Polyprotic AcidBase Equilibria Introduction 1 Polyprotic systems Acid

Polyprotic Acid-Base Equilibria Introduction 1. ) Polyprotic systems Ø Ø Acid or bases that can donate or accept more than one proton Proteins are a common example of a polyprotic system - Ø why activity of proteins are p. H dependent Polymer of amino acids - Some amino acids have acidic or basic substituents

Polyprotic Acid-Base Equilibria Introduction 1. ) Polyprotic systems Ø Amino acids (basic) (acidic) - Carboxyl group is stronger acid of ammonium group R is different group for each amino acid Overall charge is still neutral Ø Amino acids are zwitterion – molecule with both positive and negative charge - At low p. H, both ammonium and carboxy group are protonated At high p. H, neither group is protonated Stabilized by interaction with solvent
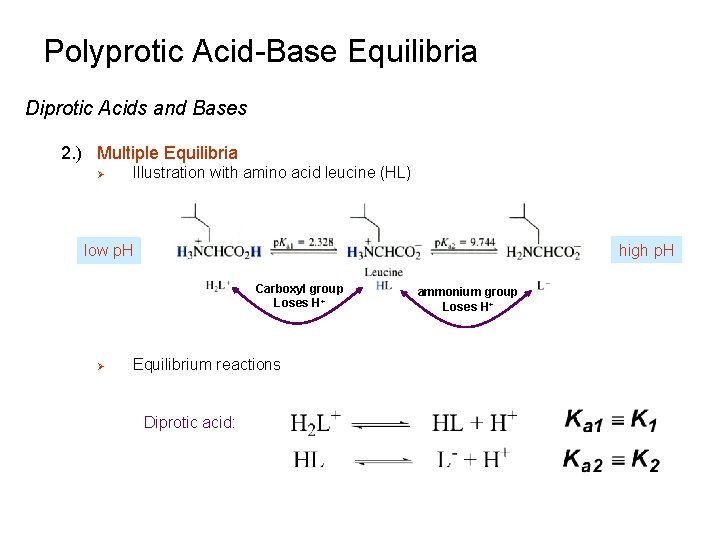
Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 2. ) Multiple Equilibria Ø Illustration with amino acid leucine (HL) high p. H low p. H Carboxyl group Loses H+ Ø Equilibrium reactions Diprotic acid: ammonium group Loses H+

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 2. ) Multiple Equilibria Ø Equilibrium reactions Diprotic base: Relationship between Ka and Kb
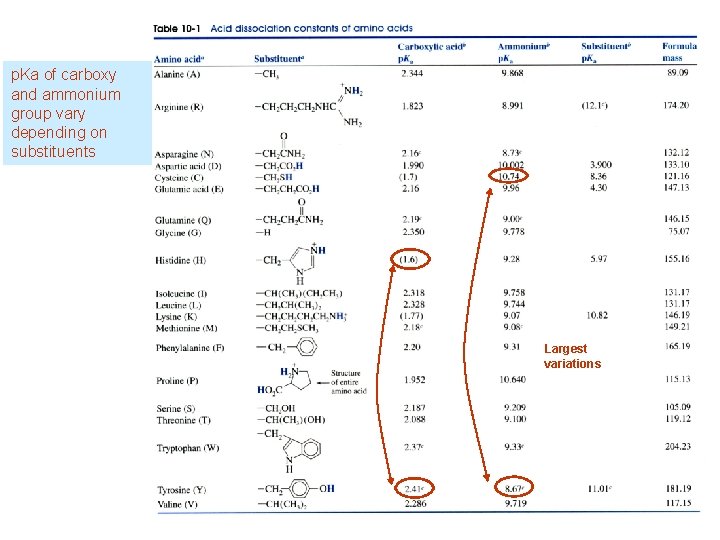
p. Ka of carboxy and ammonium group vary depending on substituents Largest variations
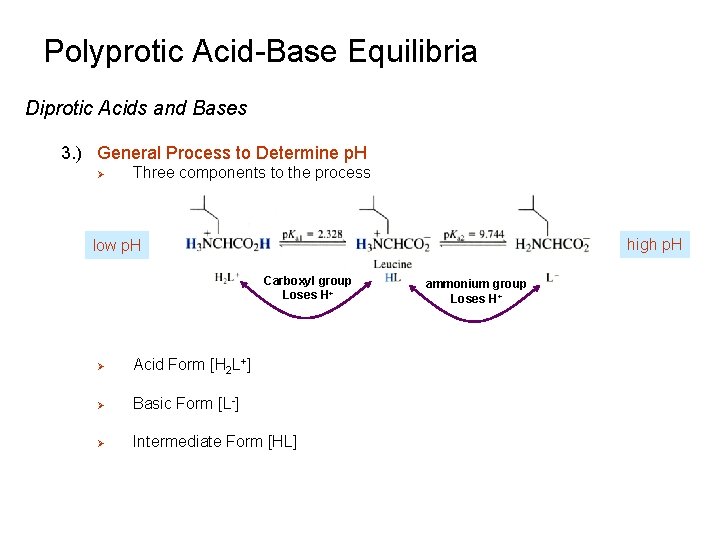
Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Three components to the process high p. H low p. H Carboxyl group Loses H+ Ø Acid Form [H 2 L+] Ø Basic Form [L-] Ø Intermediate Form [HL] ammonium group Loses H+

