POPULATION PHARMACOKINETICS RAYMOND MILLER D Sc Pfizer Global

POPULATION PHARMACOKINETICS RAYMOND MILLER, D. Sc. Pfizer Global Research and Development

Population Pharmacokinetics Definition Advantages/Disadvantages Objectives of Population Analyses Impact in Drug Development

Definition Population pharmacokinetics describe The typical relationships between physiology (both normal and disease altered) and pharmacokinetics, The interindividual variability in these relationships, and Their residual intraindividual variability. Sheiner-LB Drug-Metab-Rev. 1984; 15(1 -2): 153 -71
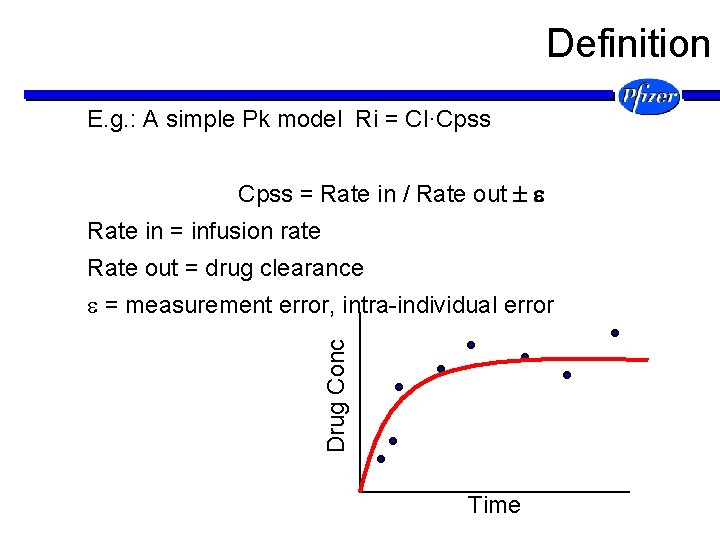
Definition E. g. : A simple Pk model Ri = Cl·Cpss = Rate in / Rate out Rate in = infusion rate Rate out = drug clearance Drug Conc = measurement error, intra-individual error Time
