POLYPHENOL COLORATION OBJECTIVES 1 Polyphenols classification and representatives

POLYPHENOL COLORATION OBJECTIVES 1. Polyphenols – classification and representatives; 2. Non-enzymatic polyphenol coloration; 3. Enzymatic polyphenol coloration;
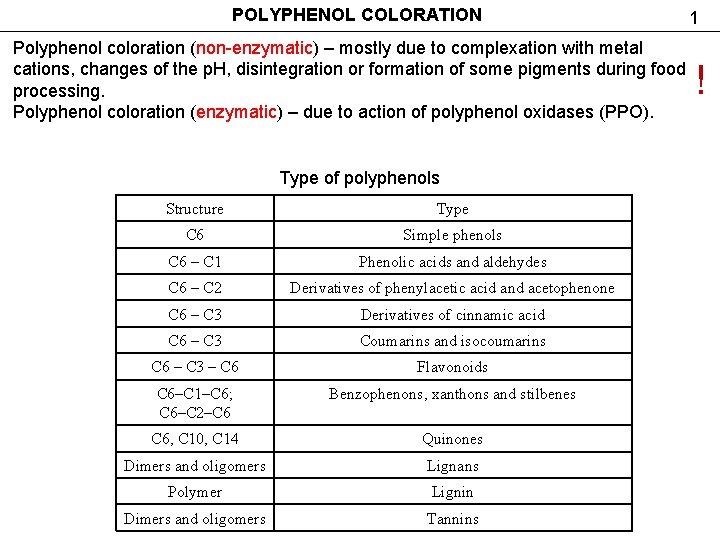
POLYPHENOL COLORATION 1 Polyphenol coloration (non-enzymatic) – mostly due to complexation with metal cations, changes of the р. Н, disintegration or formation of some pigments during food processing. Polyphenol coloration (enzymatic) – due to action of polyphenol oxidases (PPO). Type of polyphenols Structure Type С 6 Simple phenols С 6 – С 1 Phenolic acids and aldehydes С 6 – С 2 Derivatives of phenylacetic acid and acetophenone С 6 – С 3 Derivatives of cinnamic acid С 6 – С 3 Coumarins and isocoumarins С 6 – С 3 – С 6 Flavonoids С 6–С 1–С 6; С 6–С 2–С 6 Benzophenons, xanthons and stilbenes С 6, С 10, С 14 Quinones Dimers and oligomers Lignans Polymer Lignin Dimers and oligomers Tannins !
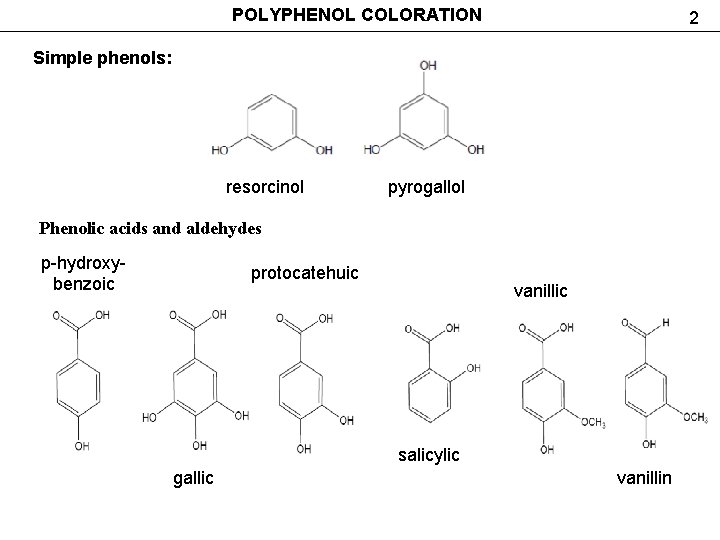
POLYPHENOL COLORATION 2 Simple phenols: resorcinol pyrogallol Phenolic acids and aldehydes р-hydroxybenzoic protocatehuic vanillic salicylic gallic vanillin
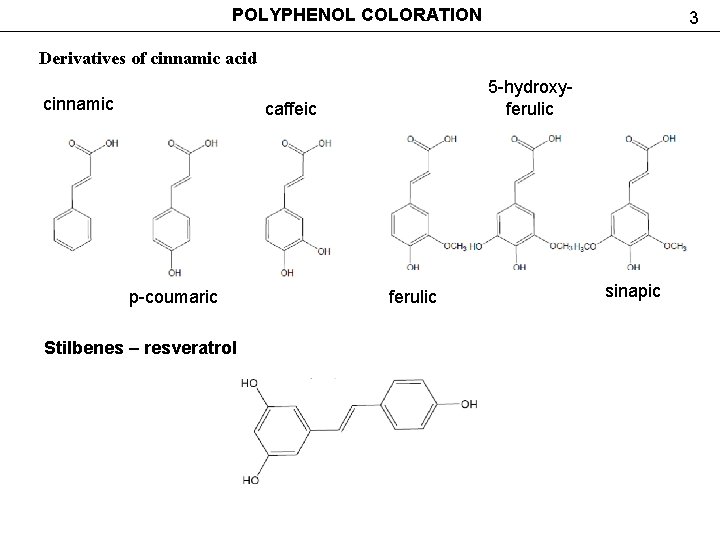
POLYPHENOL COLORATION 3 Derivatives of cinnamic acid cinnamic 5 -hydroxyferulic caffeic р-coumaric Stilbenes – resveratrol ferulic sinapic
