POSITIVE CONTROL SAMPLES FOR PARENTERAL PRODUCT CLASSES ASTM
















- Slides: 21

POSITIVE CONTROL SAMPLES FOR PARENTERAL PRODUCT CLASSES ASTM F 02 Committee on Barrier Packaging

CHALLENGE • Various methods for positive controls • Dependent on package contents ØCompressible fluid (gas) ØNon-Compressible fluid (liquids) ØVarious liquid characteristics • “Proteinaceous product may interfere with defect detection. ” 1 1 Li, Lei. American Pharmaceutical Review. February 20, 2013. Container Development and Validation for Prefilled Syringes. Closure Integrity Testing Method

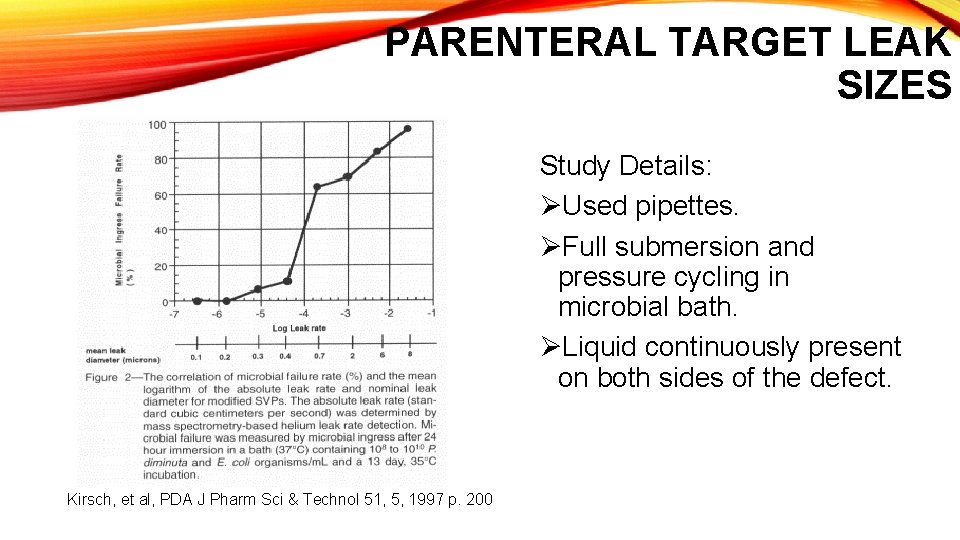
PARENTERAL TARGET LEAK SIZES Study Details: ØUsed pipettes. ØFull submersion and pressure cycling in microbial bath. ØLiquid continuously present on both sides of the defect. Kirsch, et al, PDA J Pharm Sci & Technol 51, 5, 1997 p. 200

POSITIVE CONTROL TYPES • Mechanical (drilling, puncture, crack formation, etc. ) • Pipette (“Sharp Edge”) • Capillary • Laser Drilled Defect • Controlled Air Flow Natural Vial Defect

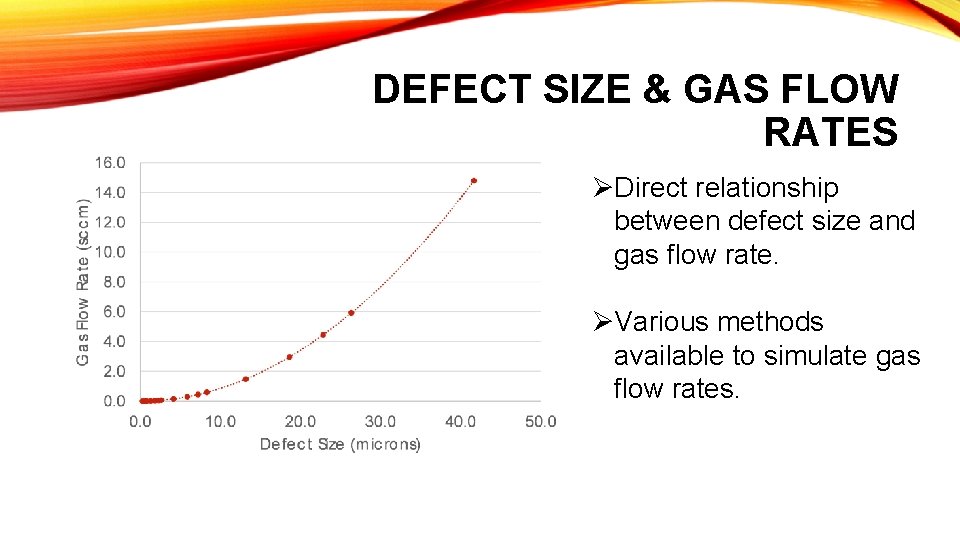
DEFECT SIZE & GAS FLOW RATES ØDirect relationship between defect size and gas flow rate. ØVarious methods available to simulate gas flow rates.