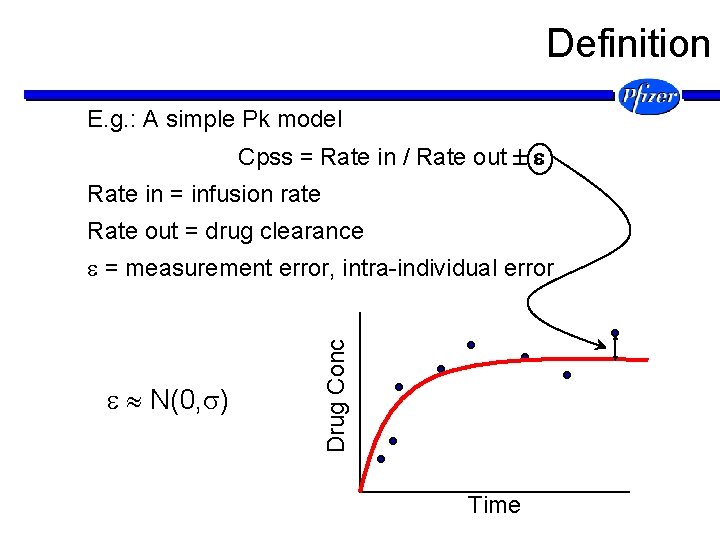
Definition E. g. : A simple Pk model Cpss = Rate in / Rate out Rate in = infusion rate Rate out = drug clearance N(0, ) Drug Conc = measurement error, intra-individual error Time
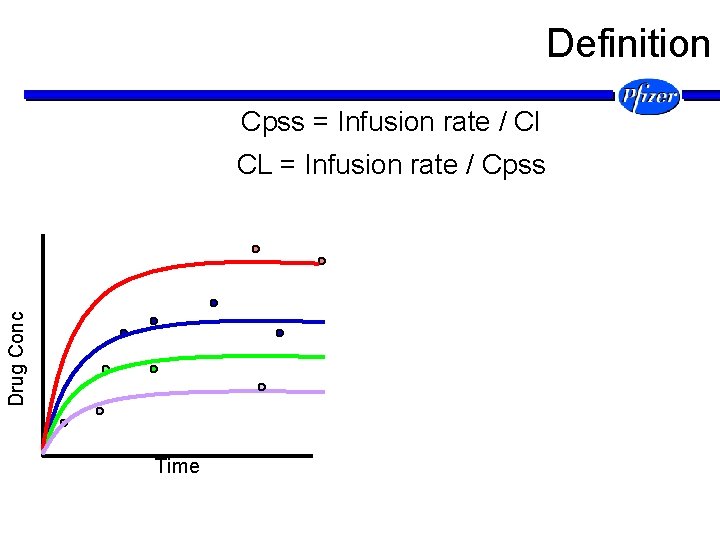
Definition Cpss = Infusion rate / Cl Drug Conc CL = Infusion rate / Cpss Time
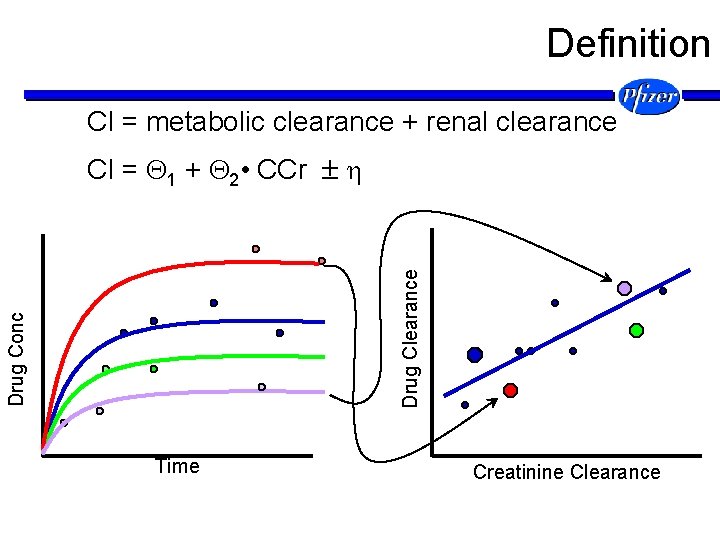
Definition Cl = metabolic clearance + renal clearance Drug Conc Drug Clearance Cl = 1 + 2 • CCr Time Creatinine Clearance
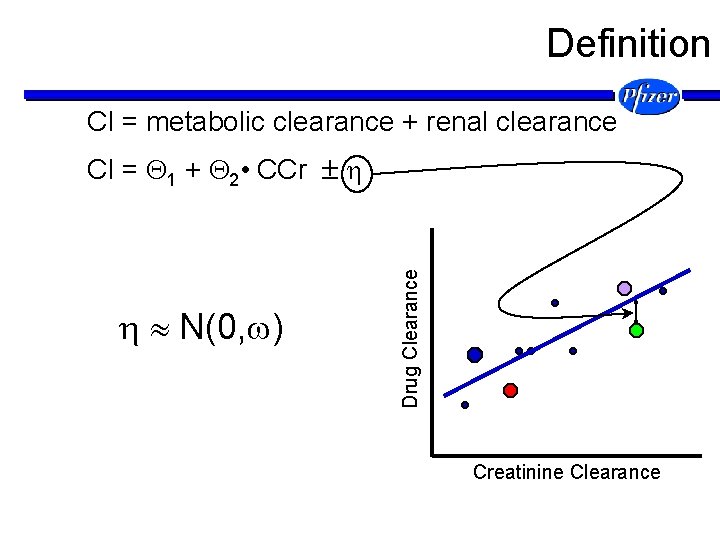
Definition Cl = metabolic clearance + renal clearance N(0, ) Drug Clearance Cl = 1 + 2 • CCr Creatinine Clearance