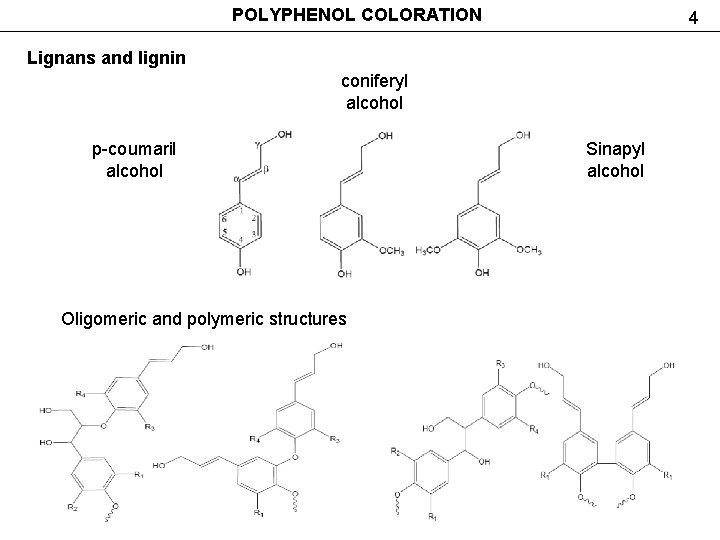
POLYPHENOL COLORATION 4 Lignans and lignin coniferyl alcohol р-coumaril alcohol Oligomeric and polymeric structures Sinapyl alcohol
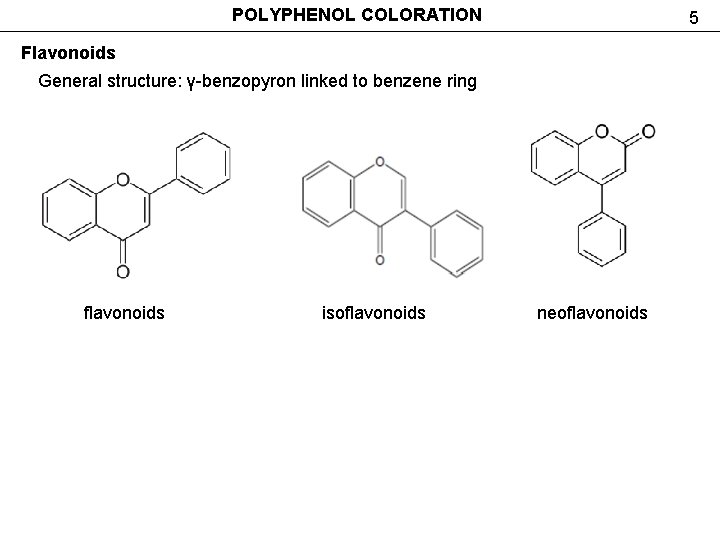
POLYPHENOL COLORATION 5 Flavonoids General structure: γ-benzopyron linked to benzene ring flavonoids isoflavonoids neoflavonoids
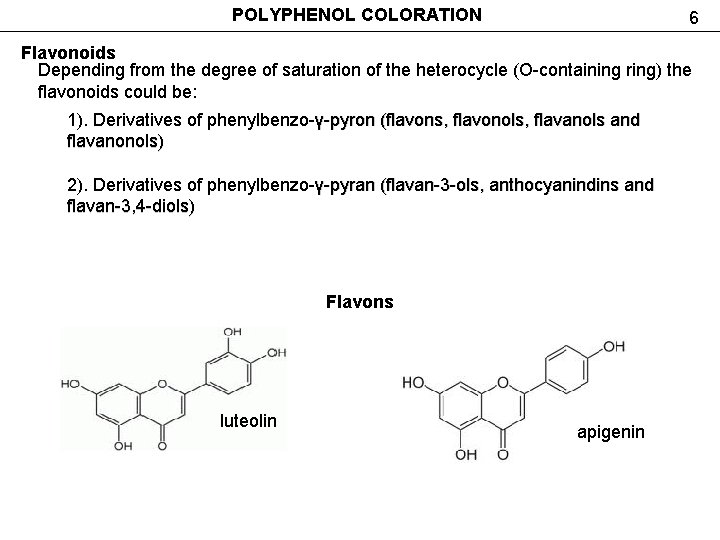
POLYPHENOL COLORATION 6 Flavonoids Depending from the degree of saturation of the heterocycle (O-containing ring) the flavonoids could be: 1). Derivatives of phenylbenzo-γ-pyron (flavons, flavonols, flavanols and flavanonols) 2). Derivatives of phenylbenzo-γ-pyran (flavan-3 -ols, anthocyanindins and flavan-3, 4 -diols) Flavons luteolin apigenin
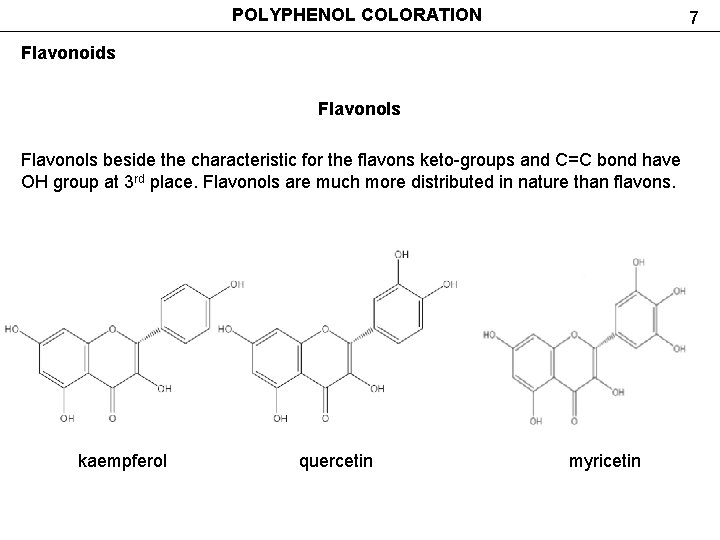
POLYPHENOL COLORATION 7 Flavonoids Flavonols beside the characteristic for the flavons keto-groups and C=C bond have OH group at 3 rd place. Flavonols are much more distributed in nature than flavons. kaempferol quercetin myricetin
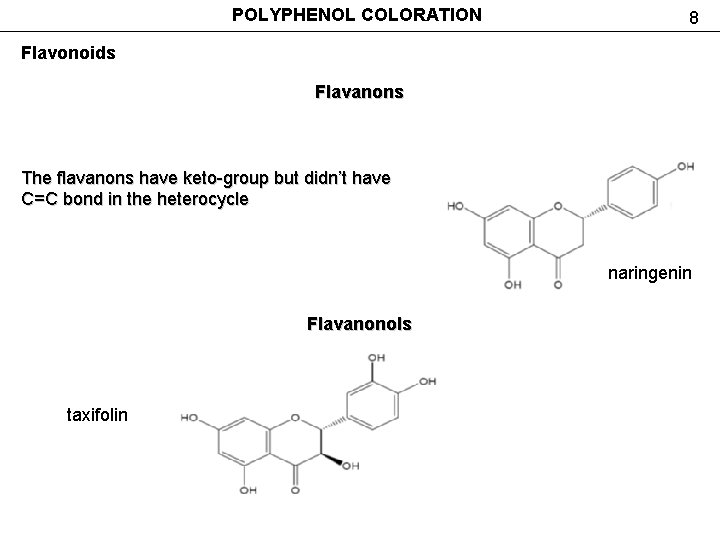
POLYPHENOL COLORATION 8 Flavonoids Flavanons The flavanons have keto-group but didn’t have C=C bond in the heterocycle naringenin Flavanonols taxifolin
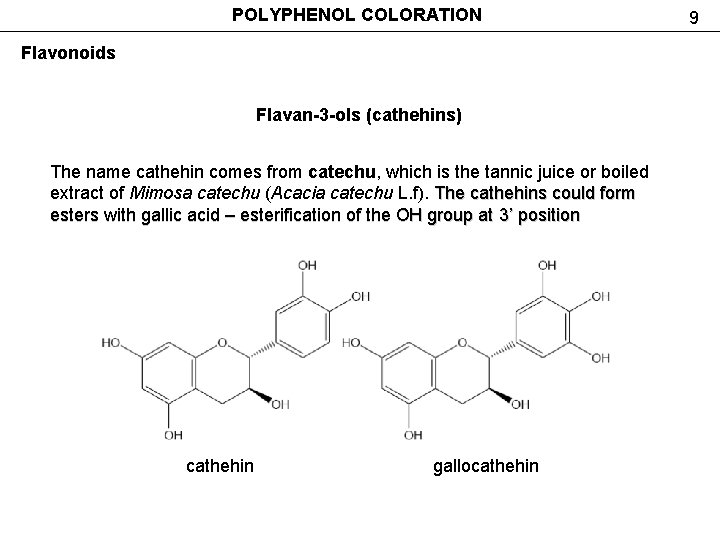
