POLYSACCHARIDE STRUCTURE References Tombs M P Harding S
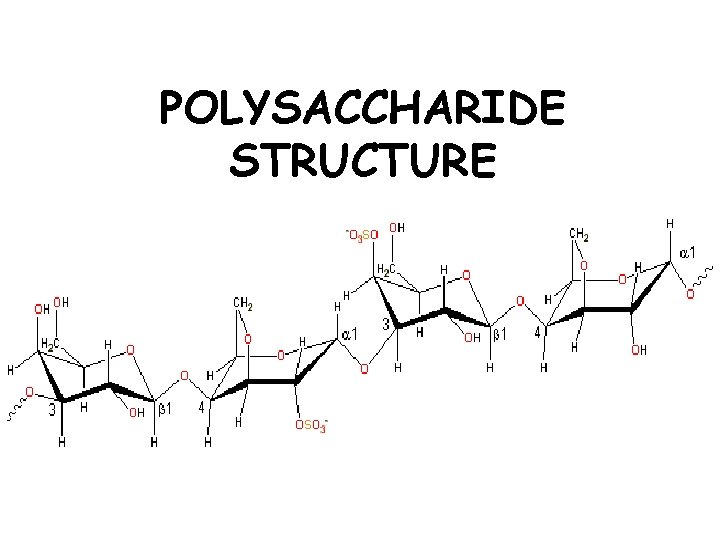
POLYSACCHARIDE STRUCTURE



References • Tombs, M. P. & Harding, S. E. , An Introduction to Polysaccharide Biotechnology, Taylor & Francis, London, 1997 • D. A. Rees, Polysaccharide Shapes, Chapman & Hall, 1977 • E. R. Morris in ‘Polysaccharides in Food’, J. M. V. Blanshard & J. R. Mitchell (eds. ), Butterworths, London. 1979, Chapter 2 • The Polysaccharides, G. O. Aspinall (ed. ), Academic Press, London, 1985 • Carbohydrate Chemistry for Food Scientists, R. L. Whistler, J. N. Be. Miller, Eagan Press, St. Paul, USA, 1997

Proteins: • well defined • Coded precisely by genes, hence monodisperse • ~20 building block residues (amino acids) • Standard peptide link (apart from proline) • Normally tightly folded structures • {some proteins do not possess folded structure – gelatin – an “honorary polysaccharide”}
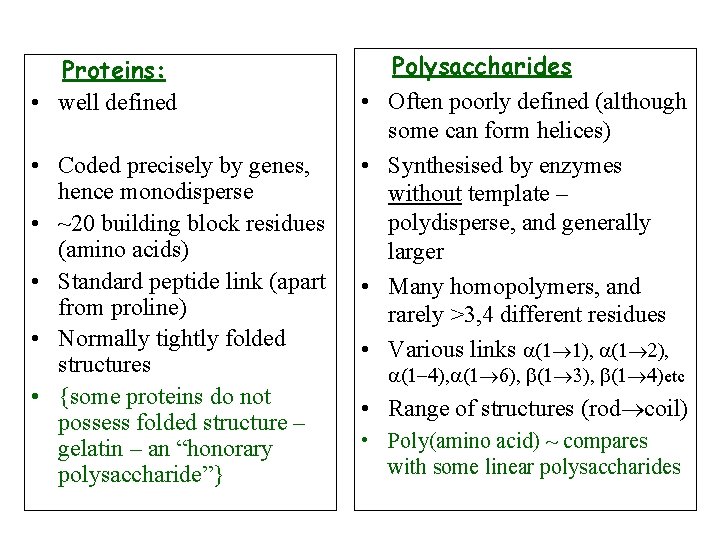
Proteins: • well defined • Coded precisely by genes, hence monodisperse • ~20 building block residues (amino acids) • Standard peptide link (apart from proline) • Normally tightly folded structures • {some proteins do not possess folded structure – gelatin – an “honorary polysaccharide”} • • Polysaccharides Often poorly defined (although some can form helices) Synthesised by enzymes without template – polydisperse, and generally larger Many homopolymers, and rarely >3, 4 different residues Various links a(1 1), a(1 2), a(1 -4), a(1 6), b(1 3), b(1 4)etc • Range of structures (rod coil) • Poly(amino acid) ~ compares with some linear polysaccharides

Monosaccharides • Contain between 3 and 7 C atoms • empirical formula of simple monosaccharides (CH 2 O)n • aldehydes or ketones from http: //ntri. tamuk. edu/cell/carbohydrates. html

Some. Terminology • Asymmetric (Chiral) Carbon – has covalent bonds to four different groups, cannot be superimposed on its mirror image • Enantiomers - pair of isomers that are (non -superimposable) mirror images