Graphical illustration of the statistical model used in NONMEM for the special case of a one compartment model with first order absorption. (Vozeh et al. Eur J Clin Pharmacol 1982; 23: 445 -451)
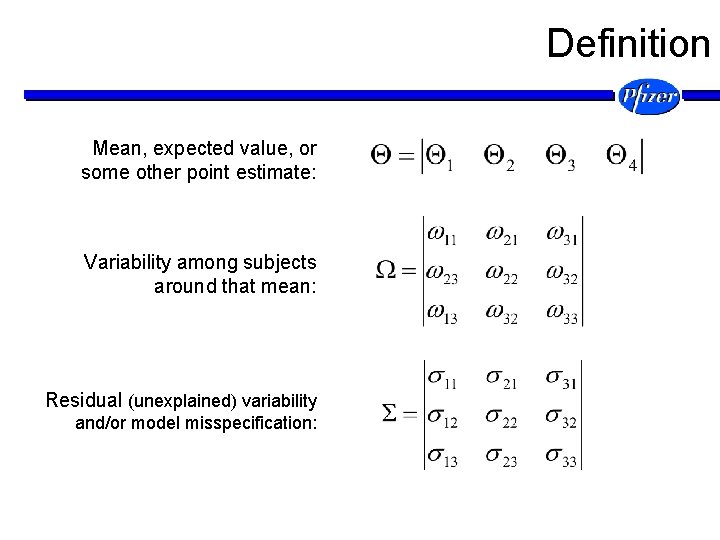
Definition Mean, expected value, or some other point estimate: Variability among subjects around that mean: Residual (unexplained) variability and/or model misspecification:
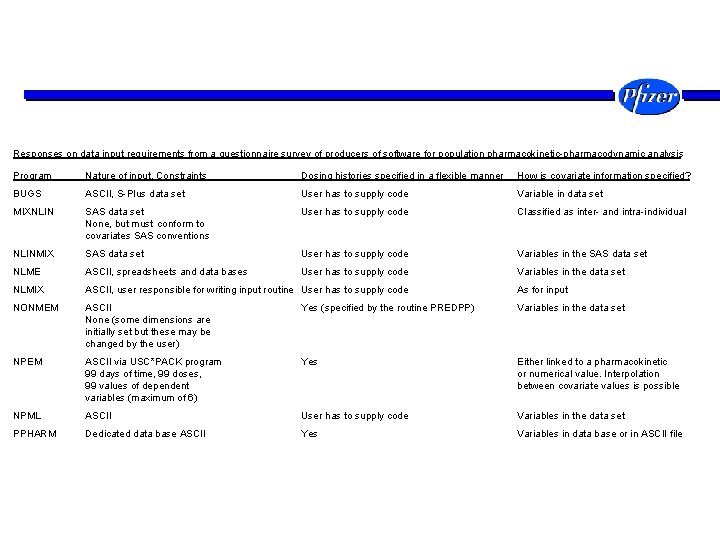
Responses on data input requirements from a questionnaire survey of producers of software for population pharmacokinetic-pharmacodynamic analysis Program Nature of input, Constraints Dosing histories specified in a flexible manner How is covariate information specified? BUGS ASCII, S-Plus data set User has to supply code Variable in data set MIXNLIN SAS data set None, but must conform to covariates SAS conventions User has to supply code Classified as inter- and intra-individual NLINMIX SAS data set User has to supply code Variables in the SAS data set NLME ASCII, spreadsheets and data bases User has to supply code Variables in the data set NLMIX ASCII, user responsible for writing input routine User has to supply code As for input NONMEM ASCII None (some dimensions are initially set but these may be changed by the user) Yes (specified by the routine PREDPP) Variables in the data set NPEM ASCII via USC*PACK program 99 days of time, 99 doses, 99 values of dependent variables (maximum of 6) Yes Either linked to a pharmacokinetic or numerical value. Interpolation between covariate values is possible NPML ASCII User has to supply code Variables in the data set PPHARM Dedicated data base ASCII Yes Variables in data base or in ASCII file