POLYPHENOL COLORATION Flavonoids Flavan-3 -ols (cathehins) The name cathehin comes from catechu, which is the tannic juice or boiled The name cathehin comes from extract of Mimosa catechu (Acacia catechu L. f). The cathehins could form esters with gallic acid – esterification of the OH group at 3’ position cathehin gallocathehin 9
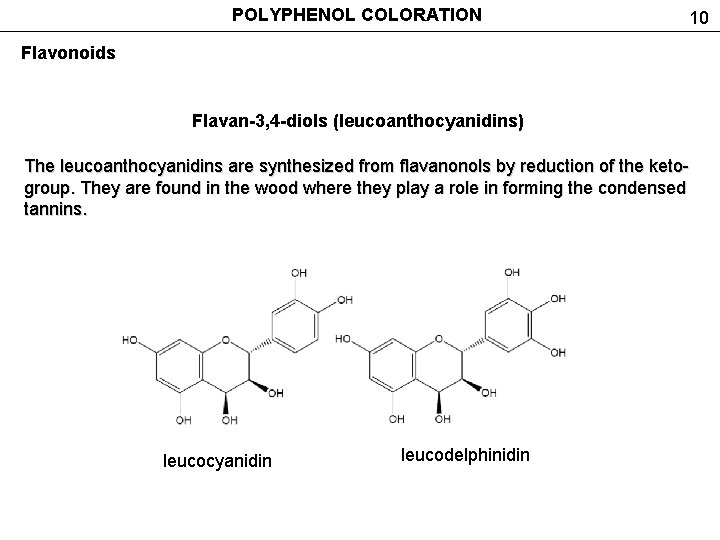
POLYPHENOL COLORATION 10 Flavonoids Flavan-3, 4 -diols (leucoanthocyanidins) The leucoanthocyanidins are synthesized from flavanonols by reduction of the ketogroup. They are found in the wood where they play a role in forming the condensed tannins. leucocyanidin leucodelphinidin
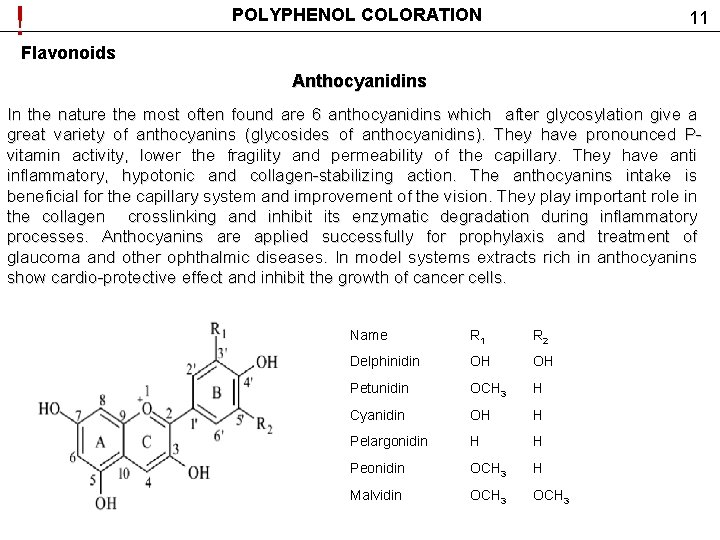
! POLYPHENOL COLORATION 11 Flavonoids Anthocyanidins In the nature the most often found are 6 anthocyanidins which after glycosylation give a great variety of anthocyanins (glycosides of anthocyanidins). They have pronounced Pvitamin activity, lower the fragility and permeability of the capillary. They have anti inflammatory, hypotonic and collagen-stabilizing action. The anthocyanins intake is beneficial for the capillary system and improvement of the vision. They play important role in the collagen crosslinking and inhibit its enzymatic degradation during inflammatory processes. Anthocyanins are applied successfully for prophylaxis