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Acid Form (H 2 L+) Ø Illustration with amino acid leucine K 1=4. 70 x 10 -3 Ø K 2=1. 80 x 10 -10 H 2 L+ is a weak acid and HL is a very weak acid Assume H 2 L+ behaves as a monoprotic acid
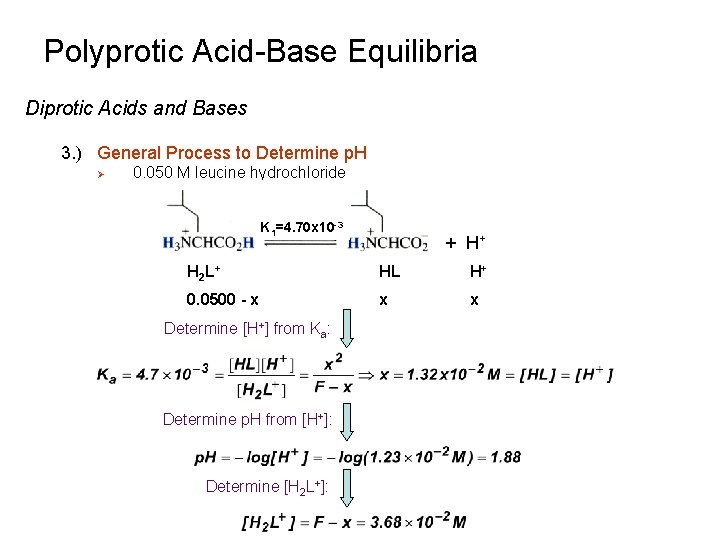
Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø 0. 050 M leucine hydrochloride K 1=4. 70 x 10 -3 + H+ H 2 L + HL H+ 0. 0500 - x x x Determine [H+] from Ka: Determine p. H from [H+]: Determine [H 2 L+]:

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Acid Form (H 2 L+) What is the concentration of L- in the solution? [L-] is very small, but non-zero. Calculate from Ka 2 Approximation [H+] ≈ [HL], reduces Ka 2 equation to [L-]=Ka 2 Validates assumption

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø For most diprotic acids, K 1 >> K 2 - Ø Assumption that diprotic acid behaves as monoprotic is valid Ka ≈ Ka 1 Even if K 1 is just 10 x larger than K 2 - Error in p. H is only 4% or 0. 01 p. H units Ø Basic Form (L-) Ø L- is a weak base and HL is an extremely weak base Assume L- behaves as a monoprotic base
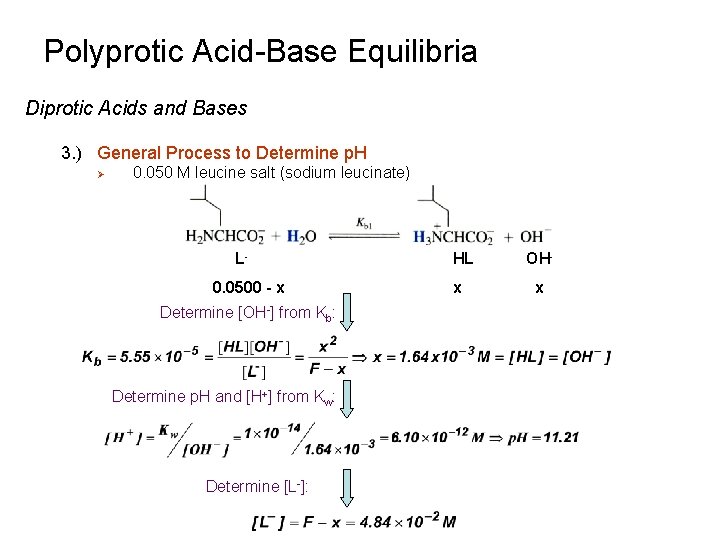
Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø 0. 050 M leucine salt (sodium leucinate) L 0. 0500 - x Determine [OH-] from Kb: Determine p. H and [H+] from Kw: Determine [L-]: HL x OHx

