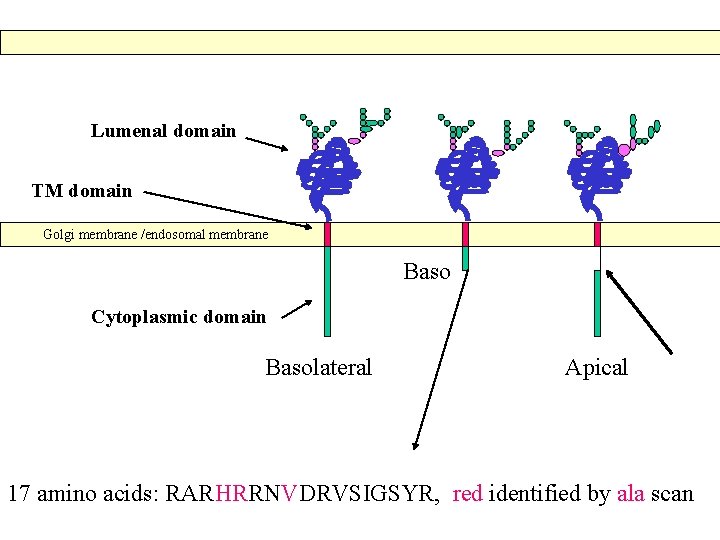
Lumenal domain TM domain Golgi membrane /endosomal membrane Baso Cytoplasmic domain Basolateral Apical 17 amino acids: RARHRRNVDRVSIGSYR, red identified by ala scan
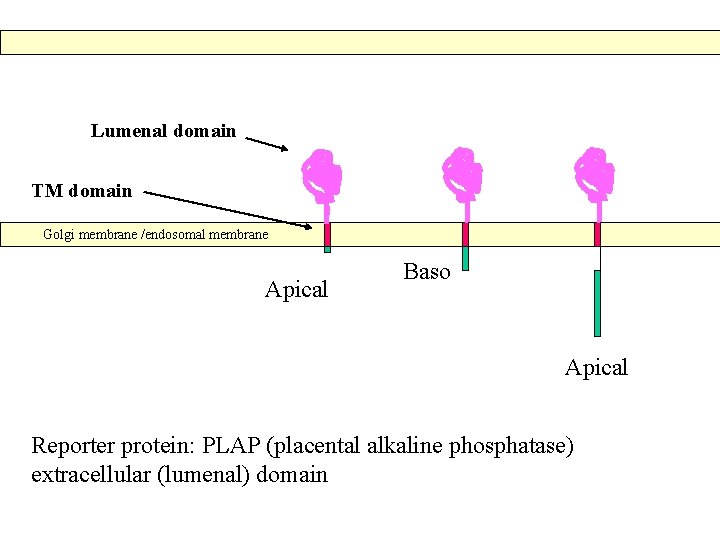
Lumenal domain TM domain Golgi membrane /endosomal membrane Apical Baso Apical Reporter protein: PLAP (placental alkaline phosphatase) extracellular (lumenal) domain

WHAT MOTIFS ARE BASOLATERAL SORTING SIGNALS? YXX (YVEL/YTDI/YXRF) bulky/hydrophobic TGN 38, M 6 PR LL / IL / LEL Fc Receptor, MHC II Invariant chain, EGF receptor NPXY Low density lipoprotein (LDL) receptor H/R-XXV Polymeric Ig. A receptor, Mannose-6 -phosphate rec. (? ) PXXP Epidermal growth factor (EGF) receptor Other Transferrin receptor (VDGDNSHVEMKLA) Some proteins have several sorting signals in their cytoplasmic tail. These are recognized at different intracellular sites, to provide recycling to the basolateral domain.