LASER DRILLED DEFECTS Nominal hole size 5 µm

CAPILLARY (TUBE) DEFECTS Accuracy of a 20 micron ID capillary; Avg. = 20. 1 μm, σ = 0. 54 μm.

CAPILLARY FLOW RATE 30µ Pinhole – ~3. 26 sccm 30µ 20 mm Channel – ~0. 16 sccm 30µ 40 mm Channel – ~0. 08 sccm

PIPETTE GAS FLOW • Pipette tip formed to create specific diameter defect. • Certified with flowrate. • Sharp defect geometry. Target defect diameter at tip.

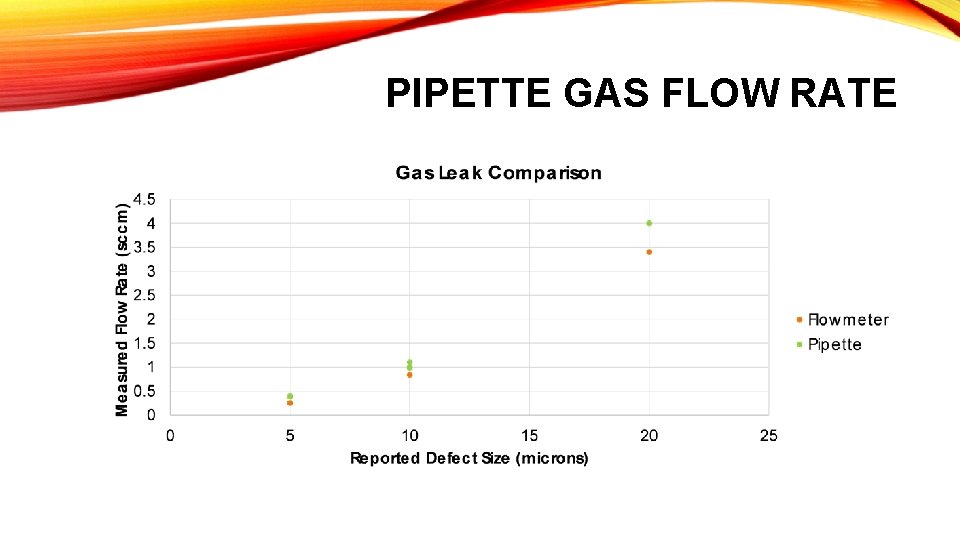
PIPETTE GAS FLOW RATE

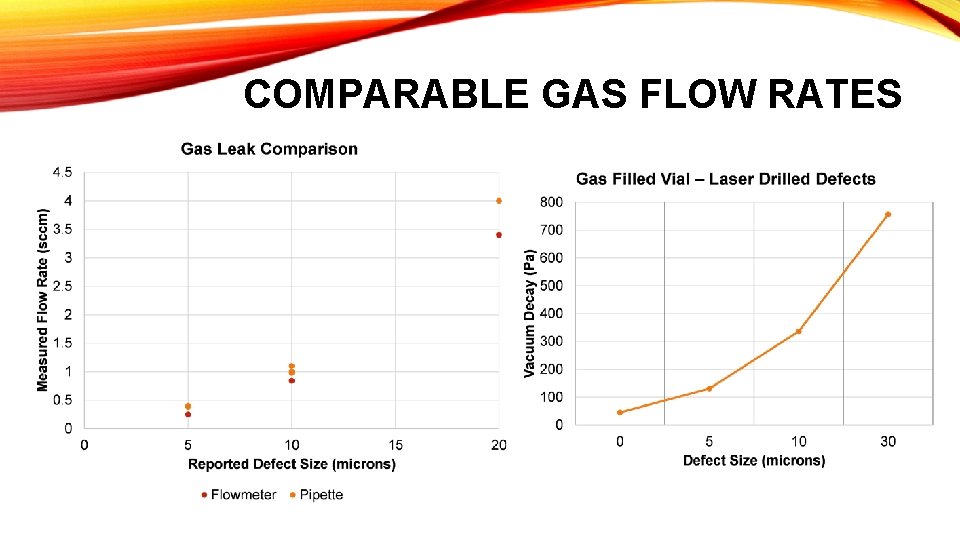
COMPARABLE GAS FLOW RATES

PIPETTE MENISCUS Significant liquid surface area within pipette.