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Basic Form (L-) What is the concentration of H 2 L+ in the solution? [H 2 L+] is very small, but non-zero. Calculate from Kb 2 Validates assumption [OH-] ≈ [HL], Fully basic form of a diprotic acid can be treated as a monobasic, K b=Kb 1

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Intermediate Form (HL) - More complicated HL is both an acid and base Ø Amphiprotic – can both donate and accept a proton Ø Since Ka > Kb, expect solution to be acidic - Ø Can not ignore base equilibrium Need to use Systematic Treatment of Equilibrium

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Intermediate Form (HL) Step 1: Pertinent reactions: Step 2: Charge Balance: Step 3: Mass Balance: Step 4: Equilibrium constant expression (one for each reaction):

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Intermediate Form (HL) Step 6: Solve: Substitute Acid Equilibrium Equations into charge balance: All Terms are related to [H+] Multiply by [H+]
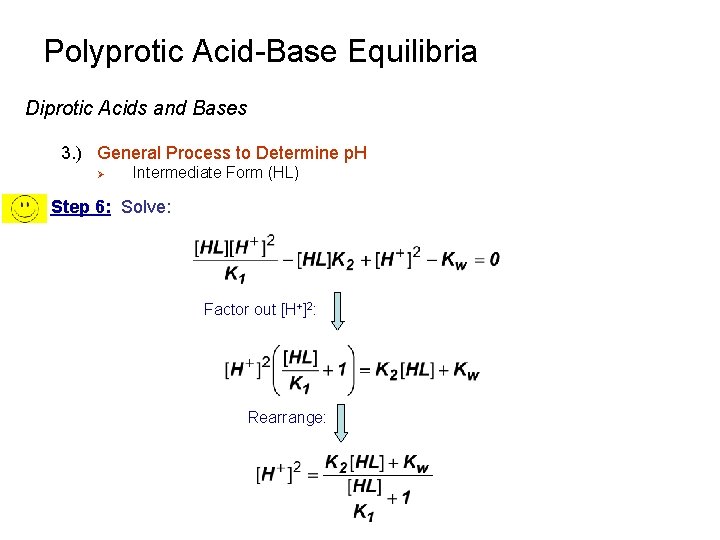
Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Intermediate Form (HL) Step 6: Solve: Factor out [H+]2: Rearrange:

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Intermediate Form (HL) Step 6: Solve: Multiply by K 1 and take square-root: Assume [HL]=F, minimal dissociation: (K 1 & K 2 are small)

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Intermediate Form (HL) Step 6: Solve: Calculate a p. H:

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Intermediate Form (HL) Step 7: Validate Assumptions Assume [HL]=F=0. 0500 M, minimal dissociation (K 1 & K 2 are small). Calculate [L-] & [H 2 L+] from K 1 & K 2: [HL]=0. 0500 M >> 9. 36 x 10 -6 [H 2 L+] & 1. 02 x 10 -5 [L-] Assumption Valid
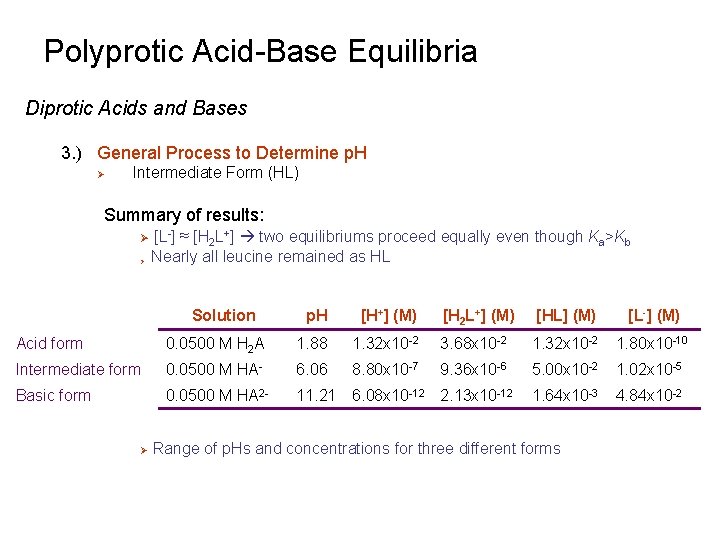
Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Intermediate Form (HL) Summary of results: Ø Ø [L-] ≈ [H 2 L+] two equilibriums proceed equally even though Ka>Kb Nearly all leucine remained as HL Solution p. H [H+] (M) [H 2 L+] (M) [HL] (M) [L-] (M) Acid form 0. 0500 M H 2 A 1. 88 1. 32 x 10 -2 3. 68 x 10 -2 1. 32 x 10 -2 1. 80 x 10 -10 Intermediate form 0. 0500 M HA- 6. 06 8. 80 x 10 -7 9. 36 x 10 -6 5. 00 x 10 -2 1. 02 x 10 -5 Basic form 0. 0500 M HA 2 - 11. 21 6. 08 x 10 -12 2. 13 x 10 -12 1. 64 x 10 -3 4. 84 x 10 -2 Ø Range of p. Hs and concentrations for three different forms

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Simplified Calculation for the Intermediate Form (HL) Assume K 2 F >> Kw: Assume K 1<< F:

Polyprotic Acid-Base Equilibria Diprotic Acids and Bases 3. ) General Process to Determine p. H Ø Simplified Calculation for the Intermediate Form (HL) Cancel F: Take the -log: Independent of concentration: p. H of intermediate form of a diprotic acid is close to midway between p. K 1 and p. K 2

Polyprotic Acid-Base Equilibria Diprotic Buffers 1. ) Same Approach as Monoprotic Buffer Ø Write two Henderson-Hasselbalch equations Ø Both Equations are always true Ø Solution only has one p. H Ø Choice of equation is based on what is known - [H 2 A] and [HA-] known use p. K 1 equation [HA-] and [A 2 -] known use p. K 2 equation
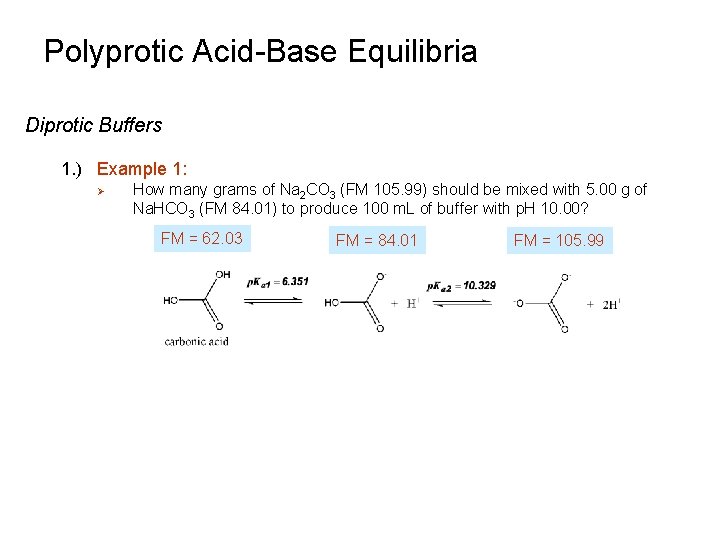
Polyprotic Acid-Base Equilibria Diprotic Buffers 1. ) Example 1: Ø How many grams of Na 2 CO 3 (FM 105. 99) should be mixed with 5. 00 g of Na. HCO 3 (FM 84. 01) to produce 100 m. L of buffer with p. H 10. 00? FM = 62. 03 FM = 84. 01 FM = 105. 99
Polyprotic Acid-Base Equilibria Diprotic Buffers 1. ) Example 2: Ø How many milliliters of 0. 202 M Na. OH should be added to 25. 0 m. L of 0. 0233 M of salicylic acid (2 -hydroxybenzoic acid) to adjust the p. H to 3. 50?