BASOLATERAL SORTING SIGNALS Some basolateral sorting signals overlap with endocytosis signals. *Fc receptor *Asialoglycoprotein receptor *Lysosomal acid phosphatase Other basolateral sorting signals are distinct from endocytosis signals. *Polymeric Ig. A receptor *LDL receptor *Transferrin recptor (? )
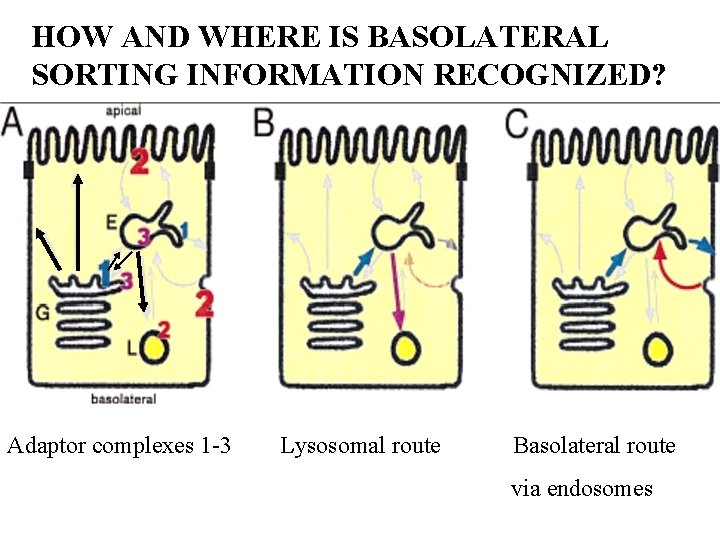
HOW AND WHERE IS BASOLATERAL SORTING INFORMATION RECOGNIZED? Adaptor complexes 1 -3 Lysosomal route Basolateral route via endosomes
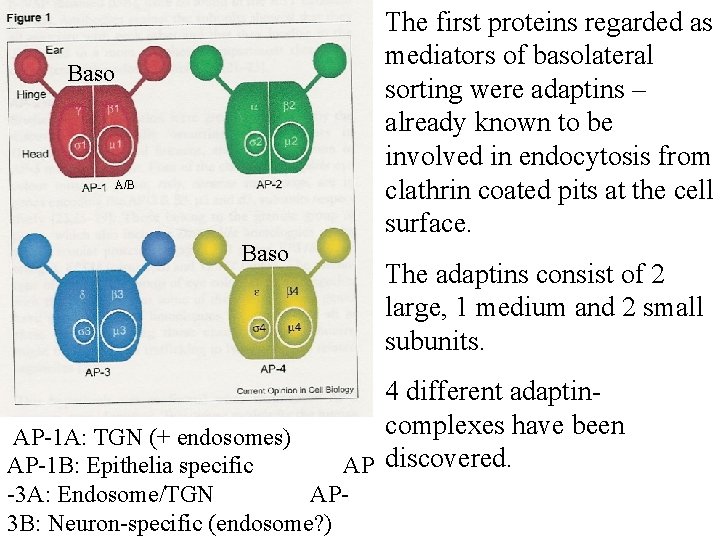
The first proteins regarded as mediators of basolateral sorting were adaptins – already known to be involved in endocytosis from clathrin coated pits at the cell surface. Baso A/B Baso The adaptins consist of 2 large, 1 medium and 2 small subunits. 4 different adaptincomplexes have been AP discovered. AP-1 A: TGN (+ endosomes) AP-1 B: Epithelia specific -3 A: Endosome/TGN AP 3 B: Neuron-specific (endosome? )
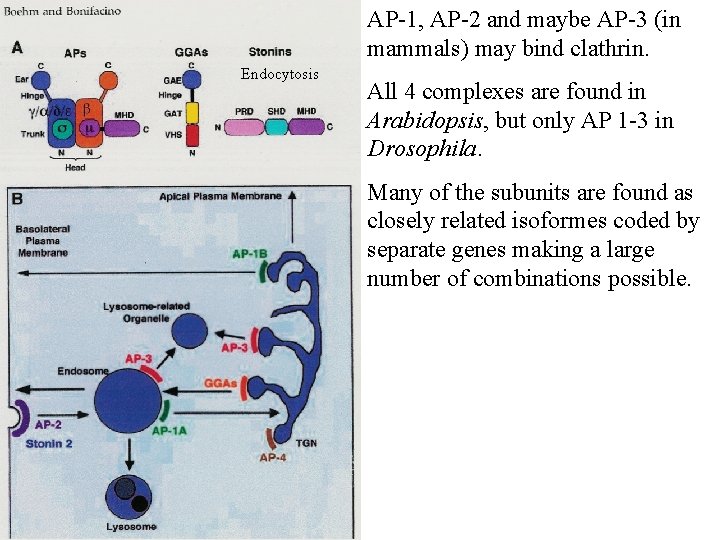
AP-1, AP-2 and maybe AP-3 (in mammals) may bind clathrin. Endocytosis All 4 complexes are found in Arabidopsis, but only AP 1 -3 in Drosophila. Many of the subunits are found as closely related isoformes coded by separate genes making a large number of combinations possible.