and treatment of glaucoma and other ophthalmic diseases. In model systems extracts rich in anthocyanins show cardio-protective effect and inhibit the growth of cancer cells. Name R 1 R 2 Delphinidin OH OH Petunidin OCH 3 H Cyanidin OH H Pelargonidin H H Peonidin OCH 3 H Malvidin OCH 3

POLYPHENOL COLORATION Complexation and color formation The flavonoids – found in almost all fruits and vegetables. Complexation with metals like Fe 3+, Ca 2+, Al 3+, Sn 2+, Cu 2+ are the reason for coloration. Fe 3+ - the complexes with tannins are used for tannins measurements in beers. The observed coloration of potatoes after boiling – complexation of Fe 3+ with ophenols. Sometimes coloration of processed cauliflower – complexes of Fe 2+ kaempferol and quercetin. The canned cauliflower could give yellow colored complexes with tin. Formation of pink coloration (pinking) of pears and peaches after thermal treatments (and subsequent slow cooling) – due to formation of cyanidin complexes with copper, iron and zinc. More at CRC Handbook of Food Additives, Ed. Thomas E. Furia (2 nd edition), CRC Press 1972 NY. (pages 281 -283) 12

POLYPHENOL COLORATION Complexation and color formation Coloration of canned asparagus shoots (dark-greenish to black) – complexes of rutin (quercetin-3 -O-rutinoside) with Fe 2+ (EDTA or presence of Sn 2+ - eliminate the coloration problems). Generally use of sequestrants is the solution for complexation of polyphenols with metals and obtaining of unusual coloration or discoloration The formation of the color of ripe olives – dark-brown to black is due to formation of oxidative products of flavonoids (luteolin 7 -glycoside). Other compounds responsible: peonidin-3 -glucoside, cyanidin-3 -rhamnoglucoside (acylated with caffeic acide), hydroxytyrosol, vanillic acid, caffeic acid, pcoumaric acid etc. 13

POLYPHENOL COLORATION Changes of the color with change of the p. H – example: anthocyanins Anthocyanins of red cabbage: left to right – increase of the р. Н 14
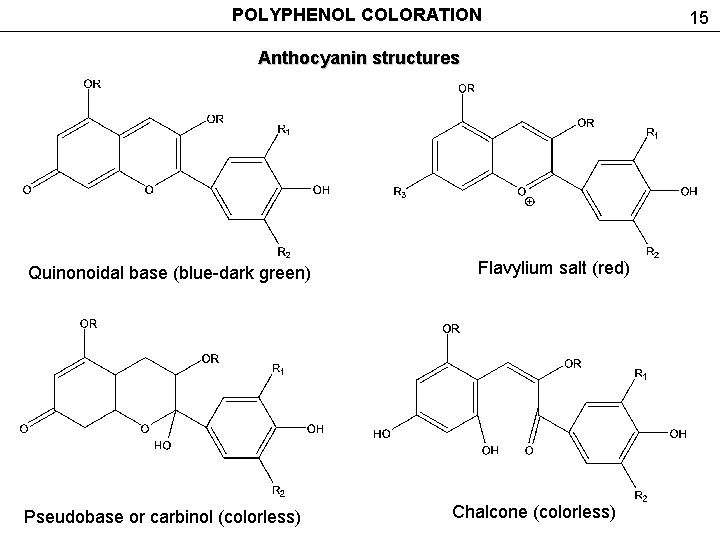
POLYPHENOL COLORATION Anthocyanin structures Quinonoidal base (blue-dark green) Pseudobase or carbinol (colorless) Flavylium salt (red) Chalcone (colorless) 15
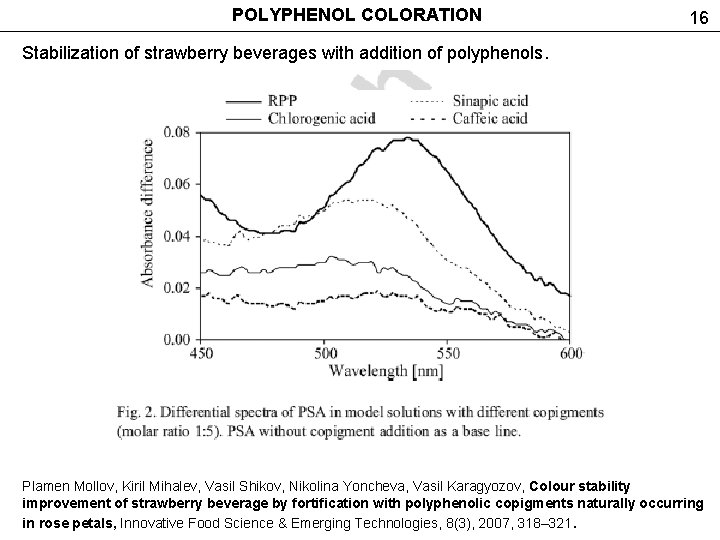
POLYPHENOL COLORATION 16 Stabilization of strawberry beverages with addition of polyphenols. Plamen Mollov, Kiril Mihalev, Vasil Shikov, Nikolina Yoncheva, Vasil Karagyozov, Colour stability improvement of strawberry beverage by fortification with polyphenolic copigments naturally occurring in rose petals, Innovative Food Science & Emerging Technologies, 8(3), 2007, 318– 321.