Chirality rules 1. Monosaccharides contain one or more asymmetric Catoms: get D- and L-forms, where D- and L- designate absolute configuration 2. D-form: -OH group is attached to the right of the asymmetric carbon 3. L-form: -OH group is attached to the left of the asymmetric carbon 4. If there is more than one chiral C-atom: absolute configuration of chiral C furthest away from carbonyl group determines whether D- or L-
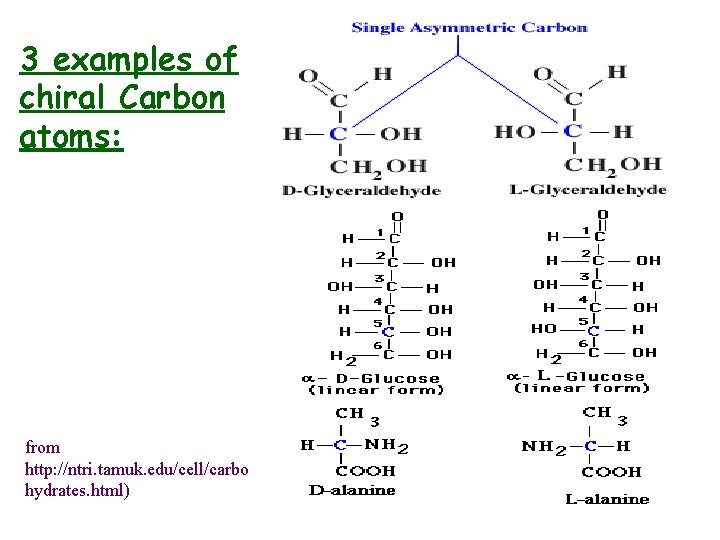
3 examples of chiral Carbon atoms: from http: //ntri. tamuk. edu/cell/carbo hydrates. html)
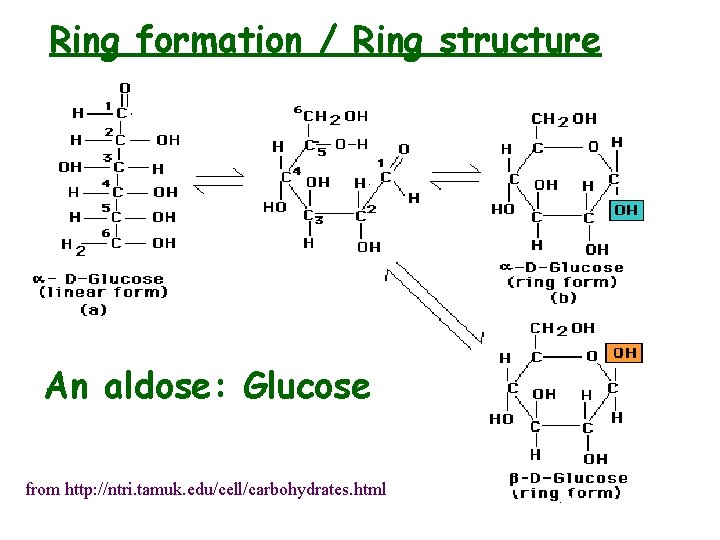
Ring formation / Ring structure An aldose: Glucose from http: //ntri. tamuk. edu/cell/carbohydrates. html
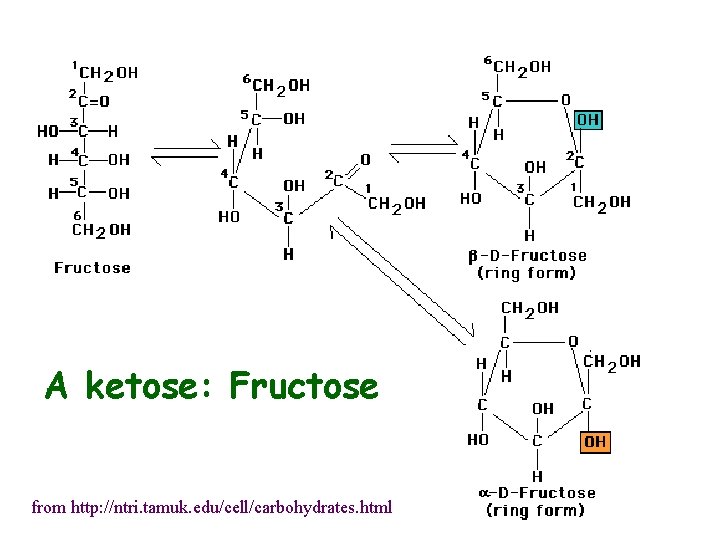
A ketose: Fructose from http: //ntri. tamuk. edu/cell/carbohydrates. html