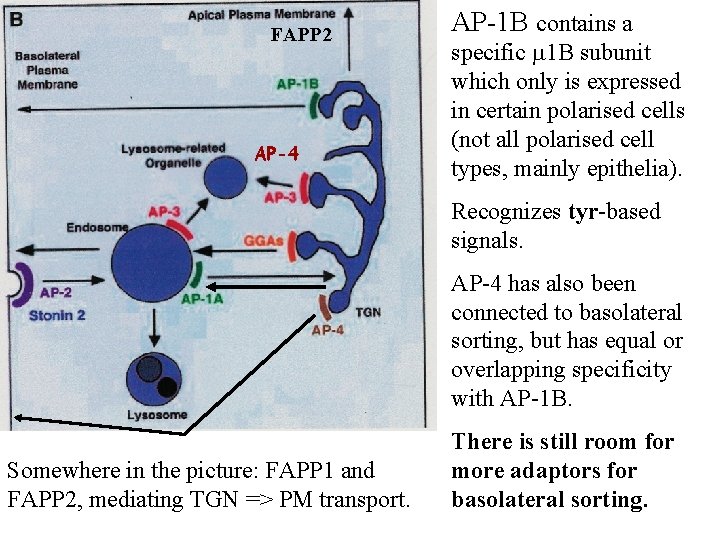
FAPP 2 AP-4 AP-1 B contains a specific 1 B subunit which only is expressed in certain polarised cells (not all polarised cell types, mainly epithelia). Recognizes tyr-based signals. AP-4 has also been connected to basolateral sorting, but has equal or overlapping specificity with AP-1 B. Somewhere in the picture: FAPP 1 and FAPP 2, mediating TGN => PM transport. There is still room for more adaptors for basolateral sorting.
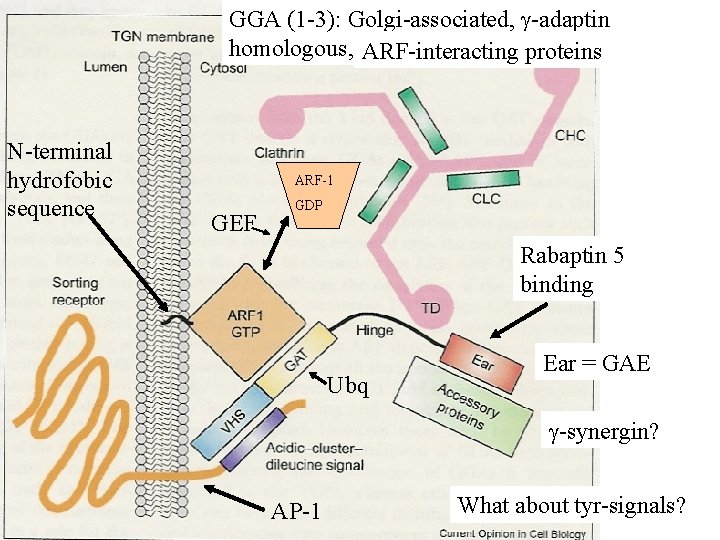
GGA (1 -3): Golgi-associated, -adaptin homologous, ARF-interacting proteins N-terminal hydrofobic sequence ARF-1 GEF GDP Rabaptin 5 binding Ubq Ear = GAE -synergin? AP-1 What about tyr-signals?