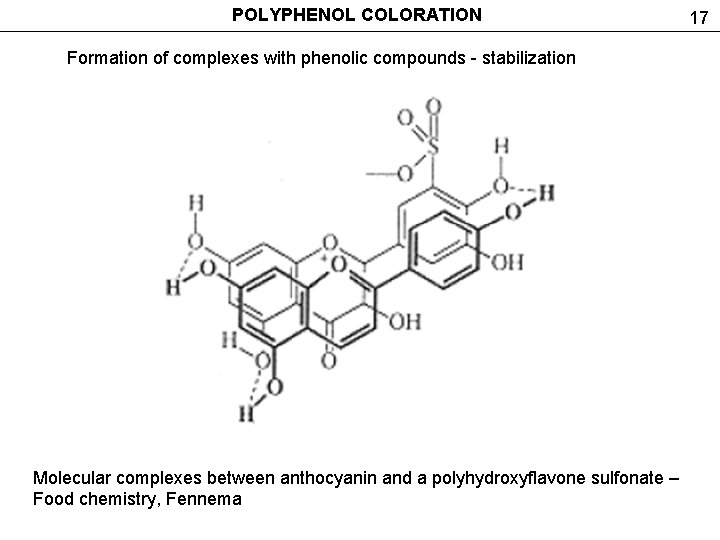
POLYPHENOL COLORATION Formation of complexes with phenolic compounds - stabilization Molecular complexes between anthocyanin and a polyhydroxyflavone sulfonate – Food chemistry, Fennema 17
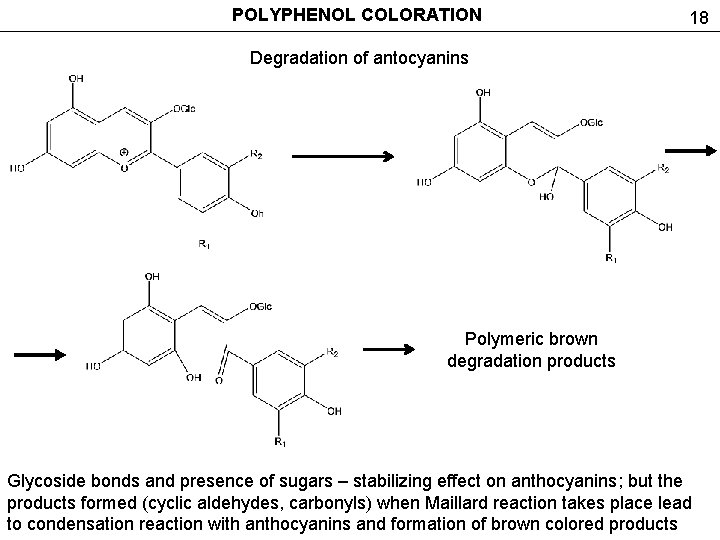
POLYPHENOL COLORATION 18 Degradation of antocyanins Polymeric brown degradation products Glycoside bonds and presence of sugars – stabilizing effect on anthocyanins; but the products formed (cyclic aldehydes, carbonyls) when Maillard reaction takes place lead to condensation reaction with anthocyanins and formation of brown colored products
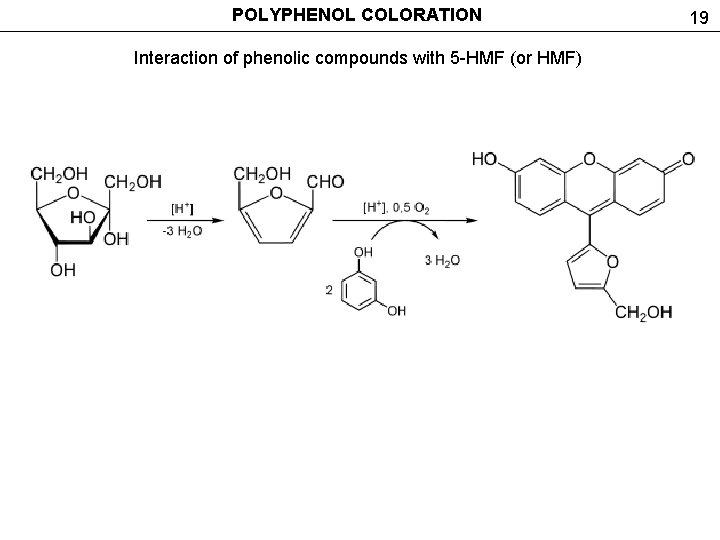
POLYPHENOL COLORATION Interaction of phenolic compounds with 5 -HMF (or HMF) 19

ENZYMATIC BROWNING 20
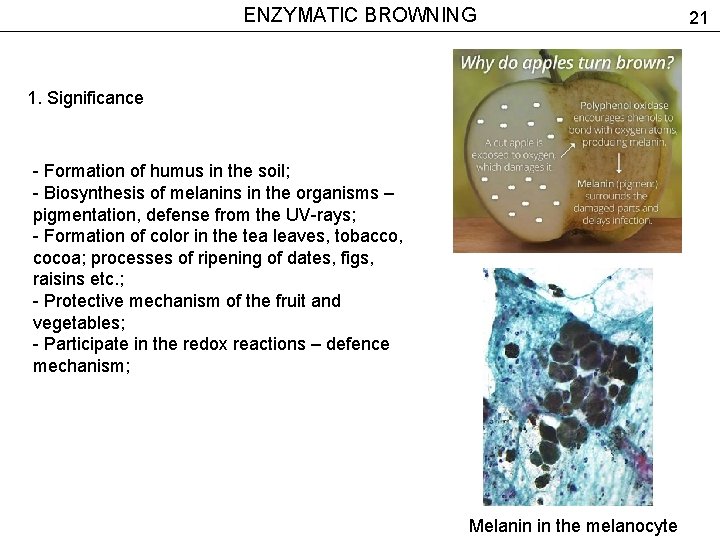
ENZYMATIC BROWNING 1. Significance - Formation of humus in the soil; - Biosynthesis of melanins in the organisms – pigmentation, defense from the UV-rays; - Formation of color in the tea