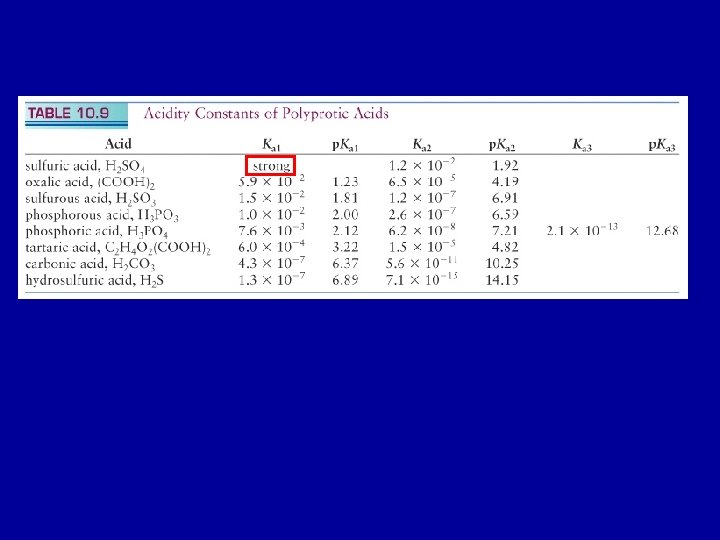
Polyprotic Acids Bases A polyprotic acid can donate

Polyprotic Acids & Bases A polyprotic acid can donate more than one H+ Carbonic acid: H 2 CO 3(aq); dissolved CO 2 in water Sulfuric acid: H 2 SO 4(aq) Phosphoric acid: H 3 PO 4(aq) A polyprotic base: can accept more than one proton Carbonate ion: CO 32 -(aq) Sulfate ion: SO 42 -(aq) Phophate ion: PO 43 -(aq) Treat each step of protonation or deprotonation sequentially

H 2 CO 3 (aq) + H 2 O(l) H 3 O+(aq) + HCO 3 -(aq) Ka 1 = 4. 3 x 10 -7 HCO 3 -(aq) + H 2 O(l) H 3 O+(aq) + CO 32 -(aq) Ka 2 = 4. 8 x 10 -11 Typically: Ka 1 >> Ka 2 >> Ka 3 >>… Harder to loose a positively charged proton from a negatively charged ion, because of attraction between opposite charges.

Calculate the p. H of 0. 010 M H 2 SO 4(aq) at 25 o. C. Sulfuric acid is the only common polyprotic acid where the first deprotonation step is complete. The second deprotonation step is much weaker and adds slightly to the H 3 O+(aq) concentration. For the first step assume all H 2 SO 4(aq) deprotonates H 2 SO 4 (aq) + H 2 O(l) H 3 O+ (aq) + HSO 4 -(aq) From the first step [H 3 O+(aq)] = 0. 010 M

Second deprotonation HSO 4 - (aq) + H 2 O(l) H 3 O+ (aq) + SO 42 - (aq) Ka 2 = 0. 012 Initial Change Equilibrium HSO 4 - (aq) SO 42 - (aq) 0. 010 0 -x +x 0. 010 -x x H 3 O+ (aq) 0. 010 + x Ka 2 = ([H 3 O+ (aq)])([SO 42 - (aq)]) / ([HSO 4 - (aq) ]) 0. 012 = (0. 010+x)(x) / (0. 010 -x) Solve the quadratic equation for x. Ka 2 is large; cannot assume that x << 0. 010 [H 3 O+ (aq)] = 1. 4 x 10 -2 M p. H = 1. 9