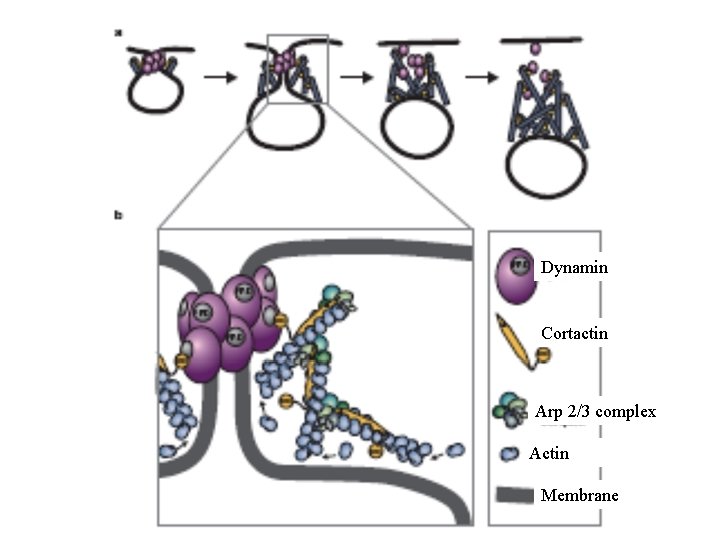
Dynamin Cortactin Arp 2/3 complex Actin Membrane

Aktiv membranbundet At least 6 ARFs exist in mammals, 5 are localised to the Golgi-apparatus and 1 to the plasma membrane. ARF 1 -GTP (myristoylated), a tyrosine based signal, and cytoplasmatisk phosfatidylinositol 4, 5 bisphosphate are necessary to recruit AP-1 clathrin 4 families adaptors to membranes. GEFs with Phosphatidylinositides of the 4 -series has several been regarded as important for Golgi. members

GGA dependent receptors CI-Mannose-6 -phosphate receptor CD-Mannose-6 -phosphate receptor Sortilin Sor. LA/LR 11 LRP-3 -secretase
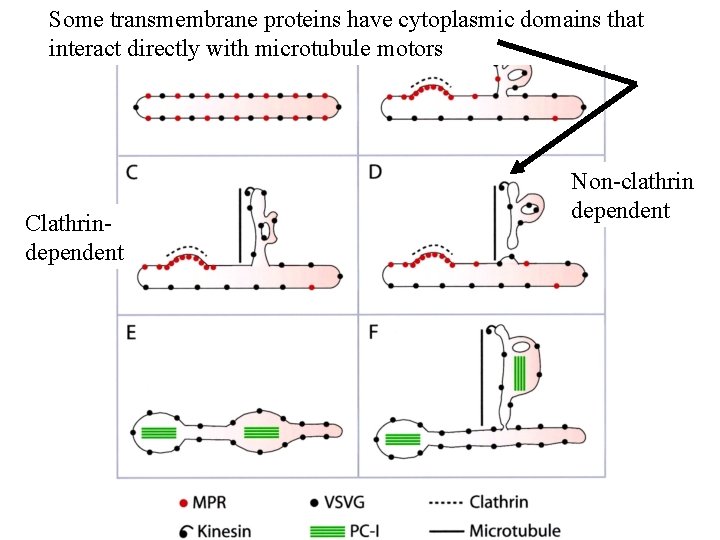
Some transmembrane proteins have cytoplasmic domains that interact directly with microtubule motors Clathrindependent Non-clathrin dependent

What about APICAL SORTING IN EPITHELIAL CELLS? Glycans: N-glycans, O-glycans, CS glycosaminoglycans Yes (maybe and no), yes (maybe), yes. GPI-anchors? NOT REALLY – OR? LIPID DOMAINS? ? Protein motifs for apical sorting: Megalin NPXY. The second of three NPXY motifs is crucial (distance from membrane…).
