Polymers large molecules made by linking smaller single

Polymers �large molecules made by linking smaller, single unit (monomers) together �Many natural polymers form important organic molecules such as DNA, and proteins �Synthetic polymers are also often made into plastics and textiles such as nylon, teflon, PVC, polyethene, etc.

Polymerization �formation of polymers from the reaction of monomers �May occur in nature, however, manufacturers have learned how to synthetically produce many types of useful polymers.
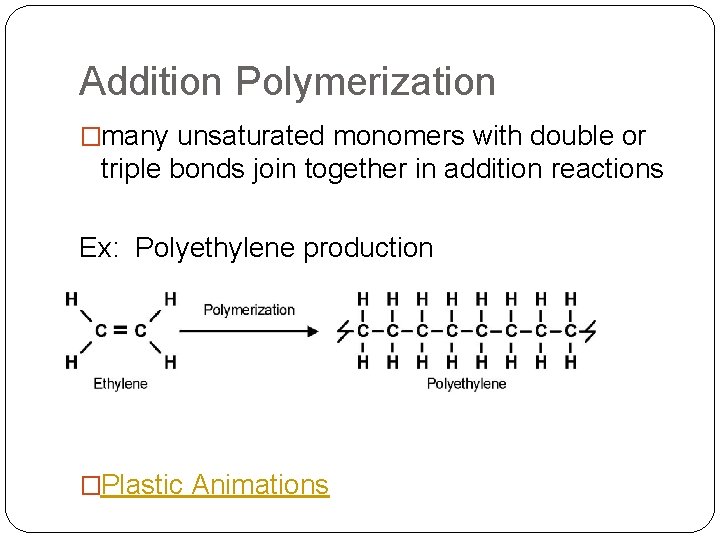
Addition Polymerization �many unsaturated monomers with double or triple bonds join together in addition reactions Ex: Polyethylene production �Plastic Animations
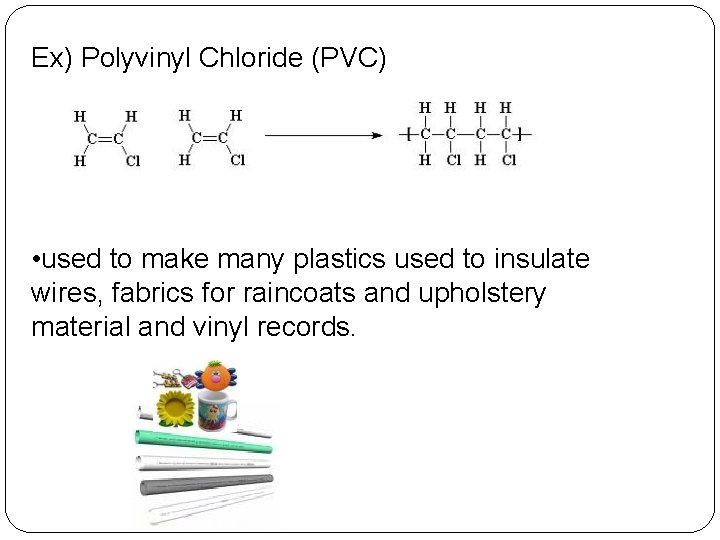
Ex) Polyvinyl Chloride (PVC) • used to make many plastics used to insulate wires, fabrics for raincoats and upholstery material and vinyl records.
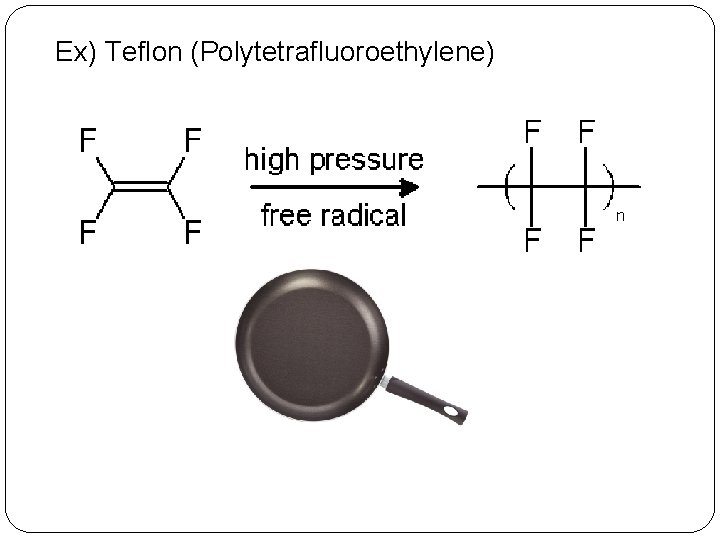
Ex) Teflon (Polytetrafluoroethylene)
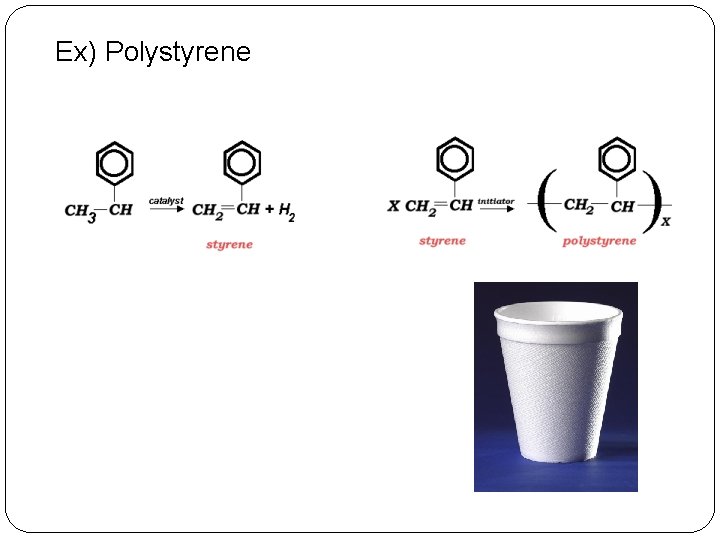
Ex) Polystyrene