Objectives 1. Provide Estimates of Population PK Parameters (CL, V) - Fixed Effects 2. Provide Estimates of Variability - Random Effects • Intersubject Variability • Interoccasion Variability (Day to Day Variability) • Residual Variability (Intrasubject Variability, Measurement Error, Model Misspecification)

Objectives 3. Identify Factors that are Important Determinants of Intersubject Variability • Demographic: Age, Body Weight or Surface Area, gender, race • Genetic: CYP 2 D 6, CYP 2 C 19 • Environmental: Smoking, Diet • Physiological/Pathophysiological: Renal (Creatinine Clearance) or Hepatic impairment, Disease State • Concomitant Drugs • Other Factors: Meals, Circadian Variation, Formulations

Advantages • Sparse Sampling Strategy (2 -3 concentrations/subject) –Routine Sampling in Phase II/III Studies –Special Populations (Pediatrics, Elderly) • Large Number of Patients –Fewer restrictions on inclusion/exclusion criteria • Unbalanced Design –Different number of samples/subject • Target Patient Population –Representative of the Population to be Treated

Disadvantages • Quality Control of Data –Dose and Sample Times/Sample Handling/ Inexperienced Clinical Staff • Timing of Analytical Results/Data Analyses • Complex Methodology –Optimal Study Design (Simulations) –Data Analysis • Resource Allocation • Unclear Cost/Benefit Ratio
Drug Conc Models are critical in sparse sampling situations: Time
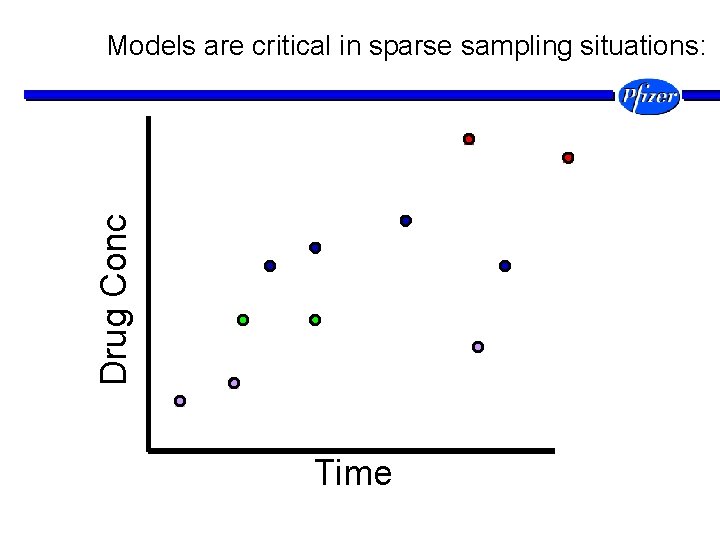
Drug Conc Models are critical in sparse sampling situations: Time
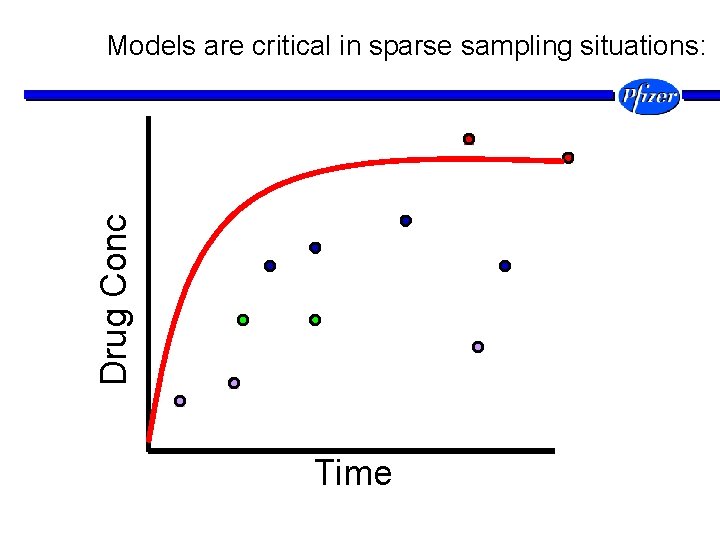
Drug Conc Models are critical in sparse sampling situations: Time
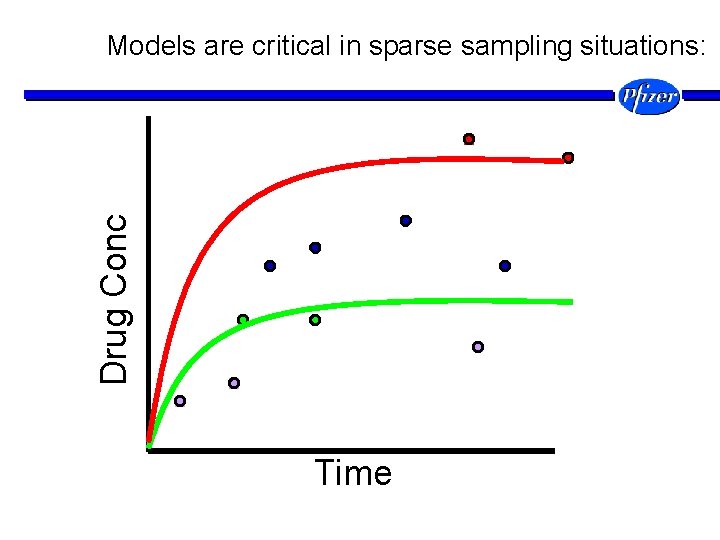
Drug Conc Models are critical in sparse sampling situations: Time
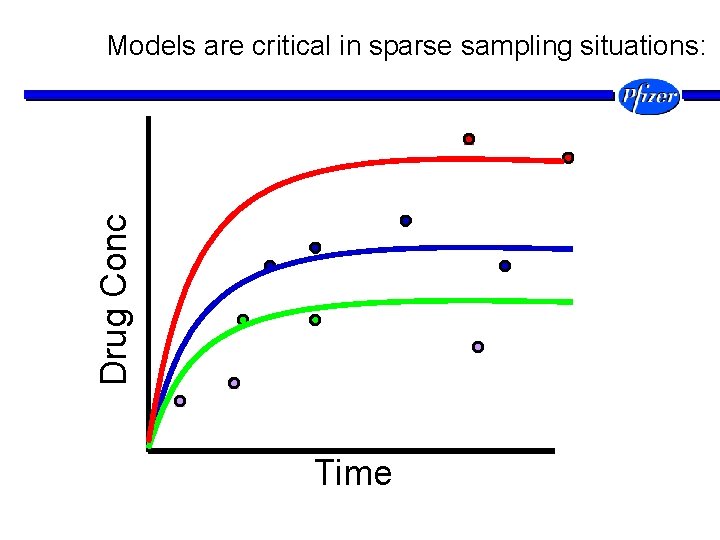
Drug Conc Models are critical in sparse sampling situations: Time
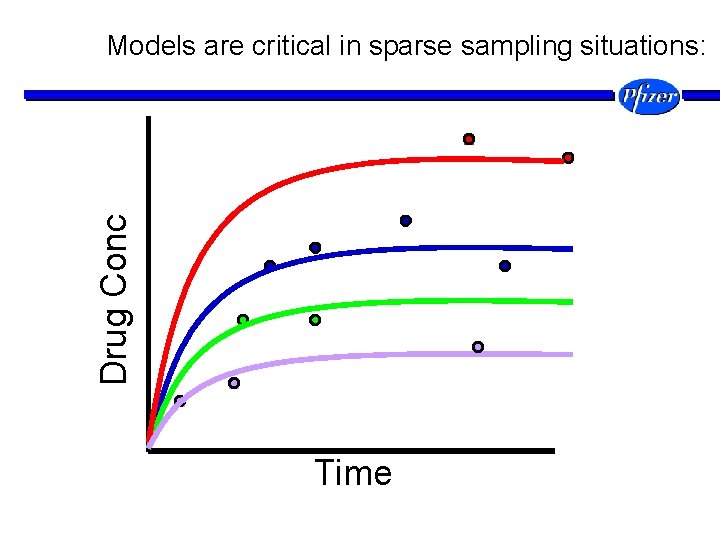
Drug Conc Models are critical in sparse sampling situations: Time