Determine the p. H of 0. 20 M H 2 S(aq) at 25 o. C H 2 S (aq) + H 2 O(l) H 3 O+ (aq) + HS- (aq) + H 2 O(l) H 3 O+ (aq) + S 2 - (aq) Ka 1 = 1. 3 x 10 -7 Ka 2 = 7. 1 x 10 -15 For the first deprotonation step determine [H 3 O+(aq)] using equilibrium tables. [H 3 O+(aq)] = 1. 6 X 10 -4 M Can assume that x << 0. 20 since Ka 1 is small Second deprotonation constant is very small, so ignore addition of H 3 O+(aq) due to second step. p. H determined by first step alone. p. H = 3. 8

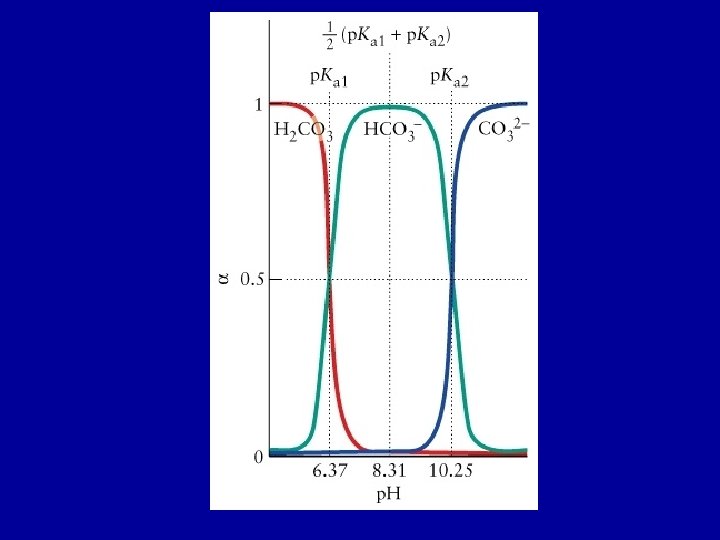
Composition and p. H For a solution of H 2 CO 3(aq): at low p. H the fully protonated species (H 2 CO 3) dominates; at high p. H the fully deprotonated form (CO 32 -) dominates; and at intermediate p. H the intermediate species (HCO 3 -) dominates. Le. Chatelier’s principle at work H 2 CO 3 (aq) + H 2 O(l) H 3 O+(aq) + HCO 3 -(aq) Ka 1 = 4. 3 x 10 -7 HCO 3 -(aq) + H 2 O(l) H 3 O+(aq) + CO 32 -(aq) Ka 2 = 4. 8 x 10 -11

H 2 CO 3(aq) + H 2 O(l) H 3 O+(aq) + HCO 3 -(aq) Ka 1 = [H 3 O+(aq)] [HCO 3 -(aq)] / [H 2 CO 3(aq) ] HCO 3 -(aq) + H 2 O(l) H 3 O+(aq) + CO 32 -(aq) Ka 2 = [H 3 O+(aq)] [CO 32 -(aq)] / [HCO 3 -(aq) ] Define a(X): fraction of species X a(X) = X [H 2 CO 3(aq) ] + [HCO 3 -(aq)] + [CO 32 -(aq)] The fraction of deprotonated species increases as the p. H increases
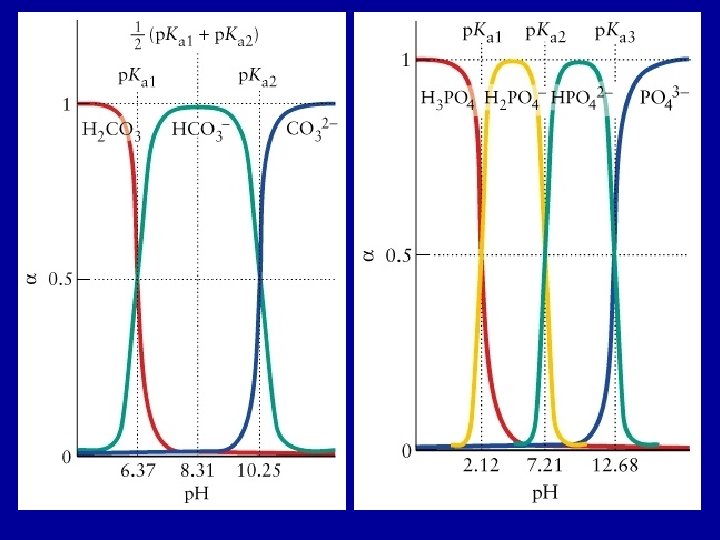