CAPILLARY ACTION Factors of Capillary Action Ø liquid-air surface tension(force/unit length), Ø θ is the contact angle, Ø ρ is the density of liquid Ø g is local acceleration due to gravity Ø r is radius of tube Thus the more narrow the space in which the water can travel, the further up it goes. Variables for Water: γ = 0. 0728 N/m at 20 °C, θ = 0° (cos(0) = 1), ρ is 1000 kg/m 3, and g = 9. 81 m/s 2.

PIPETTE MENISCUS 10 micron – 0. 48 mm meniscus diameter 5 micron – 0. 26 mm meniscus diameter

PIPETTE SUBMERSION 5 Micron - 15 Seconds Submersion 5 Micron - 1 Minute Submersion 5 Micron - 10 Minutes Submersion 10 Micron - 15 Seconds Submersion Damaged Pipette Tip – 5 seconds

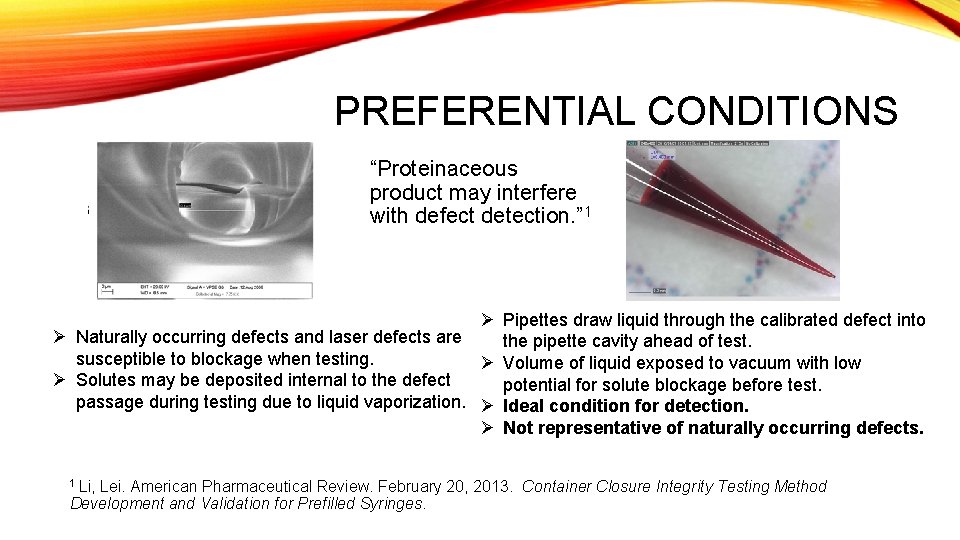
PREFERENTIAL CONDITIONS “Proteinaceous product may interfere with defect detection. ” 1 Ø Pipettes draw liquid through the calibrated defect into Ø Naturally occurring defects and laser defects are the pipette cavity ahead of test. susceptible to blockage when testing. Ø Volume of liquid exposed to vacuum with low Ø Solutes may be deposited internal to the defect potential for solute blockage before test. passage during testing due to liquid vaporization. Ø Ideal condition for detection. Ø Not representative of naturally occurring defects. 1 Li, Lei. American Pharmaceutical Review. February 20, 2013. Container Development and Validation for Prefilled Syringes. Closure Integrity Testing Method

PIPETTE FLOW RATE COMPARISON

CAPILLARY ACTION AND DEEP VACUUM 1. 0 mm 3 of water exposed to the test chamber before any vacuum force, the liquid vaporization rate occurs would cause significant vacuum decay at deep vacuum regardless of the tip ID.

CAPILLARY LIQUID FLOW RATE Ø Liquid defects will similarly affect capillaries. Ø No sharp edge to simulate natural defect geometry. Ø The meniscus will produce a liquid surface area the same area of the internal geometry of the capillary.

LASER DRILLED DEFECTS Nominal hole size 5 µm “Better resembles natural defects in glass (cracks) and polymers (pinholes). ” 1 1 Li, Lei. American Pharmaceutical Review. February 20, 2013. Container Development and Validation for Prefilled Syringes. Closure Integrity Testing Method

CONCLUSION • A variety of positive control types provide accurate a reliable methods for creating defects for gas filled defects. • Micropipettes and capillaries acceptable for gas flow (dry product) leak testing. • Use of capillary and pipette defects will provide a unique circumstance, improving the detection capability of liquid defects with deep vacuum based methods.