Polypharmacy Anticoagulation DAPT C Michael Gibson M S

Polypharmacy Anticoagulation & DAPT C. Michael Gibson, M. S. , M. D. Interventional Cardiologist Chief, Clinical Research, Beth Israel Deaconess CV Division Professor of Medicine Harvard Medical School President & CEO of Non-Profit Baim Institute & PERFUSE Study Group Founder, Editor-In-Chief www. wikidoc. org Harvard Medical School

Disclosure • Dr. Gibson has received research grant support and consulting fees in the past from all major manufacturers of antiplatelets and antithrombins • This is an educational lecture and is not intended to be an inducement to use any drug or drug in a fashion that is inconsistent with the drug or device label. Rivaroxaban is not approved for use in acute coronary syndromes in the US, but is so in many other countries • The slides were prepared by C. Michael Gibson, M. S. , M. D. and / or were under the editorial control of C. Michael Gibson, M. S. , M. D. The use of 2. 5 mg of Rivaroxaban is approved by the FDA for chronic coronary and/or peripheral artery disease when combined with aspirin. Rivaroxaban is not FDA approved for ACS. It is in many countries. Check your local label. Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019
Disclosures Present Research/Grant Funding Angel Medical Corporation Bayer Corp. / Janssen CSL Behring Portola Pharmaceuticals Baim Institute (no salary) Patents and Stocks: None Equity: nference, Inc. Consultant Boston Clinical Research Institute Cardiovascular Research Foundation Eli Lilly Medtelligence Novo Nordisk Pharma. Mar Portola Bayer / Janssen/ J&J Wed. MD Spouse: Employee of Boston Clinical Research Institute, she has equity position Amarin Amgen Bayer/Janssen/ J&J Boehringer Ingelheim Boston Scientific Cardiovascular Research Foundation CSL Behring Chiesi DCRI Eli Lilly Impact Bio, LTD Gilead Sciences, Inc. The Medicines Company Medtelligence Med. Immune The Medicine’s Co. Merck & Co. Inc. Microport Novo Nordisk PERT Consortium Pharma. Mar Portola Sanofi Somahlution Verseon Corporation The use of 2. 5 mg of Rivaroxaban is approved by the FDA for chronic coronary and/or peripheral artery disease when combined with aspirin. Rivaroxaban is not FDA approved for ACS. It is in many countries. Check your local label. Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019
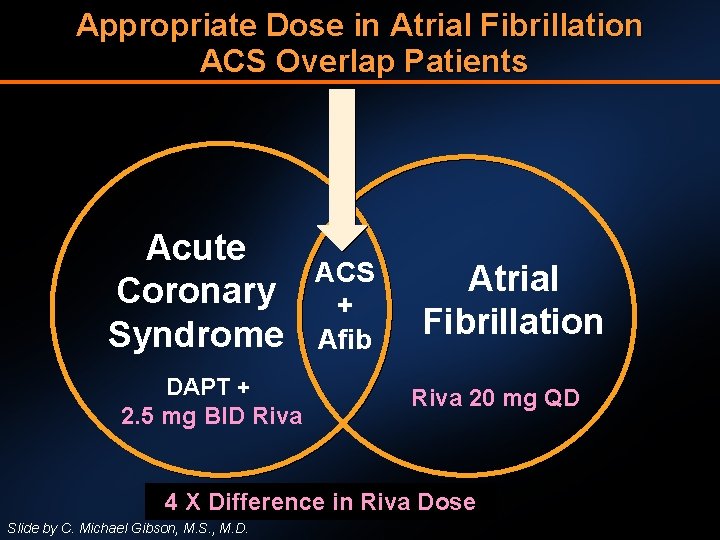
Appropriate Dose in Atrial Fibrillation ACS Overlap Patients Acute Coronary Syndrome DAPT + 2. 5 mg BID Riva ACS + Afib Atrial Fibrillation Riva 20 mg QD 4 X Difference in Riva Dose Slide by C. Michael Gibson, M. S. , M. D.