Ring Structure • Linear known as “Fischer” structure” • Ring know as a “Haworth projection” • Cyclization via intramolecular hemiacetal (hemiketal) formation • C-1 becomes chiral upon cyclization - anomeric carbon • Anomeric C contains -OH group which may be a or b (mutarotation a b) • Chair conformation usual (as opposed to boat) • Axial and equatorial bonds
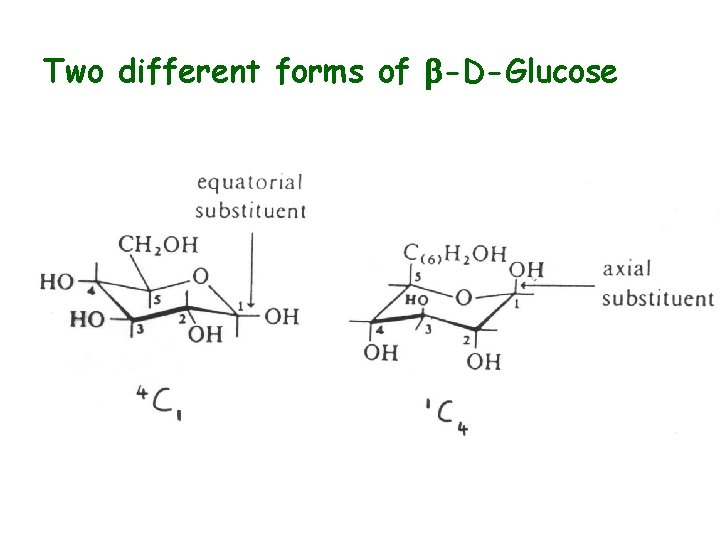
Two different forms of b-D-Glucose
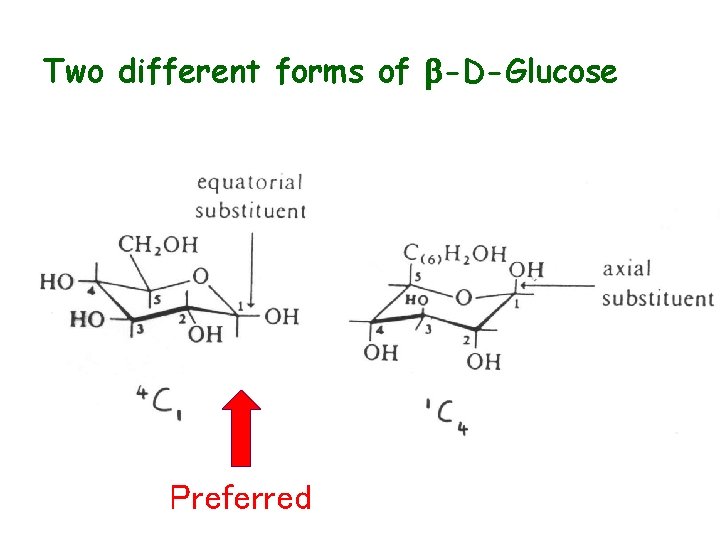
Two different forms of b-D-Glucose Preferred
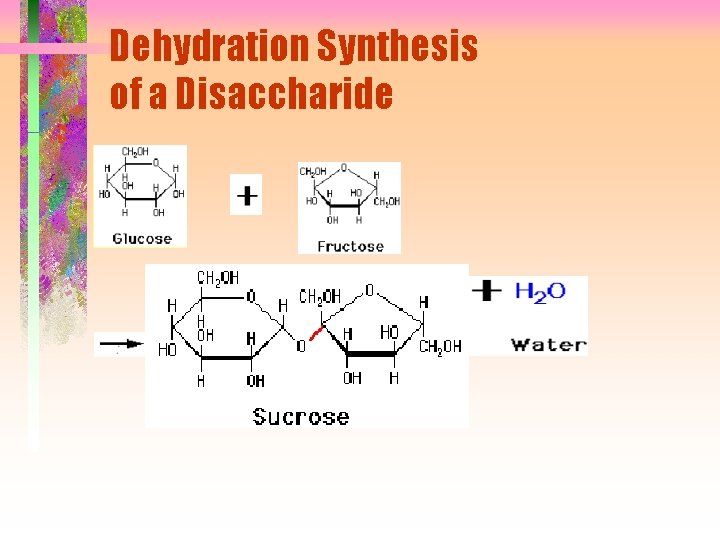
Dehydration Synthesis of a Disaccharide
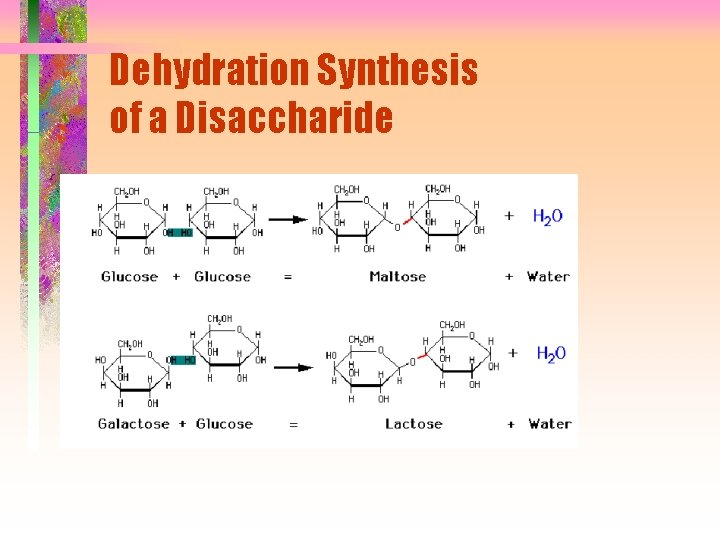
Dehydration Synthesis of a Disaccharide
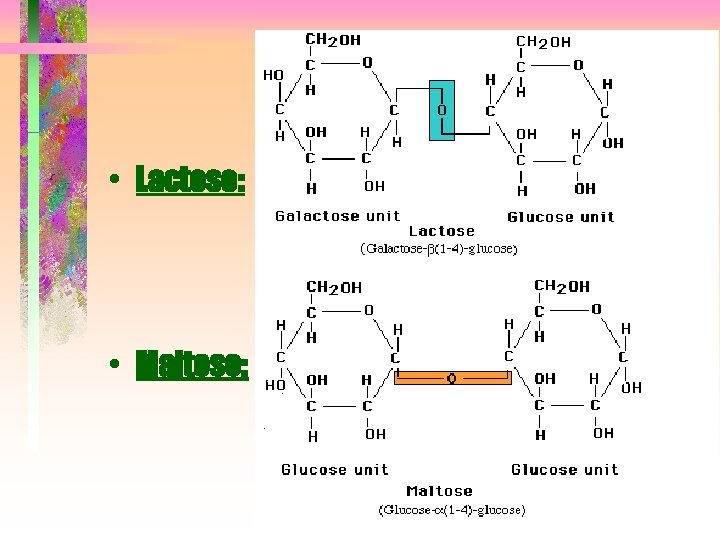
• Lactose: • Maltose:
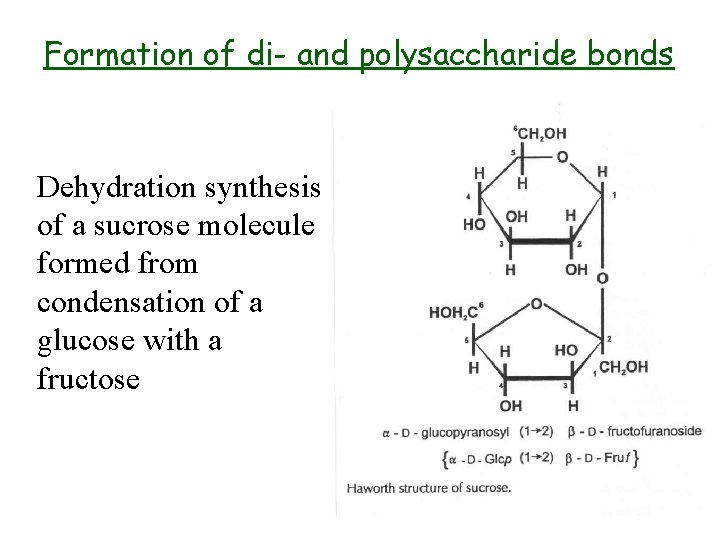
Formation of di- and polysaccharide bonds Dehydration synthesis of a sucrose molecule formed from condensation of a glucose with a fructose
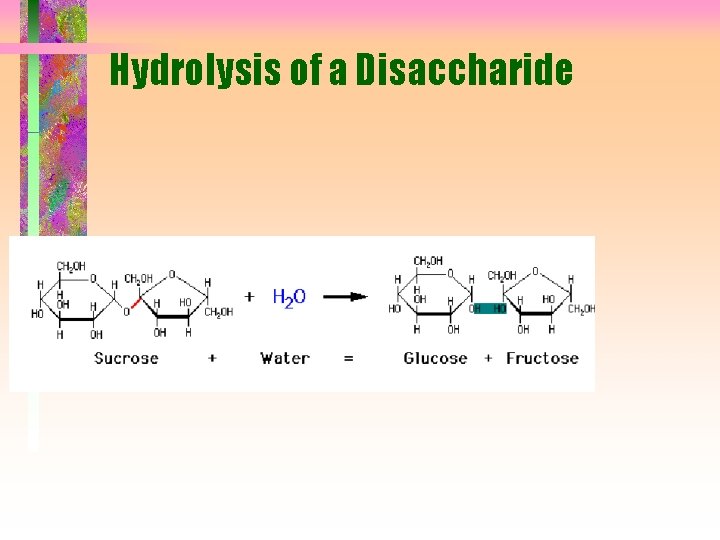
Hydrolysis of a Disaccharide
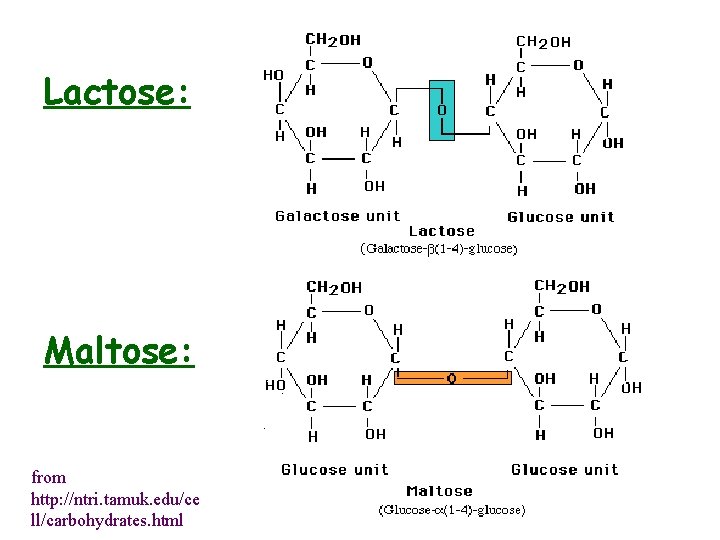
Lactose: Maltose: from http: //ntri. tamuk. edu/ce ll/carbohydrates. html