Study Objectives w To evaluate the efficacy of drug treatment or placebo as add on treatment in patients with partial seizures.
Data Structure

Baseline Placebo

Count Model represents the expected number of events per unit time E(Yij)= itij The natural estimator of is the overall observed rate for the group.

Suppose there are typically 5 occurrences per month in a group of patients: - =5

The mean number of seizure episodes per month (λ) was modeled using NONMEM as a function of drug dose, placebo, baseline and subject specific random effects. Baseline = estimated number of seizures reported during baseline period Placebo = function describing placebo response Drug = function describing the drug effect = random effect

Sub-population analysis w Some patients are refractory to any particular drug at any dose. w Interest is in dose-response in patients that respond w Useful in adjusting dose in patients who would benefit from treatment w Investigate the possibility of at least two subpopulations.

Mixture Model A model that implicitly assumes that some fraction p of the population has one set of typical values of response, and that the remaining fraction 1 -p has another set of typical values Population A (p) Population B (1 -p)

Final Model



Expected percent reduction in seizure frequency w Monte Carlo simulation using parameters and variance for Subgroup A w 8852 individuals (51% female) w % reduction from baseline seizure frequency calculated w Percentiles calculated for % reduction in seizure frequency at each dose


Results

Conclusions w A comparison of the dose-response relationship for gabapentin and pregabalin reveals that pregabalin was 2. 5 times more potent, as measured by the dose that reduced seizure frequency by 50% (ED 50). w Pregabalin was more effective than gabapentin based on the magnitude of the reduction in seizure frequency (Emax) w Three hundred clinical trials for each drug were simulated conditioned on the original study designs. Each simulated trial was analyzed to estimate % median change in seizure frequency. The observed and model-predicted treatment effects of median reduction in seizure frequency for gabapentin and pregabalin are illustrated for all subjects and for responders. Data points represent median percentage change from baseline in seizure frequency for each treatment group (including placebo). The shaded area corresponds to predicted 10 th and 90 th percentiles for median change from baseline in seizure frequency.

Relationship Between %Change in Seizure Frequency (Relative to Baseline) and Daily Dosage of Gabapentin and Pregabalin • Dose-response model in epilepsy using pooled analysis of 4 gabapentin studies + 3 pregabalin studies

Relationship Between %Change in Seizure Frequency (Relative to Baseline) and Daily Dosage of Gabapentin and Pregabalin in Responders to Treatment • Dose-response model in epilepsy using pooled analysis of 4 gabapentin studies + 3 pregabalin studies