Bewildering Number of Strategies in the ACS Patient with Atrial Fibrillation • • • ASA Dose: None Low High 2 ASA Duration (mos): 1 3 6 12 4 Thineopyridine: None Clop Ticlid Pras Ticag 4 Thienopyridine duration (mos): 1 3 6 12 4 AC: None Warf Dabi Riva Apix Edox 5 AC INR/Dose: Low High 2 1+8 = 9 ASA 1+16 = 17 Thieno 1+10 = 11 ACs Permutations of Single, Dual or Triple Therapy as Early Initial Therapy (0, 1, 3, 6 mos) following ACS: 9 X 17 X 11 = 1, 683 Permutations of Single or Dual Therapy Late After Early Therapy (0, 1, 3, 6 mos) following ACS: 1, 683 Total Permutations throughout one year: 2. 8 Million Slide by C. Michael Gibson, M. S. , M. D.

Ways to Reduce Bleeding With Triple Therapy Replace warfarin with alternate anticoagulant Reduce dosing of anticoagulant Drop an antiplatelet agent All of the above Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. AHA 2016
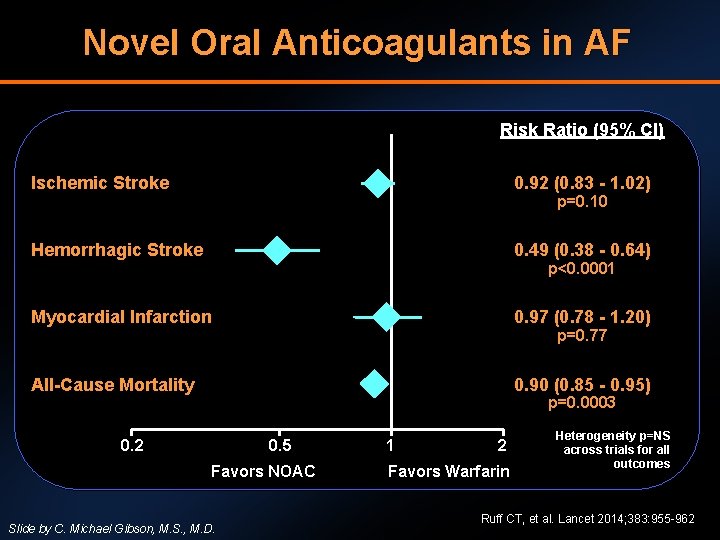
Novel Oral Anticoagulants in AF Risk Ratio (95% CI) Ischemic Stroke 0. 92 (0. 83 - 1. 02) Hemorrhagic Stroke 0. 49 (0. 38 - 0. 64) Myocardial Infarction 0. 97 (0. 78 - 1. 20) All-Cause Mortality 0. 90 (0. 85 - 0. 95) p=0. 10 p<0. 0001 p=0. 