leaves, tobacco, cocoa; processes of ripening of dates, figs, raisins etc. ; - Protective mechanism of the fruit and vegetables; - Participate in the redox reactions – defence mechanism; Melanin in the melanocyte 21

ENZYMATIC BROWNING 2. Mechanism of action Polyphenol oxidase (РРО) Substrates of the reaction – monophenols, L-tyrosine, hydroxy-derivatives of cinnamic acid (p-coumaric, caffeic, ferulic, chlorogenic acid) Catechol oxidase – catalyzes oxidation of o-diphenols to quinones. 22
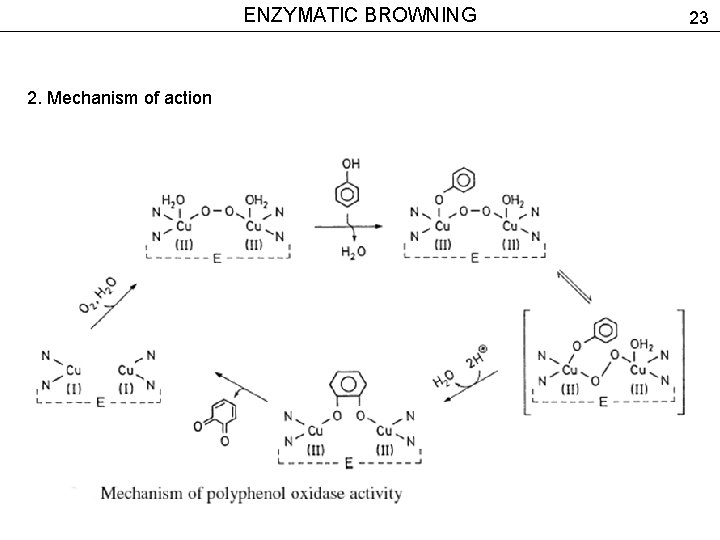
ENZYMATIC BROWNING 2. Mechanism of action 23
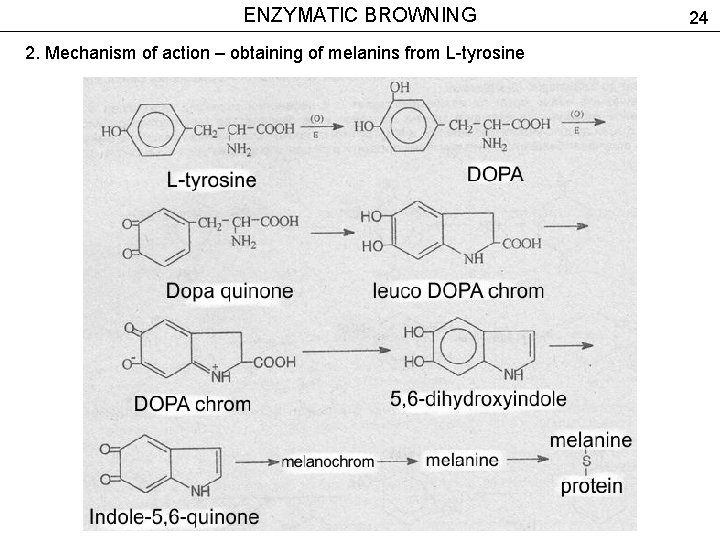
ENZYMATIC BROWNING 2. Mechanism of action – obtaining of melanins from L-tyrosine 24
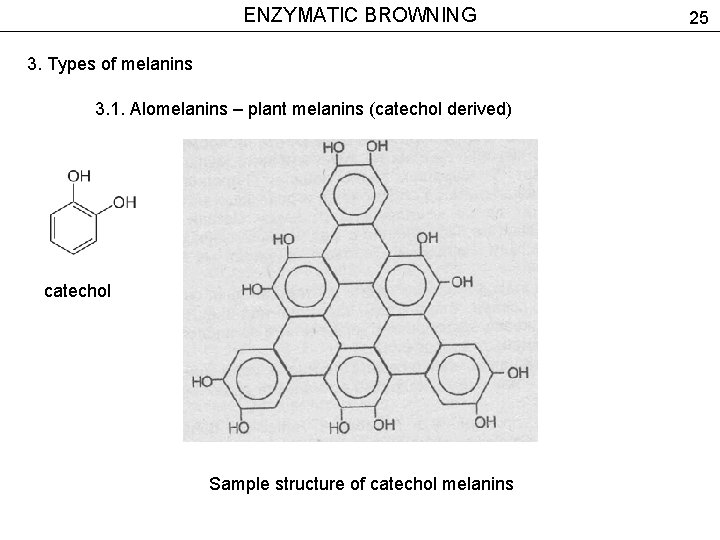
ENZYMATIC BROWNING 3. Types of melanins 3. 1. Alomelanins – plant melanins (catechol derived) catechol Sample structure of catechol melanins 25
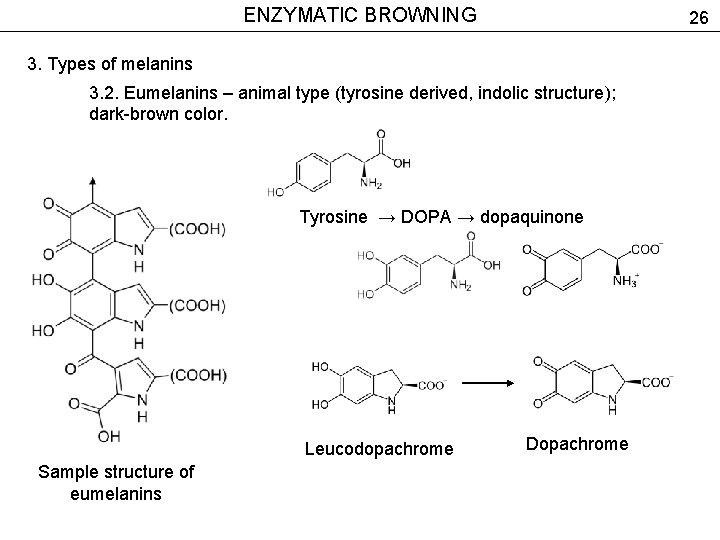