Disaccharides • Composed of two monosaccharide units by glycosidic link from C-1 of one unit and -OH of second unit • 1 3, 1 4, 1 6 links most common but 1 1 and 1 2 are possible • Links may be a or b • Link around glycosidic bond is fixed but anomeric forms on the other C-1 are still in equilibrium

Polysaccharides Primary Structure: Sequence of residues N. B. Many are homopolymers. Those that are heteropolymers rarely have >3, 4 different residues

Secondary & Tertiary Structure • • Rotational freedom hydrogen bonding oscillations local (secondary) and overall (tertiary) random coil, helical conformations
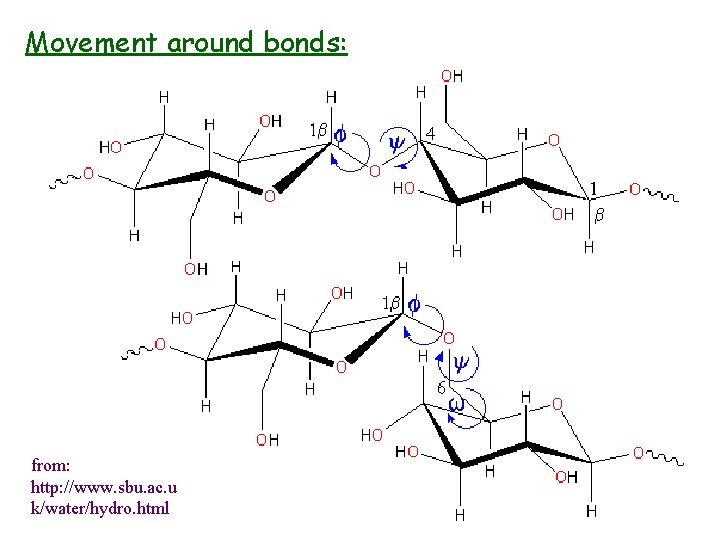
Movement around bonds: from: http: //www. sbu. ac. u k/water/hydro. html