77 p=0. 0003 0. 2 0. 5 Favors NOAC Slide by C. Michael Gibson, M. S. , M. D. 1 2 Favors Warfarin Heterogeneity p=NS across trials for all outcomes Ruff CT, et al. Lancet 2014; 383: 955 -962
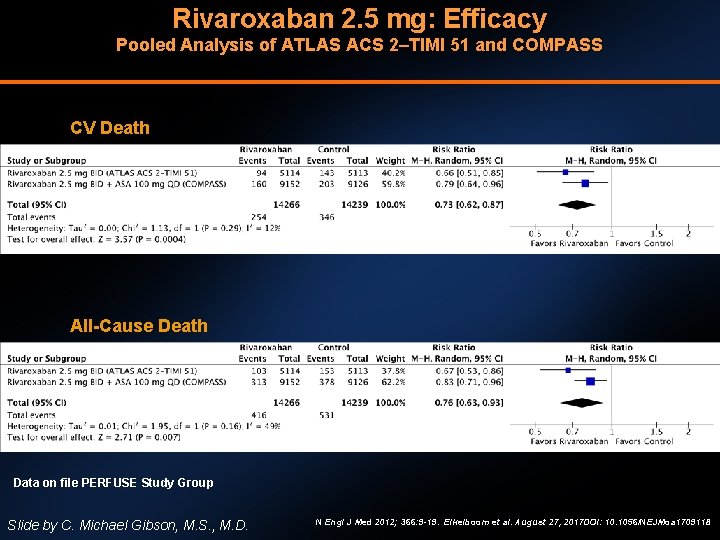
Rivaroxaban 2. 5 mg: Efficacy Pooled Analysis of ATLAS ACS 2–TIMI 51 and COMPASS CV Death All-Cause Death Data on file PERFUSE Study Group Slide by C. Michael Gibson, M. S. , M. D. N Engl J Med 2012; 366: 9 -19. Eikelboom et al. August 27, 2017 DOI: 10. 1056/NEJMoa 1709118
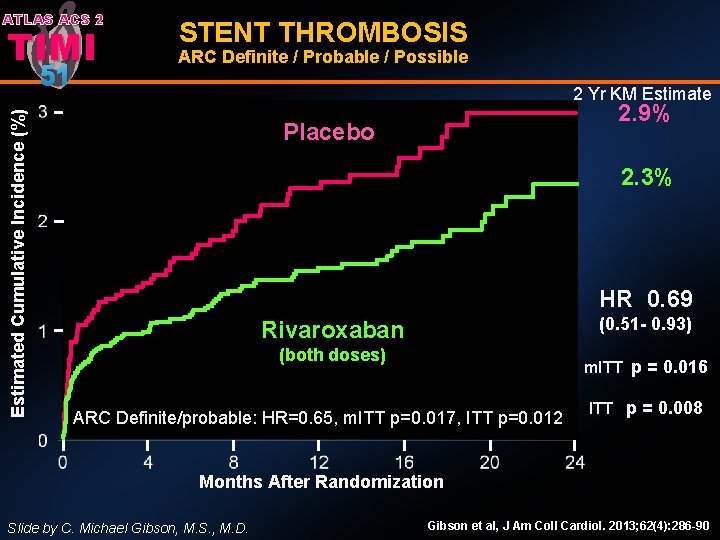
ATLAS ACS 2 TIMI Estimated Cumulative Incidence (%) 51 STENT THROMBOSIS ARC Definite / Probable / Possible 2 Yr KM Estimate 2. 9% Placebo 2. 3% HR 0. 69 (0. 51 - 0. 93) Rivaroxaban (both doses) m. ITT p = 0. 016 ARC Definite/probable: HR=0. 65, m. ITT p=0. 017, ITT p=0. 012 ITT p = 0. 008 Months After Randomization Slide by C. Michael Gibson, M. S. , M. D. Gibson et al, J Am Coll Cardiol. 2013; 62(4): 286 -90
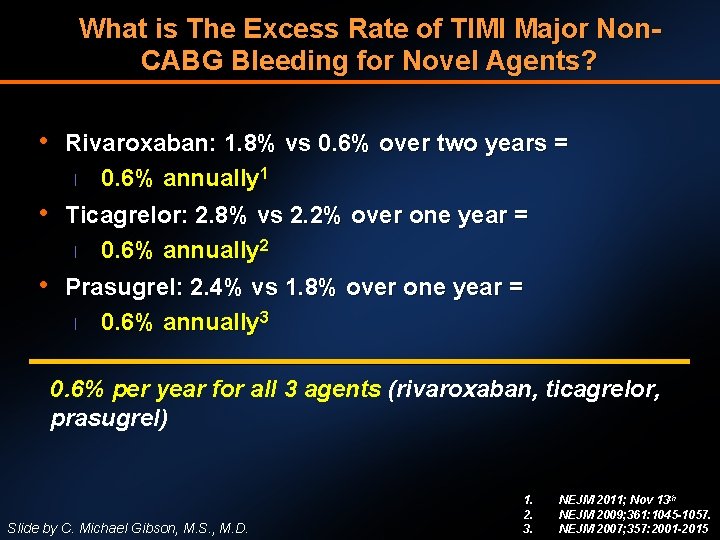
