Poster at AANP 2020 Background In clinical trials

Poster at AANP 2020

Background • In clinical trials for GBM, patient selection based on MGMT promoter methylation tests has limitations: • MGMT promoter methylation tests do not account for alternative mechanisms of gene regulation. • The technical heterogeneity and the costs for methylation tests are highly variable. • Some methylation tests demand central evaluation in larger institutions, which increases the turn-around-time. • We previously described that high MGMT expression in tumor cells is an independent prognostic factor for glioblastomas. • We hypothesized that IHC is a cost-effective and reliable prognostic/predictive tool to assess MGMT expression in glioblastomas.

Methods Our aim is to develop a CLIA-certified IHC test as a prognostic/predictive tool for glioblastomas. • MGMT quantification performed by Western blot (WB) in GBM primary cell lines with known MGMT promoter methylation status (GBM 10, GBM 14, and GBM 43) - anti-MGMT antibody (clone MT 3. 1 – Millipore). • The specificity of the 5 monoclonal antibodies was tested by WB using an input of 25µg of protein extracted from GBM 43 cell line. • IHC optimization: 10 cases predicted to be positive and 10 cases predicted to be negative for MGMT expression, based on MGMT methylation pyrosequencing were stained with the same anti-MGMT antibody (protocol for prognostic tests). • 195 glioblastomas arranged in tissue microarrays (TMAs) were stained with the best anti. MGMT antibody and independently analyzed by two experienced neuropathologists, to assess reproducibility of IHC results.

MGMT protein expression determined different survival in cell lines with similar MGMT promoter status, and the anti-MGMT m. Ab showed high specificity compared to other antibodies from different makers. OS in response to TMZ plus radiation: GBM 10: 76 days GBM 14: 188 days GBM 43: 55 days (Carlson, Grogan et al. 2009 – Curr Protoc Pharmacol) MT 3. 1 The antibody used for the IHC optimization is shown as Ab 5. Control GAPDH – data not shown.
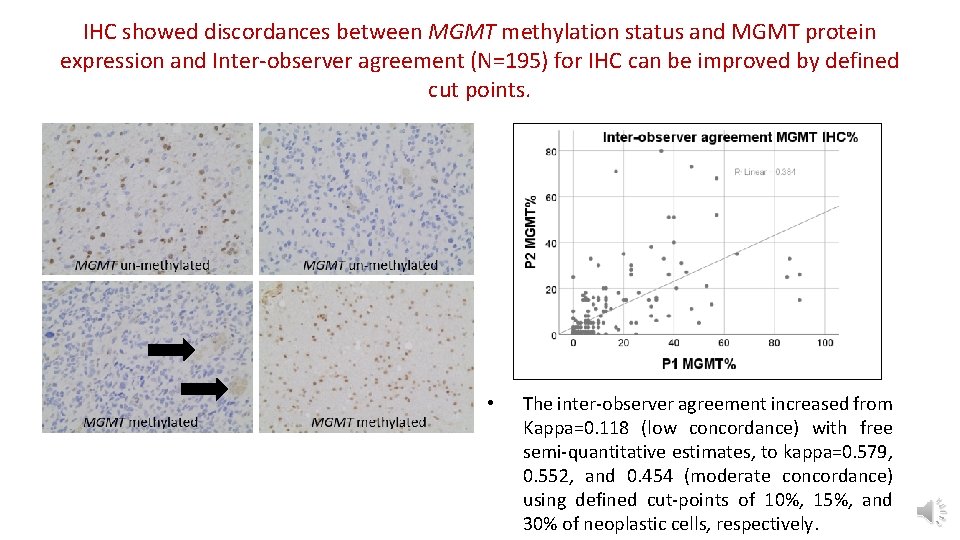
IHC showed discordances between MGMT methylation status and MGMT protein expression and Inter-observer agreement (N=195) for IHC can be improved by defined cut points. • The inter-observer agreement increased from Kappa=0. 118 (low concordance) with free semi-quantitative estimates, to kappa=0. 579, 0. 552, and 0. 454 (moderate concordance) using defined cut-points of 10%, 15%, and 30% of neoplastic cells, respectively.

Summary • IHC test for MGMT assessment in GBM is feasible and the inter-observer agreement improves with well-defined cut-points. • IHC may reduce the need for centralized tests (such as EPIC methylation array), therefore lowering costs, while strengthening the active participation of local Pathologists in decision-making for clinical trials in glioblastomas. • We are currently developing strategies to define and validate optimal cut points to establish MGMT IHC as an integral predictive marker for stratification/enrollment criteria for clinical trials.
- Slides: 6