Polyprotic Acid-Base Equilibria Polyprotic Acids and Bases 1. ) Extend Treatment of Diprotic Acids and Bases to Polyprotic Systems Ø Equilibria for triprotic system Acid equilibria: Base equilibria: Ø For a polyprotic system, would simply contain n such equilibria

Polyprotic Acid-Base Equilibria Polyprotic Acids and Bases 1. ) Extend Treatment of Diprotic Acids and Bases to Polyprotic Systems Ø Rules for triprotic system 1. 2. H 3 A is treated as a monoprotic acid, Ka = K 1 H 2 A- is treated similarly as an intermediate form of a diprotic acid 3. HA 2 - is also treated similarly as an intermediate form of a diprotic acid a. b. 4. Surrounded by H 2 A- and A 3 Use K 2 & K 3, instead of K 1 & K 2 A 3 - is treated as monobasic, with Kb=Kb 1=Kw/Ka 3

Polyprotic Acid-Base Equilibria Polyprotic Acids and Bases 1. ) Extend Treatment of Diprotic Acids and Bases to Polyprotic Systems Ø Rules for triprotic system Treat as Monoprotic Treat asacid: Intermediate Forms Treatas as Intermediate Monoprotic Forms base: For more complex system, just have additional intermediate forms K s that “bracket” or contain form, “End Forms” Use of Equilibria that Bracket Reactions are KTreated 1 & K 3 2 as Monoprotic 2 in-between the atwo monoprotic acid and base forms at “ends”

Polyprotic Acid-Base Equilibria Polyprotic Acids and Bases 3. ) Which is the Principal Species? Ø Depends on the p. H of the Sample and the p. Ka values - Ø At p. Ka, 1: 1 mixture of HA and AFor monoprotic, A- is predominant when p. H > p. Ka For monoprotic, HA is predominant when p. H < p. Ka Similar for polyprotic, but several p. Ka values Triprotic acid Diprotic acid p. H Major Species p. H < p. K 1 H 2 A p. K 1 < p. H < p. K 2 HA- p. H > p. K 2 A 2 - Determine Principal Species by Comparing the p. H of the Solution with the p. Ka Values

Polyprotic Acid-Base Equilibria Polyprotic Acids and Bases 4. ) Fractional Composition Equations Ø Ø Fraction of Each Species at a Given p. H Useful for: - Ø Acid-base titrations EDTA titrations Electrochemical equilibria Combine Mass Balance and Equilibrium Constant Rearrange:

Polyprotic Acid-Base Equilibria Polyprotic Acids and Bases 4. ) Fractional Composition Equations Ø Combine Mass Balance and Equilibrium Constant Recall: fraction of molecule in the form HA is: Divide by F: Fraction in the form HA: Fraction in the form A-:

Polyprotic Acid-Base Equilibria Polyprotic Acids and Bases 4. ) Fractional Composition Equations Ø Ø Diprotic Systems Follows same process as monoprotic systems Fraction in the form H 2 A: Fraction in the form HA-: Fraction in the form A 2 -:

Polyprotic Acid-Base Equilibria Isoelectric and Isoionic p. H 1. ) Isoionic point – is the p. H obtained when the pure, neutral polyprotic acid HA is dissolved in water Ø Ø Neutral zwitterion Only ions are H 2 A+, A-, H+ and OHConcentrations are not equal to each other Isoionic point: p. H obtained by simply dissolving alanine Remember: Net Charge of Solution is Always Zero!

Polyprotic Acid-Base Equilibria Isoelectric and Isoionic p. H 2. ) Isoelectric point – is the p. H at which the average charge of the polyprotic acid is 0 Ø p. H at which [H 2 A+] = [A-] - Ø Ø Always some A- and H 2 A+ in equilibrium with HA Most of molecule is in uncharged HA form To go from isoionic point (all HA) to isoelectric point, add acid to decrease [A-] and increase [H 2 A+] until equal - p. K 1 < p. K 2 isoionic point is acidic excess [A-] Remember: Net Charge of Solution is Always Zero!

Polyprotic Acid-Base Equilibria Isoelectric and Isoionic p. H 2. ) Isoelectric point – is the p. H at which the average charge of the polyprotic acid is 0 Ø isoelectric point: [A-] = [H 2 A+] Isoelectric point:

Polyprotic Acid-Base Equilibria Isoelectric and Isoionic p. H 3. ) Example: Ø Determine isoelectric and isoionic p. H for 0. 10 M alanine (p. K 1 = 2. 34, p. K 2=9. 87). Solution: For isoionic point: For isoelectric point: Isoelectric and isoionic points for polyprotic acid are almost the same
- Slides: 36