What is The Excess Rate of TIMI Major Non. CABG Bleeding for Novel Agents? • Rivaroxaban: 1. 8% vs 0. 6% over two years = l 0. 6% annually 1 • Ticagrelor: 2. 8% vs 2. 2% over one year = l 0. 6% annually 2 • Prasugrel: 2. 4% vs 1. 8% over one year = l 0. 6% annually 3 0. 6% per year for all 3 agents (rivaroxaban, ticagrelor, prasugrel) Slide by C. Michael Gibson, M. S. , M. D. 1. 2. 3. NEJM 2011; Nov 13 th NEJM 2009; 361: 1045 -1057. NEJM 2007; 357: 2001 -2015
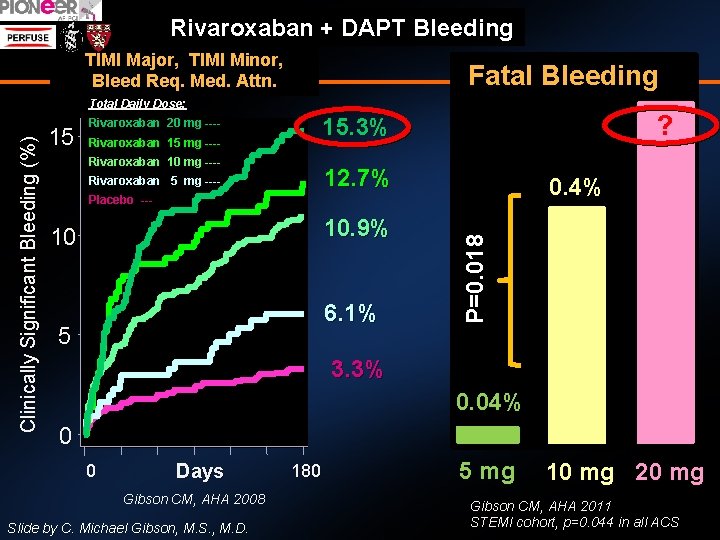
Rivaroxaban + DAPT Bleeding TIMI Major, TIMI Minor, Bleed Req. Med. Attn. Fatal Bleeding 15 ? 15. 3% Rivaroxaban 20 mg ---Rivaroxaban 15 mg ---Rivaroxaban 10 mg ---- 12. 7% Rivaroxaban 5 mg ---- 0. 4% Placebo --- 10. 9% 10 6. 1% 5 P=0. 018 Clinically Significant Bleeding (%) Total Daily Dose: 3. 3% 0. 04% 0 0 Days Gibson CM, AHA 2008 Slide by C. Michael Gibson, M. S. , M. D. 180 5 mg 10 mg 20 mg Gibson CM, AHA 2011 STEMI cohort, p=0. 044 in all ACS
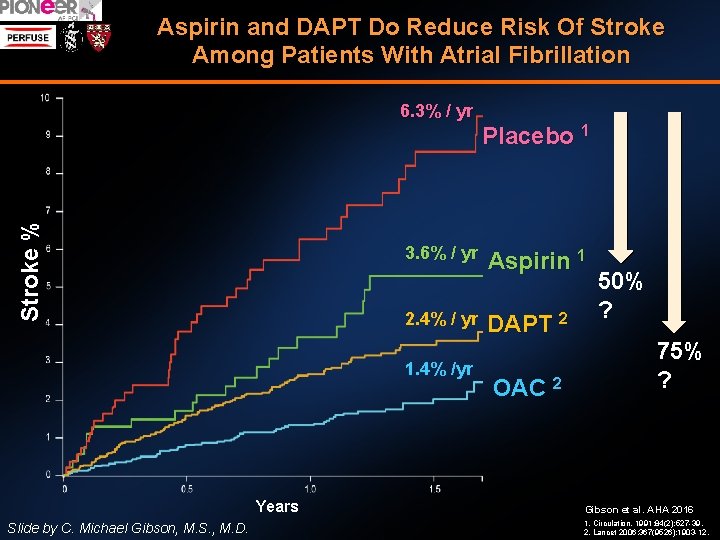
Aspirin and DAPT Do Reduce Risk Of Stroke Among Patients With Atrial Fibrillation Stroke % 6. 3% / yr 3. 6% / yr 2. 4% / yr 1. 4% /yr Years Slide by C. Michael Gibson, M. S. , M. D. Placebo 1 Aspirin 1 DAPT 2 OAC 2 50% ? 75% ? Gibson et al. AHA 2016 1. Circulation. 1991; 84(2): 527 -39. 2. Lancet 2006; 367(9526): 1903 -12.