Tertiary structure - sterical/geometrical conformations • Rule-of-thumb: Overall shape of the chain is determined by geometrical relationship within each monosaccharide unit l b(1 4) - zig-zag - ribbon like l b(1 3) & a(1 4) - U-turn - hollow helix l b(1 2) - twisted - crumpled l (1 6) - no ordered conformation
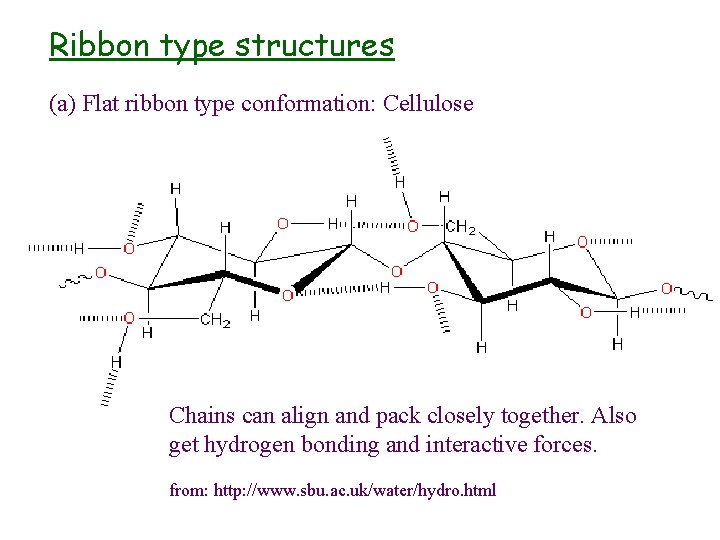
Ribbon type structures (a) Flat ribbon type conformation: Cellulose Chains can align and pack closely together. Also get hydrogen bonding and interactive forces. from: http: //www. sbu. ac. uk/water/hydro. html
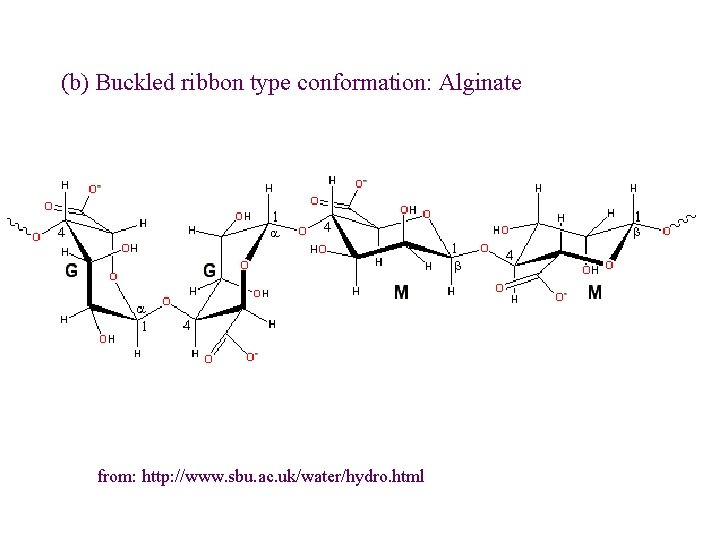
(b) Buckled ribbon type conformation: Alginate from: http: //www. sbu. ac. uk/water/hydro. html

Hollow helix type structures • Tight helix - void can be filled by including molecules of appropriate size and shape • More extended helix - two or three chains may twist around each other to form double or triple helix • Very extended helix - chains can nest, i. e. , close pack without twisting around each other
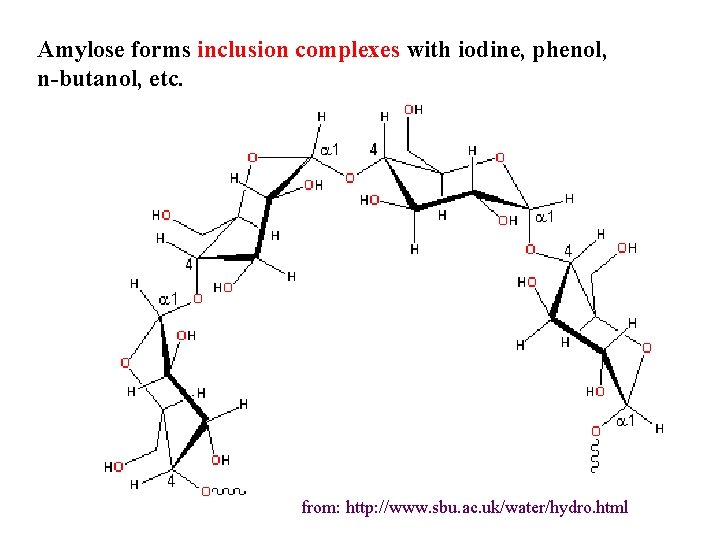
Amylose forms inclusion complexes with iodine, phenol, n-butanol, etc. from: http: //www. sbu. ac. uk/water/hydro. html