Determine the concentration of H 2 CO 3(aq), HCO 3 -(aq), CO 32(aq), H 3 O+(aq) present and the p. H at equilibrium in a solution that is initially 0. 010 M in H 2 CO 3. (Ka 1 = 4. 3 x 10 -7, Ka 2 = 4. 8 x 10 -11) Step 1 H 2 CO 3(aq) + H 2 O(l) H 3 O+(aq) + HCO 3 -(aq) Initial Change H 2 CO 3(aq) 0. 010 H 3 O+(aq) 0 -x Equilibrium 0. 010 -x x x 4. 3 x 10 -7 = x 2 / (0. 010 -x) Assume x << 0. 010; x = 6. 6 x 10 -5 [H 2 CO 3(aq)] ≈ 0. 010 M; [H 3 O+(aq)] = 6. 6 x 10 -5 M; [HCO 3 -(aq)] = 6. 6 x 10 -5 M HCO 3 -(aq) 0

HCO 3 -(aq) + H 2 O(l) H 3 O+(aq) + CO 32 -(aq) Ka 2 = [H 3 O+(aq)] [CO 32 -(aq)] / [HCO 3 -(aq) ] Initial Change HCO 3 -(aq) 6. 6 x 10 -5 -y Equilibrium 6. 6 x 10 -5 - y H 3 O+(aq) 6. 6 x 10 -5 CO 32 -(aq) 0 6. 6 x 10 -5 + y y 4. 8 x 10 -11 = (6. 6 x 10 -5 + y) y / (6. 6 x 10 -5 - y) Assume y << 6. 6 x 10 -5 4. 8 x 10 -11 = (6. 6 x 10 -5 y / (6. 6 x 10 -5 ); y = 4. 8 x 10 -11 At equilibrium: [H 2 CO 3(aq)] ≈ 0. 010 M; [H 3 O+(aq)] = 6. 6 x 10 -5 M; [HCO 3 -(aq)] = 6. 6 x 10 -5 M; [CO 32 -(aq)] = 4. 8 x 10 -11 M p. H = 4. 18

Buffers Buffer solutions : resists change in p. H even with addition of small amounts of acid or base. Buffer solutions are mixed solutions: mixture of a weak acid and its conjugate base or a weak base and its conjugate acid. Human blood has a p. H maintained at p. H = 7. 4 due to a combination of carbonate, phosphate and protein buffers. The ocean is buffered to a p. H of ~ 8. 4 by buffering that depends the presence of hydrogen carbonates and silicates.

Buffer Action An acid buffer is an aqueous solution of a weak acid and its conjugate base. It buffers solutions on the acid side of neutral (p. H < 7). Example: solution of CH 3 COOH(aq) + CH 3 COONa(aq) CH 3 COOH / CH 3 COOA base buffer is an aqueous solution of a weak base and its conjugate acid. It buffers solutions on the basic side of neutral (p. H > 7). Example: NH 3(aq) + NH 4 Cl(aq) NH 3/ NH 4+

Buffer solution of CH 3 COOH(aq) / CH 3 COO- (aq) If a small amount of strong acid is added: H 3 O+(aq) + CH 3 COO-(aq) CH 3 COOH(aq) + H 2 O(l) K for this reaction = 1/Ka(CH 3 COOH(aq)) = 5. 5 x 104 The CH 3 COO-(aq) acts as a “sink” for the added protons, and the p. H remains unchanged. If a small amount of strong base is added OH-(aq) + CH 3 COOH(aq) CH 3 COO-(aq) + H 2 O(l) K for this reaction = 1/Kb(CH 3 COO-(aq)) = 1. 8 x 109 The CH 3 COOH(aq) acts as a “sink” for the added OH-, and the p. H remains unchanged