Rivaroxaban and Ticagrelor Work Synergistically to Inhibit Thrombin Mediated Platelet Aggregation Synergistic Effect: 20% + 17% would be expected to yield 37% not 52% inhibition Slide by C. Michael Gibson, M. S. , M. D. Perzborn et al, J Cardiovasc Pharmacol Ther. 2015 Apr 6.
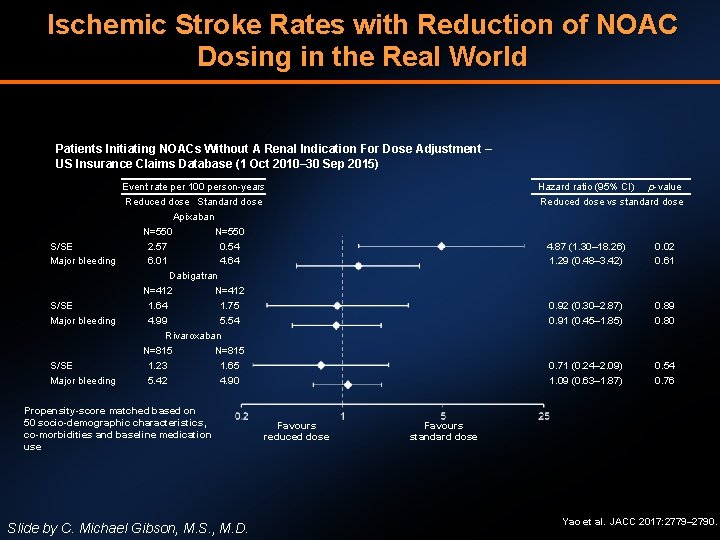
Ischemic Stroke Rates with Reduction of NOAC Dosing in the Real World Patients Initiating NOACs Without A Renal Indication For Dose Adjustment – US Insurance Claims Database (1 Oct 2010– 30 Sep 2015) Event rate per 100 person-years Reduced dose Standard dose Apixaban N=550 S/SE 2. 57 0. 54 Major bleeding 6. 01 4. 64 Dabigatran N=412 S/SE 1. 64 1. 75 Major bleeding 4. 99 5. 54 Rivaroxaban N=815 S/SE 1. 23 1. 65 Major bleeding 5. 42 4. 90 Propensity-score matched based on 50 socio-demographic characteristics, co-morbidities and baseline medication use Slide by C. Michael Gibson, M. S. , M. D. Favours reduced dose Hazard ratio (95% CI) p-value Reduced dose vs standard dose 4. 87 (1. 30– 18. 26) 1. 29 (0. 48– 3. 42) 0. 02 0. 61 0. 92 (0. 30– 2. 87) 0. 91 (0. 45– 1. 85) 0. 89 0. 80 0. 71 (0. 24– 2. 09) 1. 09 (0. 63– 1. 87) 0. 54 0. 76 Favours standard dose Yao et al. JACC 2017: 2779– 2790.