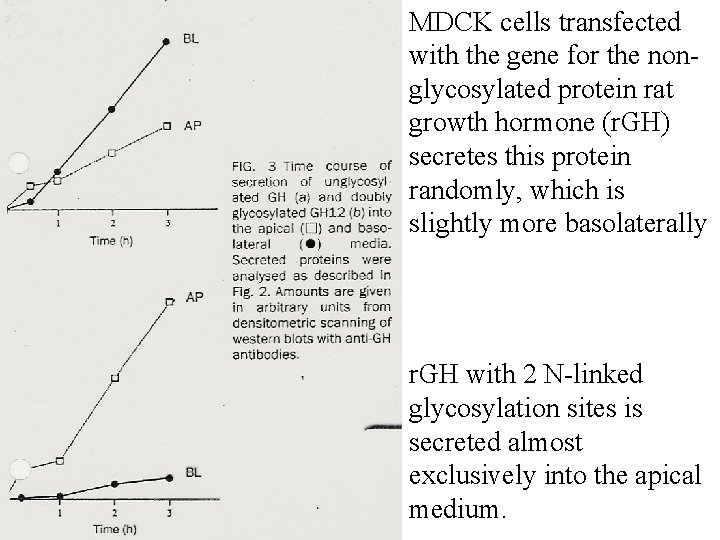
MDCK cells transfected with the gene for the nonglycosylated protein rat growth hormone (r. GH) secretes this protein randomly, which is slightly more basolaterally r. GH with 2 N-linked glycosylation sites is secreted almost exclusively into the apical medium.

Erythropoietin – three N-glycans, one is critical. Endolyn – eight N-glycans, not all equally important. O-glycans of mucin type may also mediate apical sorting: Intestinal sucrase-isomaltase Gp-40 Several other examples But several examples of non-sorted glycoproteins also exist.
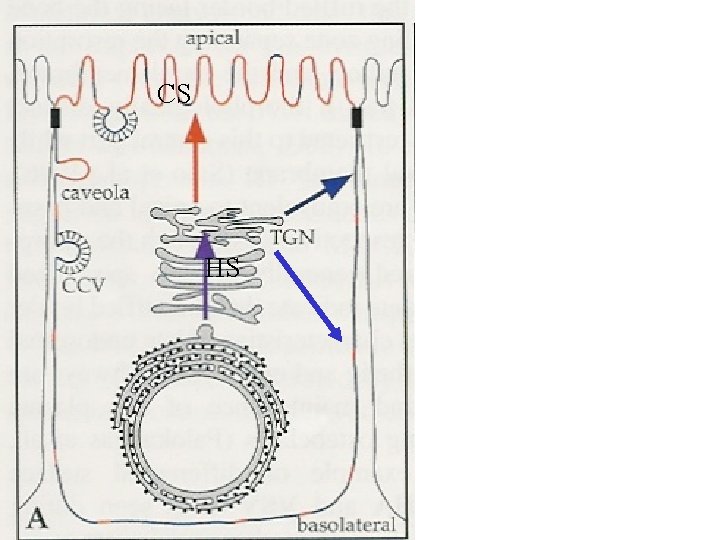
CS HS
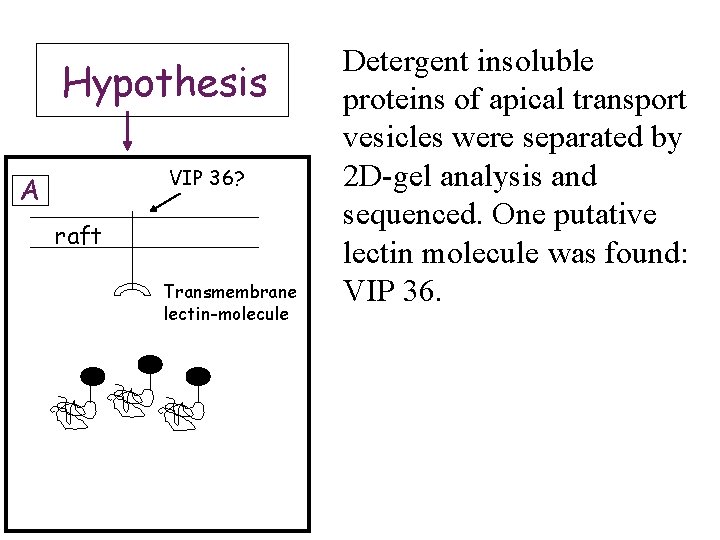
Hypothesis VIP 36? A raft Transmembrane lectin-molecule Detergent insoluble proteins of apical transport vesicles were separated by 2 D-gel analysis and sequenced. One putative lectin molecule was found: VIP 36.