ENZYMATIC BROWNING 26 3. Types of melanins 3. 2. Eumelanins – animal type (tyrosine derived, indolic structure); dark-brown color. Tyrosine → DOPA → dopaquinone Leucodopachrome Sample structure of eumelanins Dopachrome
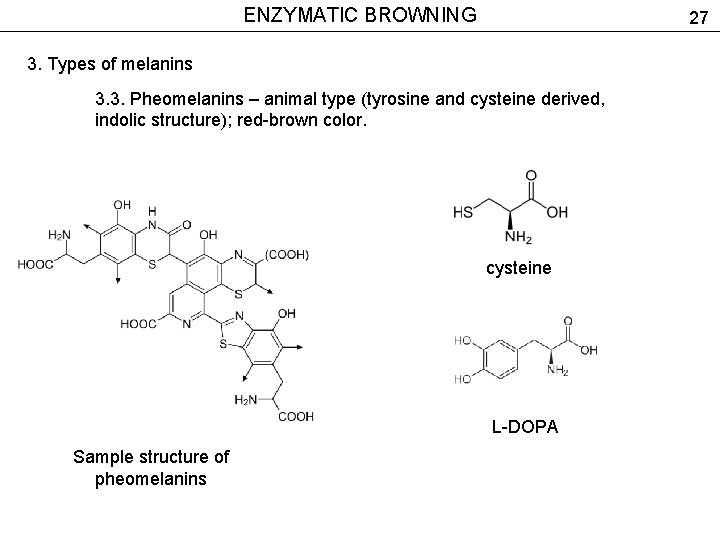
ENZYMATIC BROWNING 27 3. Types of melanins 3. 3. Pheomelanins – animal type (tyrosine and cysteine derived, indolic structure); red-brown color. cysteine L-DOPA Sample structure of pheomelanins

! ENZYMATIC BROWNING 4. Processes of enzymatic browning desired - fermentation of tea leaves; - fermentation of cocoa beans; - obtaining of sake; - obtaining of soy sauce. undesirable - discoloration of wounded tissues – as a result of microbial attack, wounding during picking, transportation, storage; - treatment of the raw materials – peeling, trimming, milling, etc. - chilling and freezing processes; - drying processes; - processing of crustaceans. inhibition - Tropolone ( grape polyphenol oxidase inhibitor); - Potassium metabisulfite, K 2 S 2 O 5; - Potassium dithionite or potassium hydrosulfite, K 2 S 2 O 4; 28
- Slides: 29