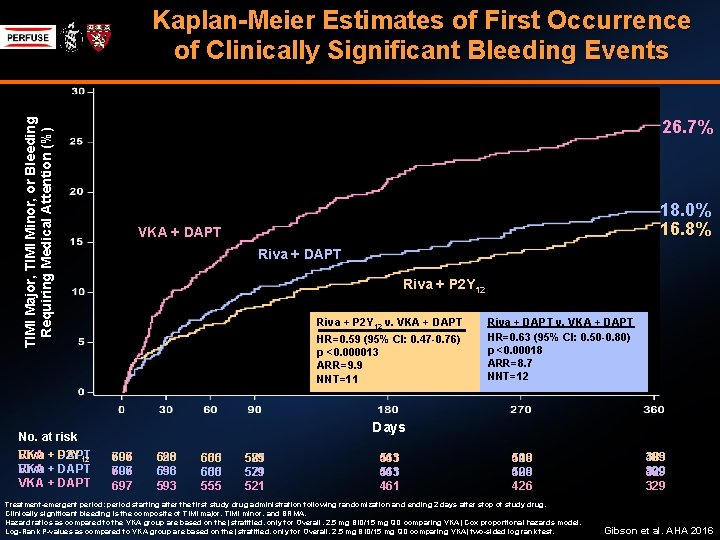
TIMI Major, TIMI Minor, or Bleeding Requiring Medical Attention (%) Kaplan-Meier Estimates of First Occurrence of Clinically Significant Bleeding Events 26. 7% p<0. 00018 p<0. 000013 18. 0% 16. 8% VKA + DAPT Riva + P 2 Y 12 HR = 0. 63 (95% CI 0. 50 -0. 80) ARR = 8. 7 HR = 0. 59 (95% CI 0. 47 -0. 76) Riva + P 2 Y 12 v. VKA + DAPT Riva + DAPT v. VKA + DAPT NNT = 12 HR=0. 63 (95% CI: 0. 50 -0. 80) HR=0. 59 (95% CI: 0. 47 -0. 76) ARR = 9. 9 p <0. 00018 p <0. 000013 NNT = 11 ARR=8. 7 ARR=9. 9 NNT=12 NNT=11 No. at risk VKA + DAPT Riva + P 2 Y 12 VKA + DAPT Riva + DAPT VKA + DAPT Days 697 706 697 593 636 628 593 636 593 555 600 606 555 600 555 521 579 585 521 579 521 461 543 461 426 509 510 426 509 426 Treatment-emergent period: period starting after the first study drug administration following randomization and ending 2 days after stop of study drug. Clinically significant bleeding is the composite of TIMI major, TIMI minor, and BRMA. Hazard ratios as compared to the VKA group are based on the (stratified, only for Overall, 2. 5 mg BID/15 mg QD comparing VKA) Cox proportional hazards model. Log-Rank P-values as compared to VKA group are based on the (stratified, only for Overall, 2. 5 mg BID/15 mg QD comparing VKA) two-sided log rank test. 329 409 383 329 409 329 Gibson et al. AHA 2016
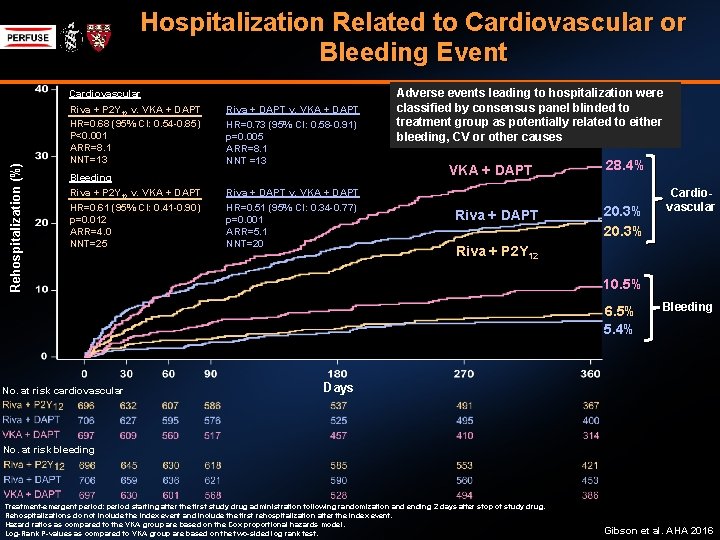
Hospitalization Related to Cardiovascular or Bleeding Event Rehospitalization (%) Cardiovascular Riva + P 2 Y 12 v. VKA + DAPT HR=0. 68 (95% CI: 0. 54 -0. 85) P<0. 001 ARR=8. 1 NNT=13 Riva + DAPT v. VKA + DAPT HR=0. 73 (95% CI: 0. 58 -0. 91) p=0. 005 ARR=8. 1 NNT =13 Bleeding Riva + P 2 Y 12 v. VKA + DAPT HR=0. 61 (95% CI: 0. 41 -0. 90) p=0. 012 ARR=4. 0 NNT=25 Riva + DAPT v. VKA + DAPT HR=0. 51 (95% CI: 0. 34 -0. 77) p=0. 001 ARR=5. 1 NNT=20 Adverse events leading to hospitalization were classified by consensus panel blinded to treatment group as potentially related to either bleeding, CV or other causes VKA + DAPT Riva + DAPT 28. 