Designing a buffer Make a solution with particular p. H so that it buffers about this p. H. Consider a solution of a weak acid and its conjugate base HA(aq) + H 2 O(l) H 3 O+(aq) + A-(aq) [H 3 O+(aq)] - log [H 3 = Ka O+(aq)] p. H = p. Ka + log [HA(aq)] [A-(aq)] [HA(aq)] = - log Ka - log [A-(aq)] [A- (aq)] [HA(aq)] Henderson-Hasselbach equation Note: for a solution of a weak base/conjugate acid, use Ka of the conjugate acid

An optimal buffer is one in which the weak acid and its conjugate base have equal concentrations Select a weak acid that has its p. Ka as close as possible to the desired p. H Having chosen the weak acid, use the Henderson Hasselbach equation to determine the ratio of [A-(aq)] and [HA(aq)] that will form solution that buffers around the desired p. H

Calculate the p. H of a buffer solution that is 0. 040 M CH 3 COONa (aq) and 0. 080 M CH 3 COOH (aq). p. Ka(CH 3 COOH(aq)) = 4. 75 CH 3 COOH(aq) + H 2 O(l) H 3 O+(aq) + CH 3 COO-(aq) CH 3 COOH(aq) Initial 0. 080 M Change -x Equilibrium 0. 080 - x Ka = (0. 040 + x) x / (0. 080 - x) Assume x << 0. 040 x = 3. 6 x 10 -5 p. H = 4. 44 H 3 O+(aq) 0 x x CH 3 COO-(aq) 0. 040 M 0. 040 + x

Suppose that a solution is made by dissolving 1. 2 g Na. OH(s) (0. 030 moles) in 500 m. L of the buffer solution in the previous problem. Calculate the p. H of the resulting solution and the change in p. H. Assume the volume of the solution to be constant. The OH-(aq) will react with CH 3 COOH(aq) + OH-(aq) CH 3 COO-(aq) + H 2 O(l) Moles of OH- added = 0. 030 moles Moles of CH 3 COOH(aq) = (0. 500 L) (0. 080 M) = 0. 040 mol Amount of unreacted CH 3 COOH(aq) = 0. 010 moles Molarity of CH 3 COOH(aq) = 0. 020 M