Condensation Polymers �involve the condensing or removal of a small molecule such as H 2 O, NH 3 or HCl from the functional groups of two different monomers to form a polymer
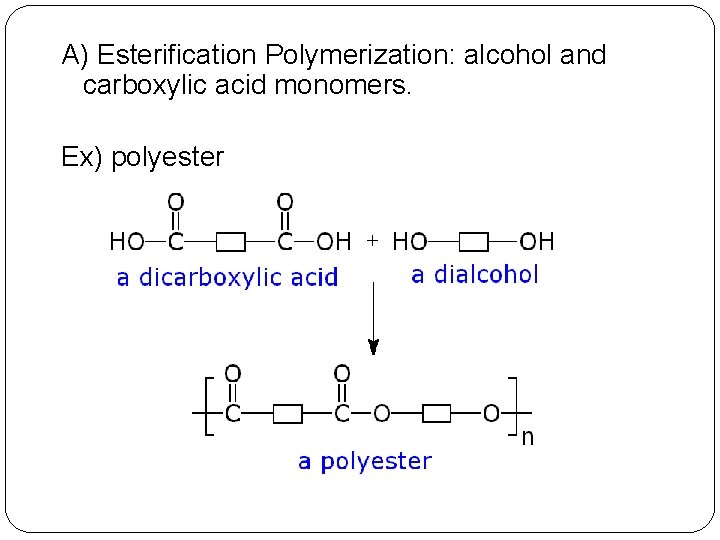
A) Esterification Polymerization: alcohol and carboxylic acid monomers. Ex) polyester
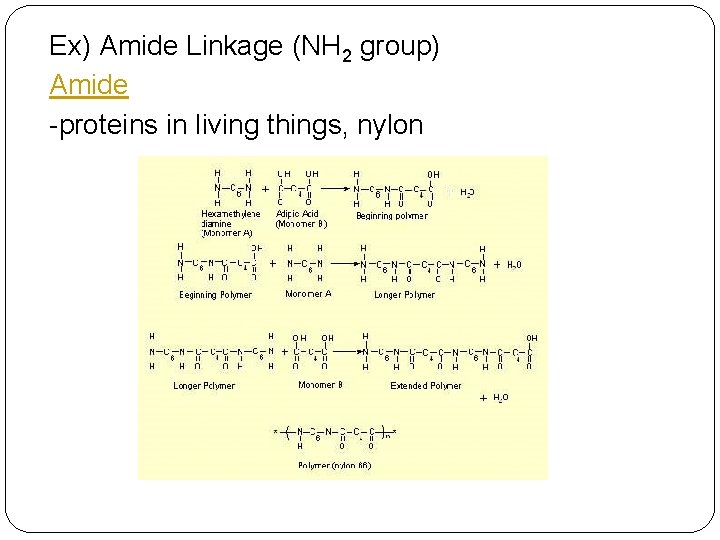
Ex) Amide Linkage (NH 2 group) Amide -proteins in living things, nylon
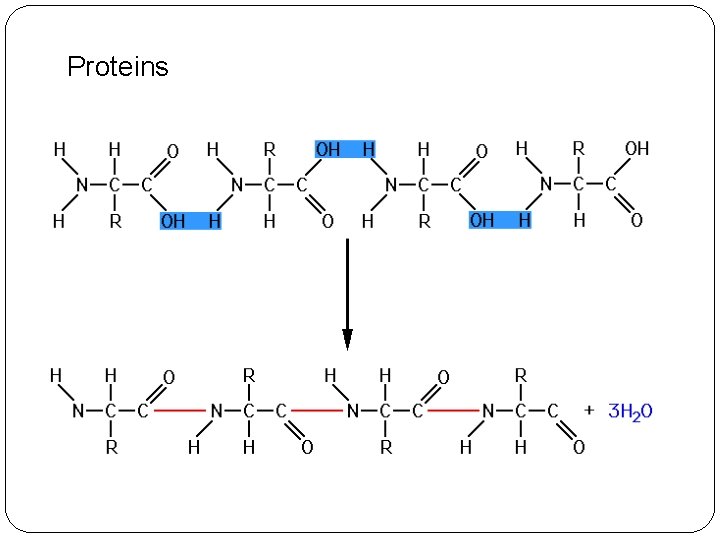
Proteins

Natural Polymers �Lipids –glycerol and fatty acids monomers �Proteins – amino acid monomers �Carbohydrates –monosaccharides (sugar) monomers �Cellulose-glucose monomers �Wood, paper, cotton �Animation Gallery

Synthetic Polymers �Polyesters �Polyamides (nylon) �Kevlar

Videos �Proteins: Structure and Function (Complete Video) �Saturated and Unsaturated Fats and Oils �Synthesis of Organic Compounds �Pharmaceuticals �Petrochemical Industry �The Age of Polymers (Complete Video) �Plastic Videos
- Slides: 13