4% 20. 3% Riva + P 2 Y 12 10. 5% 6. 5% 5. 4% No. at risk cardiovascular Cardiovascular Bleeding Days No. at risk bleeding Treatment-emergent period: period starting after the first study drug administration following randomization and ending 2 days after stop of study drug. Rehospitalizations do not include the index event and include the first rehospitalization after the index event. Hazard ratios as compared to the VKA group are based on the Cox proportional hazards model. Log-Rank P-values as compared to VKA group are based on the two-sided log rank test. Gibson et al. AHA 2016
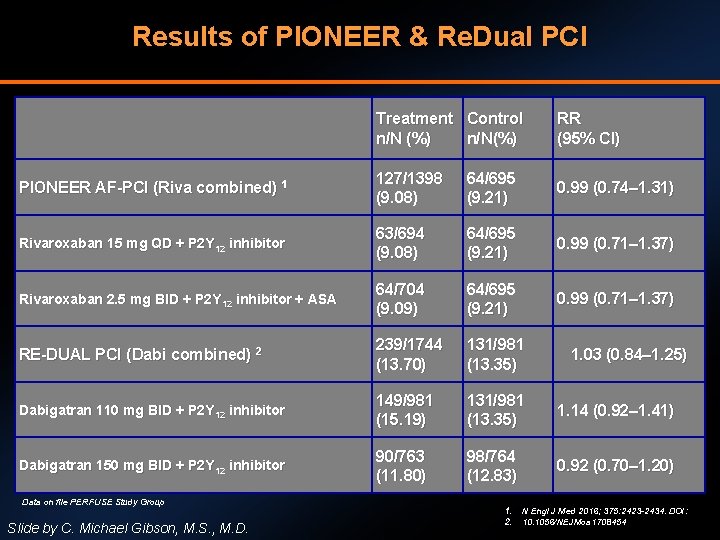
Results of PIONEER & Re. Dual PCI Treatment Control n/N (%) n/N(%) RR (95% CI) PIONEER AF-PCI (Riva combined) 1 127/1398 (9. 08) 64/695 (9. 21) 0. 99 (0. 74– 1. 31) Rivaroxaban 15 mg QD + P 2 Y 12 inhibitor 63/694 (9. 08) 64/695 (9. 21) 0. 99 (0. 71– 1. 37) Rivaroxaban 2. 5 mg BID + P 2 Y 12 inhibitor + ASA 64/704 (9. 09) 64/695 (9. 21) 0. 99 (0. 71– 1. 37) RE-DUAL PCI (Dabi combined) 2 239/1744 (13. 70) 131/981 (13. 35) Dabigatran 110 mg BID + P 2 Y 12 inhibitor 149/981 (15. 19) 131/981 (13. 35) 1. 14 (0. 92– 1. 41) Dabigatran 150 mg BID + P 2 Y 12 inhibitor 90/763 (11. 80) 98/764 (12. 83) 0. 92 (0. 70– 1. 20) Data on file PERFUSE Study Group Slide by C. Michael Gibson, M. S. , M. D. 1. 2. 1. 03 (0. 84– 1. 25) N Engl J Med 2016; 375: 2423 -2434. DOI: 10. 1056/NEJMoa 1708454
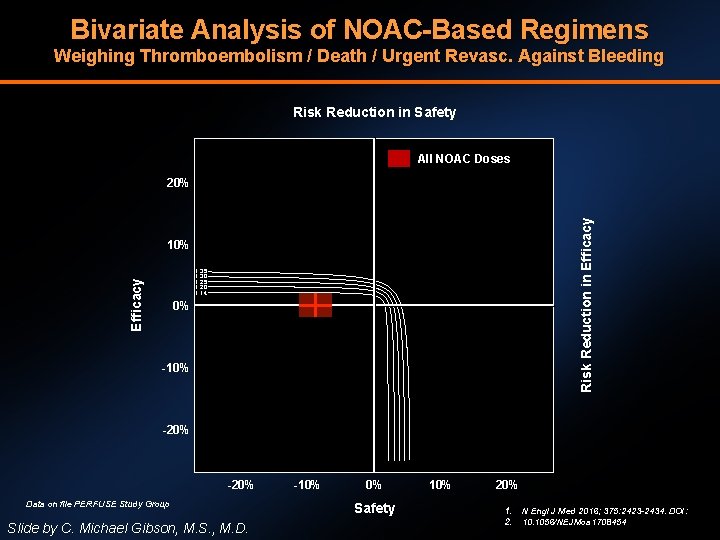
Bivariate Analysis of NOAC-Based Regimens Weighing Thromboembolism / Death / Urgent Revasc. Against Bleeding Risk Reduction in Safety All NOAC Doses Risk Reduction in Efficacy 20% 10% Efficacy 1. 35 1. 30 1. 25 1. 20 1. 14 0% -10% -20% Data on file PERFUSE Study Group Slide by C. Michael Gibson, M. S. , M. D. -10% 0% Safety 10% 20% 1. 2. N Engl J Med 2016; 375: 2423 -2434. DOI: 10. 1056/NEJMoa 1708454