Moles of CH 3 COO-(aq) = initial amount + amount formed by reaction of OH- (aq) and CH 3 COOH(aq) = (0. 040 M x 0. 500 L) + (0. 030 moles) = 0. 050 moles Molarity of CH 3 COO-(aq) = 0. 10 M CH 3 COOH(aq) + H 2 O(l) CH 3 COO-(aq) + H 3 O+(aq) p. H = p. Ka + log ([CH 3 COO-(aq) ] / [CH 3 COOH(aq) ] ) = 5. 45 If the solution had contained HCl at p. H = 4. 4, addition of the Na. OH would have raised the p. H to 12. 8
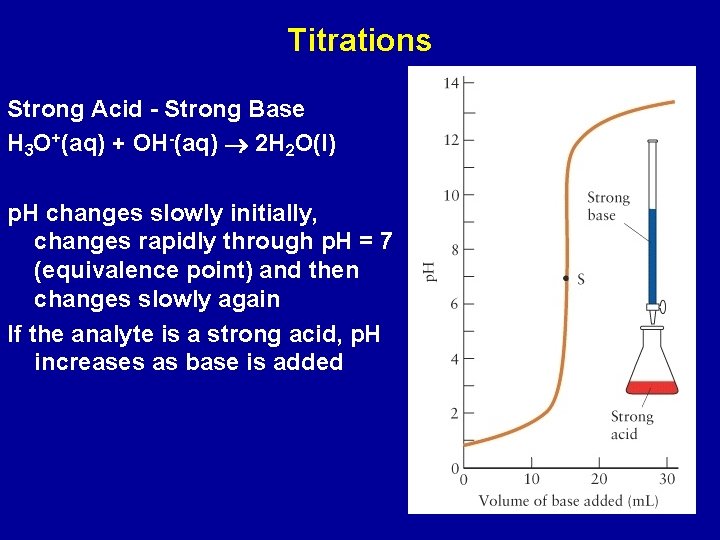
Titrations Strong Acid - Strong Base H 3 O+(aq) + OH-(aq) 2 H 2 O(l) p. H changes slowly initially, changes rapidly through p. H = 7 (equivalence point) and then changes slowly again If the analyte is a strong acid, p. H increases as base is added
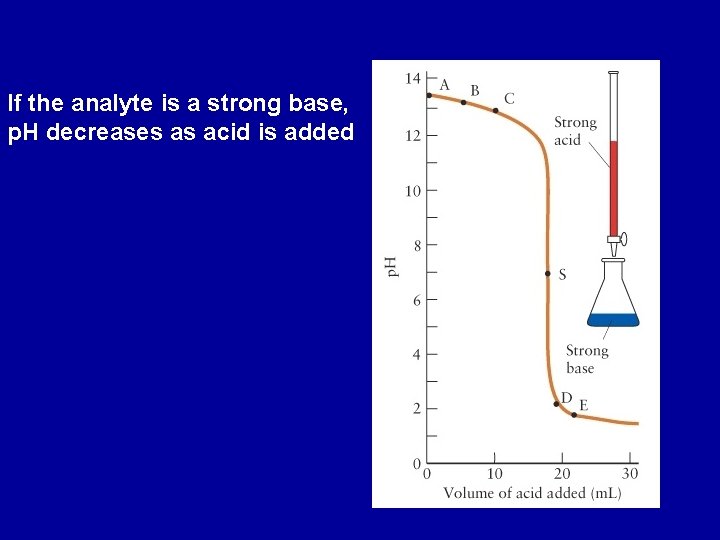
If the analyte is a strong base, p. H decreases as acid is added

Analyte: 25. 00 m. L of 0. 250 M Na. OH(aq) Titrant: 0. 340 M HCl(aq) Determine the p. H of the solution when 5. 00 m. L of titrant added Answer: p. H = 13. 18 Determine the amount of titrant that must be added to reach the equivalence point? Answer: 18. 4 m. L Determine the p. H of the solution after the addition of 20. 4 m. L of titrant. Answer: p. H = 1. 82
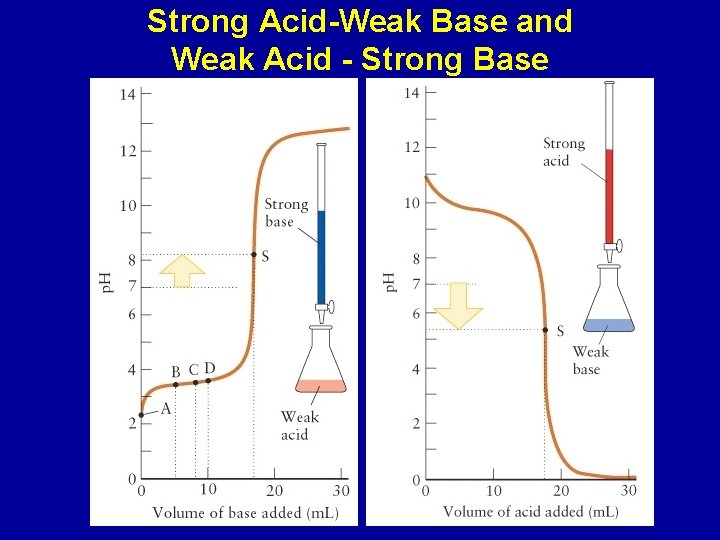
Strong Acid-Weak Base and Weak Acid - Strong Base
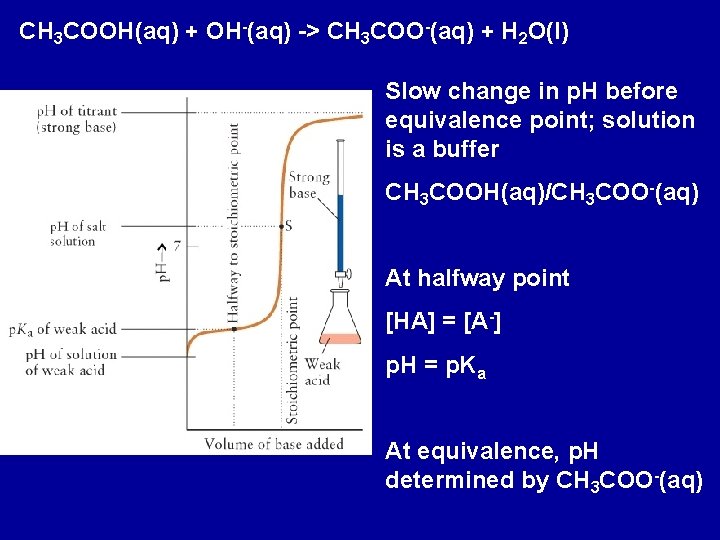
CH 3 COOH(aq) + OH-(aq) -> CH 3 COO-(aq) + H 2 O(l) Slow change in p. H before equivalence point; solution is a buffer CH 3 COOH(aq)/CH 3 COO-(aq) At halfway point [HA] = [A-] p. H = p. Ka At equivalence, p. H determined by CH 3 COO-(aq)