The liganded amyloseiodine complex: rows of iodine atoms (shown in black) neatly fit into the core of the amylose helix. N. B. Unliganded amylose normally exists as a coil rather than a helix in solution
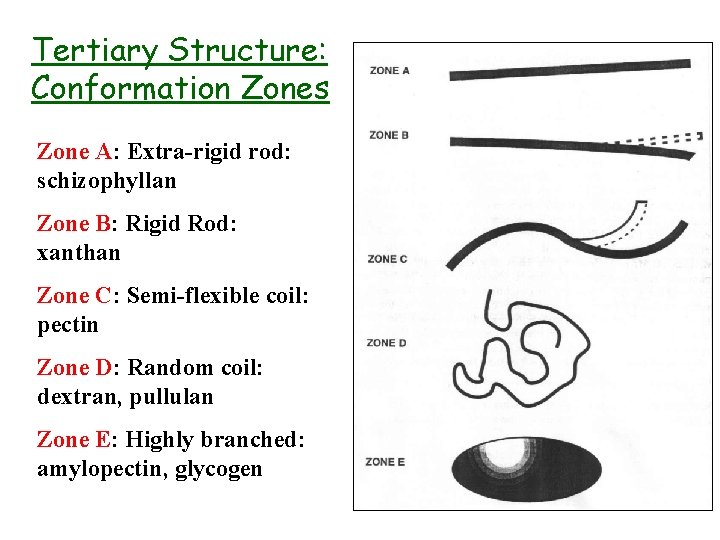
Tertiary Structure: Conformation Zones Zone A: Extra-rigid rod: schizophyllan Zone B: Rigid Rod: xanthan Zone C: Semi-flexible coil: pectin Zone D: Random coil: dextran, pullulan Zone E: Highly branched: amylopectin, glycogen

Quarternary structure aggregation of ordered structures Aggregate and gel formation: • May involve • other molecules such as Ca 2+ or sucrose • Other polysaccharides (mixed gels) …this will be covered in the lecture from Professor Mitchell

Polysaccharides – 6 case studies 1. 2. 3. 4. 5. 6. Alginates (video) Pectin Xanthan Galactomannans Cellulose Starch (Dr. Sandra Hill)

1. Alginate (E 400 -E 404) Source: Brown seaweeds (Phaeophyceae, mainly Laminaria) Linear unbranched polymers containing b-(1 4) -linked D-mannuronic acid (M) and a-(1 4)linked L-guluronic acid (G) residues Not random copolymers but consist of blocks of either MMM or GGG or MGMGMG
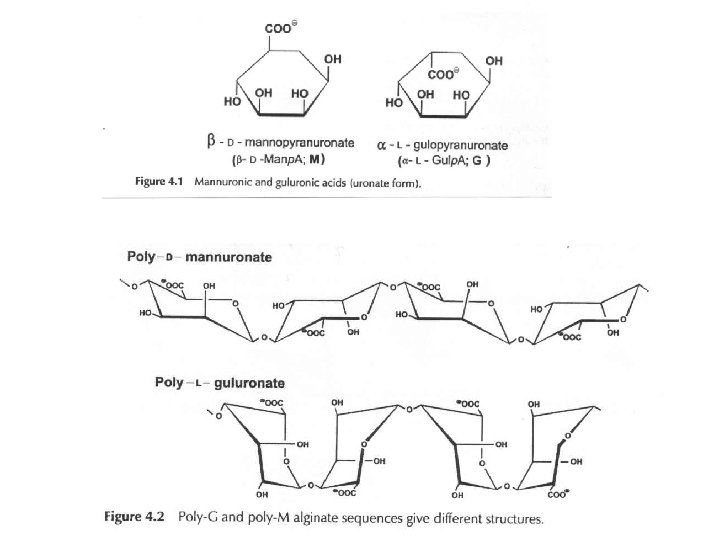
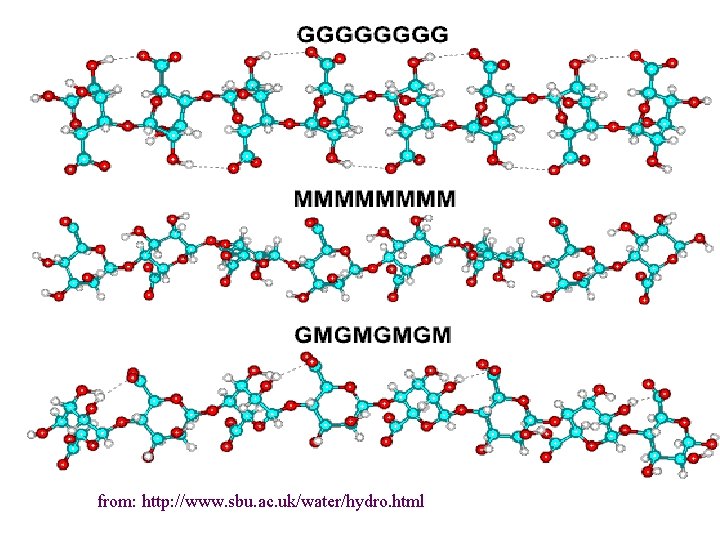
from: http: //www. sbu. ac. uk/water/hydro. html
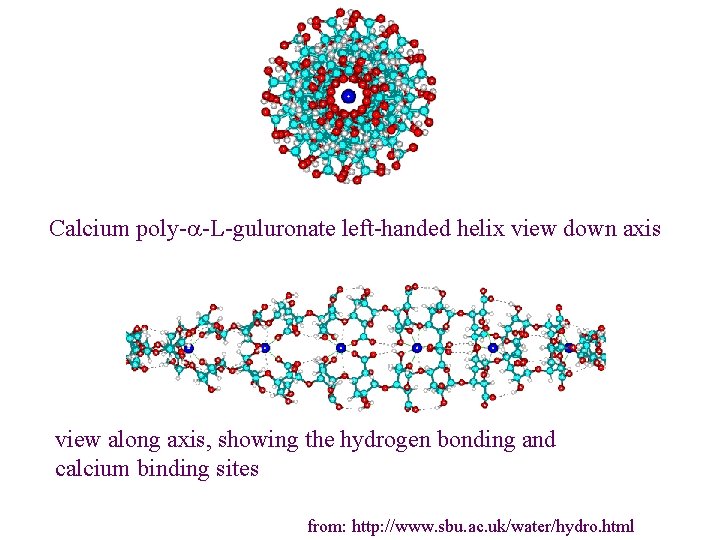
Calcium poly-a-L-guluronate left-handed helix view down axis view along axis, showing the hydrogen bonding and calcium binding sites from: http: //www. sbu. ac. uk/water/hydro. html