Impact in Drug Development w Gabapentin was subsequently approved by FDA for post-herpetic neuralgia w Approved label states under clinical studies: “Pharmacokineticpharmacodynamic modeling provided confirmatory evidence of efficacy across all doses”
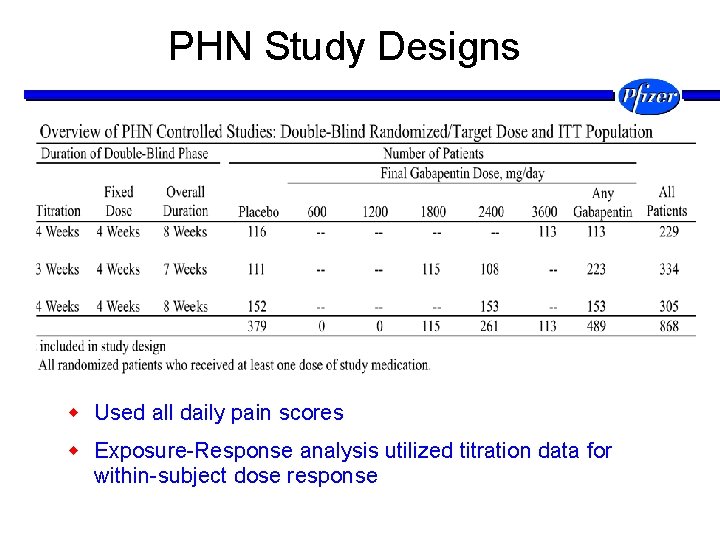
PHN Study Designs w Used all daily pain scores w Exposure-Response analysis utilized titration data for within-subject dose response
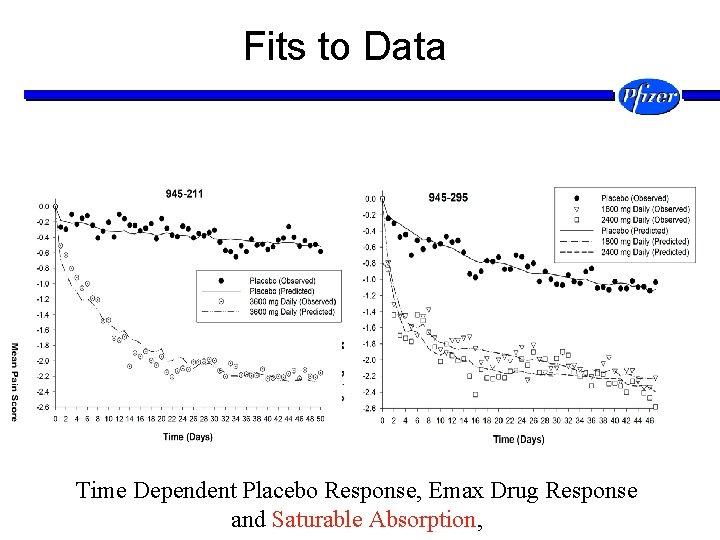
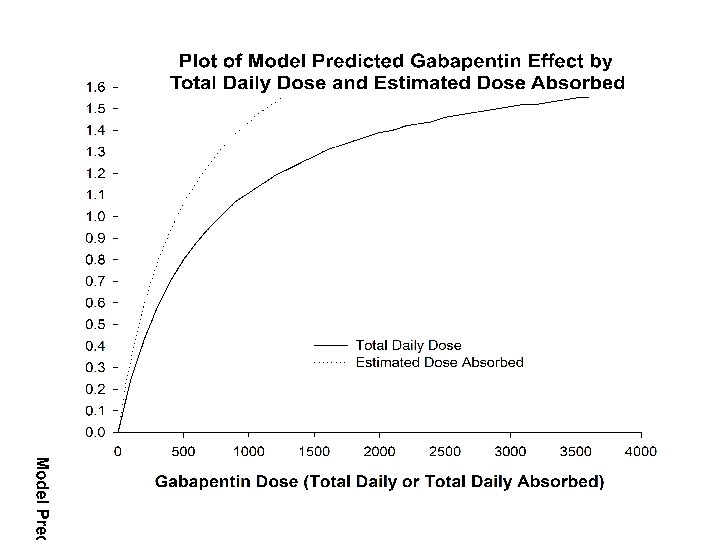
Fits to Data Time Dependent Placebo Response, Emax Drug Response and Saturable Absorption,

Outcomes w Model and Data Provided with Submission • FDA reviewers used model to test various scenarios • Supported doses and conclusions of Pfizer • Provided confidence to eliminate need for replicate doses • FDA proposed language in the label on PK-PD modeling and clinical trials w FDA/Pfizer publication to discuss modeling and impact on regulatory decision-making • clinical endpoints • similar study design • familiarity with drug class
- Slides: 43