Changes in p. H during a titration of a weak acid/base with a strong base/acid: Halfway to the stoichiometric point, the p. H = p. Ka of the acid The p. H is greater than 7 at the equivalence point of the titration of a weak acid and strong base The p. H is less that 7 at the equivalence point of the titration of a weak base and strong acid Beyond the equivalence point, the excess strong acid or base will determine the p. H of the solution

Titration of 100. 0 m. L of 0. 1000 M CH 3 COOH(aq) with 0. 1000 M Na. OH Before addition of Na. OH: p. H determined by CH 3 COOH(aq) + H 2 O(l) H 3 O+(aq) + CH 3 COO-(aq) Answer: p. H = 2. 88 Before the equivalence point: determine p. H for a buffer Addition of 30. 00 m. L of Na. OH(aq) The OH-(aq) reacts with the CH 3 COOH(aq). Determine concentration of CH 3 COOH(aq) and CH 3 COO- (aq) in solution after addition of the base. Answer: p. H = 4. 38 At half equivalence: [CH 3 COOH(aq)] = [CH 3 COO-(aq)] p. H = p. Ka

At equivalence: enough OH-(aq) added to react with all CH 3 COOH(aq). For this problem, equivalence is reached when 100. 0 m. L of OH- is added; i. e. 0. 01000 moles of OH-(aq) added Solution contains 0. 01000 moles CH 3 COO-(aq) in 200. 0 m. L solution; [CH 3 COO-(aq)] = 0. 05000 M p. H determined by CH 3 COO-(aq) + H 2 O(l) CH 3 COOH(aq) + OH- (aq) p. H = 8. 72 (note greater than 7. 0) Beyond equivalence: p. H determined by excess OH-(aq)

Estimate the p. H at the equivalence point of the titration of 25. 00 m. L of 0. 100 M HCOOH(aq) with 0. 150 M Na. OH(aq) (Ka(HCOOH) = 1. 8 x 10 -4) At the equivalence point, enough Na. OH(aq) has been added to react with all the HCOOH(aq) forming HCOO-(aq) The reaction: HCOO-(aq) + H 2 O(l) HCOOH(aq) + OHdetermines the p. H at equivalence Answer: 8. 26
- Slides: 29