Different types of alginates different properties e. g. gel strength Polyguluronate: - gelation through addition of Ca 2+ ions – egg-box Polymannuronate – less strong gels, interactions with Ca 2+ weaker, ribbon-type conformation Alternating sequences – disordered structure, no gelation

Properties and Applications • High water absorption • Low viscosity emulsifiers and shear-thinning thickeners • Stabilize phase separation in low fat-substitutes e. g. as alginate/caseinate blends in starch threephase systems • Used in pet food chunks, onion rings, stuffed olives and pie fillings, wound healing agents, printing industry (largest use)
2. Pectin (E 440) • Cell wall polysaccharide in fruit and vegetables • Main source citrus peel
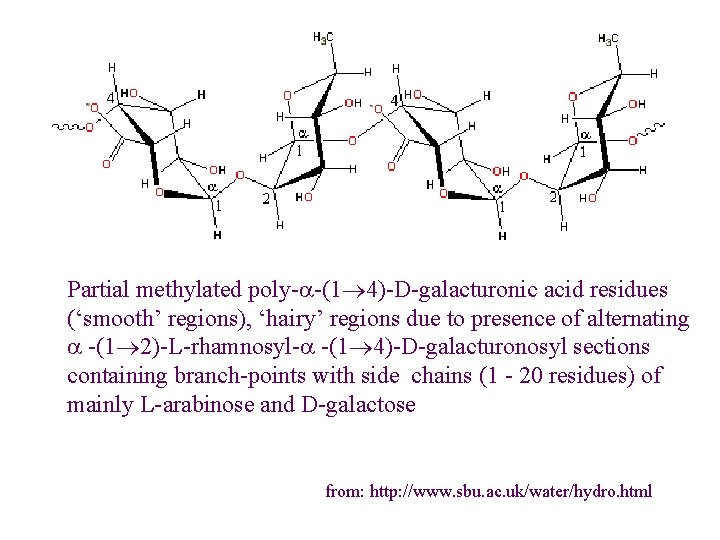
Partial methylated poly-a-(1 4)-D-galacturonic acid residues (‘smooth’ regions), ‘hairy’ regions due to presence of alternating a -(1 2)-L-rhamnosyl-a -(1 4)-D-galacturonosyl sections containing branch-points with side chains (1 - 20 residues) of mainly L-arabinose and D-galactose from: http: //www. sbu. ac. uk/water/hydro. html

Properties and applications • Main use as gelling agent (jams, jellies) – dependent on degree of methylation – high methoxyl pectins gel through H-bonding and in presence of sugar and acid – low methoxyl pectins gel in the presence of Ca 2+ (‘egg-box’ model) • Thickeners • Water binders • Stabilizers
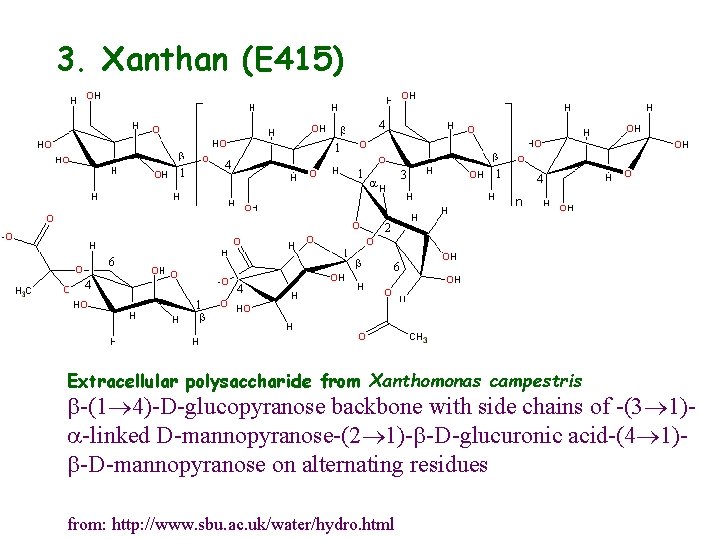
3. Xanthan (E 415) Extracellular polysaccharide from Xanthomonas campestris b-(1 4)-D-glucopyranose backbone with side chains of -(3 1)a-linked D-mannopyranose-(2 1)-b-D-glucuronic acid-(4 1)b-D-mannopyranose on alternating residues from: http: //www. sbu. ac. uk/water/hydro. html