2017 ESC Guidelines Update The duration of triple therapy should be limited or omitted after hospital discharge taking into account the ischaemic risk and bleeding risk Slide by C. Michael Gibson, M. S. , M. D. Valgimigli M. ESC 2017, Updated Guidelines

Ischemic Risk Factors Slide by C. Michael Gibson, M. S. , M. D. Valgimigli M. ESC 2017, Updated Guidelines

Bleeding Risk Factors & Strategies To Reduce Bleeding Slide by C. Michael Gibson, M. S. , M. D. Valgimigli M. ESC 2017, Updated Guidelines

2017 ESC Guidelines Update: Use of NOACs Slide by C. Michael Gibson, M. S. , M. D. Valgimigli M. ESC 2017, Updated Guidelines

Summary of key changes between 2016 and 2018 Expert Consensus on Antithrombotic Management of AF patients undergoing PCI Choice of anticoagulant Choice of P 2 Y 12 inhibitor Strategy (double vs triple therapy) 2016 Expert Consensus VKA or NOAC may be both considered, with choice of agent at the discretion of the treating physician and taking into consideration patient preference Clopidogrel is the P 2 Y 12 inhibitor of choice; avoid prasugrel or ticagrelor 2018 Expert Consensus A NOAC (rather than a VKA) should generally be preferred in most patients unless contraindicated Clopidogrel is the P 2 Y 12 inhibitor of choice; ticagrelor may represent a reasonable treatment option in patients at high ischemic/thrombotic and low bleeding risk; avoid prasugrel DAPT in adjunct to OAC (i. e. , triple. A double-therapy regimen (OAC therapy) should not extend to a full 12 plus P 2 Y 12 inhibitor) immediately months; consider SAPT (preferably after hospital discharge should be clopidogrel and dropping aspirin) in considered for most patients, while adjunct to OAC (i. e. , double-therapy) extending the use of aspirin beyond as early as possible (0 to 6 months hospital discharge (i. e. , triplepost-stenting) depending on therapy) should be considered only ischemic/thrombotic and bleeding for patients at high risk profile ischemic/thrombotic and low bleeding risks and for a limited period of time (e. g. , 1 month)
- Slides: 23