WHAT IS A (GLYCOLIPID) RAFT? Glycolipid- and cholesterol rich domains in a lipid membrane are associated in a more stable structure than lipids are according to “the fluid mosaic model. ” On the cell surface of a “regular” cell, these domains will have a diameter of 60 - 100 nm. In specialised membranes may larger areas of the plasma membrane have raft-characteristics. Example: The apical membrane of epithelial cells (MDCK). Do lipids and lipid-binding proteins play a role in sorting of molecules that are transported from the TGN to the apical membrane?

Caveolins: Proteins with affinity for specialised lipiddomains. Palmitoylation. Might be necessary for cell surface transport of GPI-anchored proteins. Not apical transport. GPI-anker: Glycosyl-phosphatidyl-inositol-anchor that might bind proteins to a membrane. For some time regarded as sorting signals for apical transport, since these proteins usually are localised to rafts. The apical sorting is most likely dependent on N-glycans (via transcytosis? ). Glycosphingolipids: are glycolipids that are mainly transported to the apical side in MDCK-cells (from the TGN). Present in rafts rich in cholesterol. MAL (VIP 17): A protein that seems to mediate apical sorting of several cargo proteins in MDCK-cells.
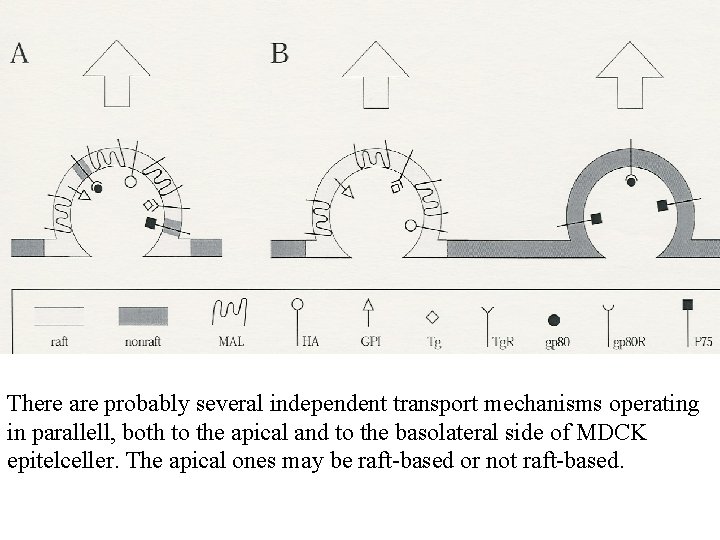
There are probably several independent transport mechanisms operating in parallell, both to the apical and to the basolateral side of MDCK epitelceller. The apical ones may be raft-based or not raft-based.
Protein kinase D Annexin II, Annexin XIIIb, FAPP 1/2, MAL (VIP 17)

All mechanisms for sorting from the TGN are not known *We have only discussed proteins with one transmembrane domain, while many proteins span the membrane several times. These may also be sorted. How? *Some apical proteins, like megalin, have been reported to have signals in the cytoplasmic tail (interaction with motor proteins? ) *Ubiquitinylation may shift the sorting from TGN to the plasma membrane towards TGN to lysosomes (the vacuole in yeast). *Lipids may play a role in sorting in many ways. *What factors are necessary for budding, transport and fusion? (v- and t- SNARES, GTP-binding proteins, etc. )
- Slides: 39