Properties and applications • • double helical conformation pseudoplastic shear-thinning thickener stabilizer emulsifier foaming agent forms synergistic gels with galactomannans

4. Galactomannans b-(1 4) mannose (M) backbone with a(1 6) galactose (G) side chains • Ratio of M to G depends on source – M: G=1: 1 - fenugreek gum – M: G=2: 1 - guar gum (E 412) – M: G=3: 1 - tara gum – M: G=4: 1 - locust bean gum (E 410) l
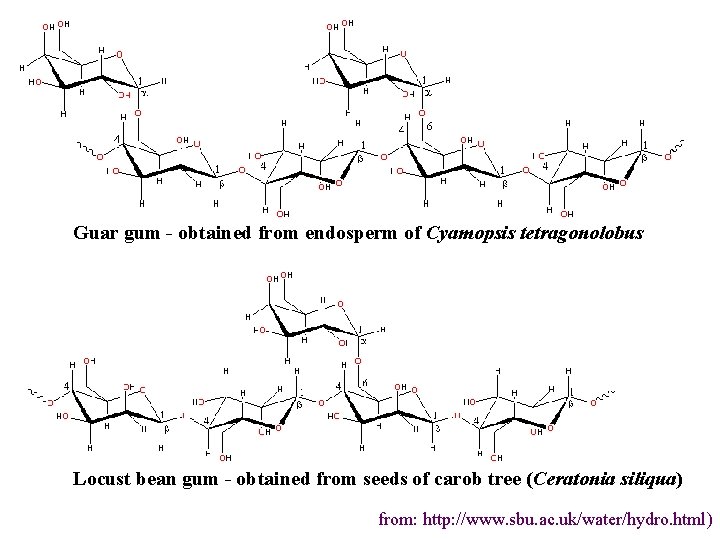
Guar gum - obtained from endosperm of Cyamopsis tetragonolobus Locust bean gum - obtained from seeds of carob tree (Ceratonia siliqua) from: http: //www. sbu. ac. uk/water/hydro. html)

Properties and applications • non-ionic • solubility decreases with decreasing galactose content • thickeners and viscosifiers • used in sauces, ice creams • LBG can form very weak gels
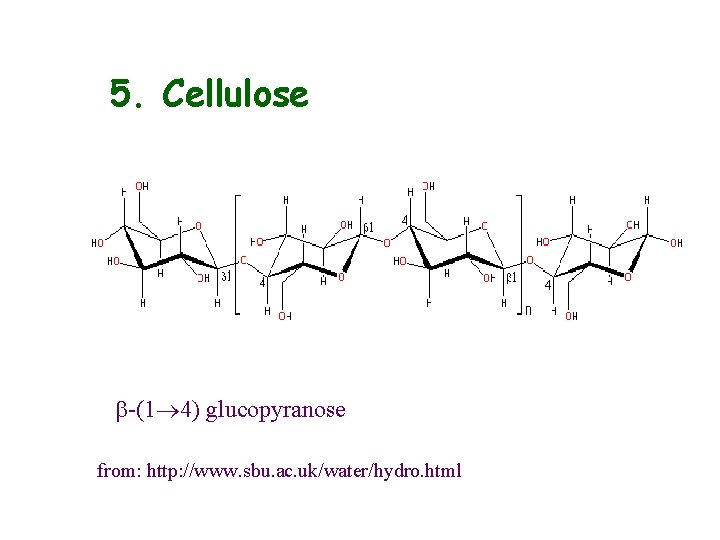
5. Cellulose b-(1 4) glucopyranose from: http: //www. sbu. ac. uk/water/hydro. html

Properties and applications • found in plants as microfibrils • very large molecule, insoluble in aqueous and most other solvents • flat ribbon type structure allows for very close packing and formation of intermolecular H-bonds • two crystalline forms (Cellulose I and II) • derivatisation increases solubility (hydroxy-propyl methyl cellulose, carboxymethyl cellulose, etc. )

Properties and applications • • Composed of glucose subunits Different bond formed than starch Structural component in plants Cannot be digested by animals
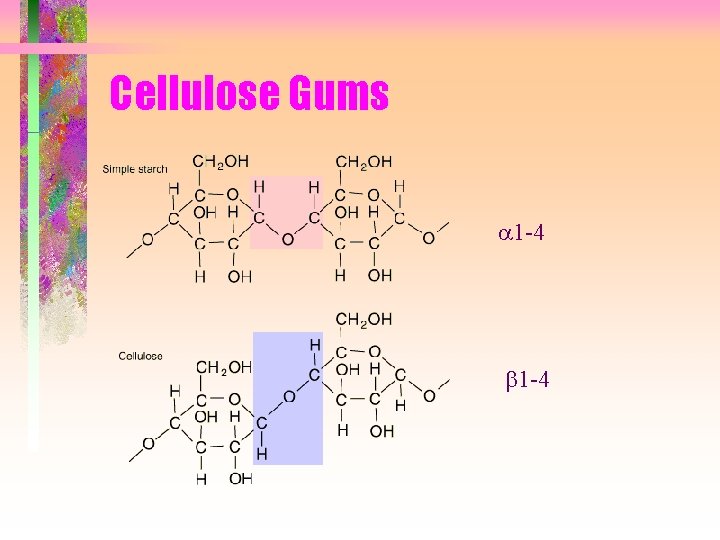
Cellulose Gums a 1 -4 b 1 -4

Starch Consists of glucose subunits Plant energy storage molecule Glycogen is a very similar molecule in animals. Starch and glycogen can be digested by animals
- Slides: 53