Polycystic Ovary Syndrome Julie Bleyenberg DO Dover Womens

Polycystic Ovary Syndrome Julie Bleyenberg, DO Dover Women’s Health August 14, 2015

Polycystic Ovary Syndrome (PCOS) ► Heterogeneous disorder of unclear etiology ► One of the most common endocrine disorders in reproductive aged women ► Diagnosis § § of exclusion Congenital adrenal hyperplasia Cushing's disease Hyperprolactinemia Thyroid disease

Polycystic Ovary Syndrome Variable spectrum of symptoms/conditions ► Hyperandrogenism ► ► hirsutism, severe acne, male pattern alopecia Diagnostic criteria (2 out of 3) * * ► Ovulatory dysfunction ► Androgen excess ► Polycystic ovaries Menstrual irregularity § oligo or amenorrhea, irregular bleeding ► Polycystic ovaries ► Central obesity * * Currently the most widely accepted criteria
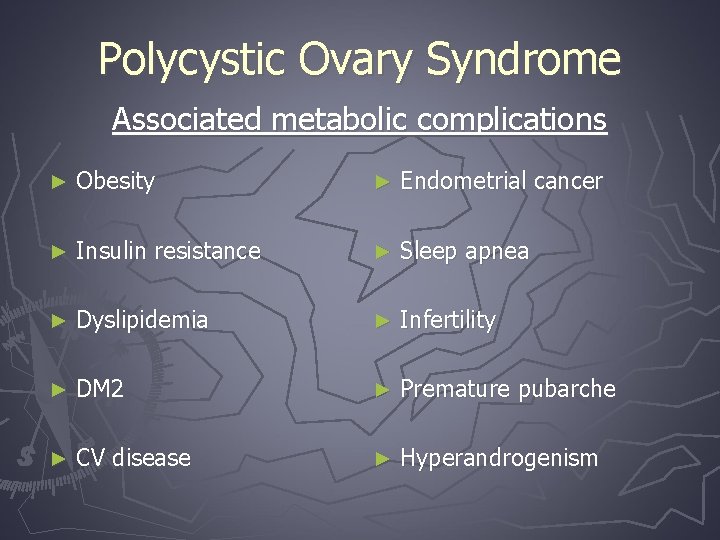
Polycystic Ovary Syndrome Associated metabolic complications ► Obesity ► Endometrial cancer ► Insulin resistance ► Sleep apnea ► Dyslipidemia ► Infertility ► DM 2 ► Premature pubarche ► CV disease ► Hyperandrogenism

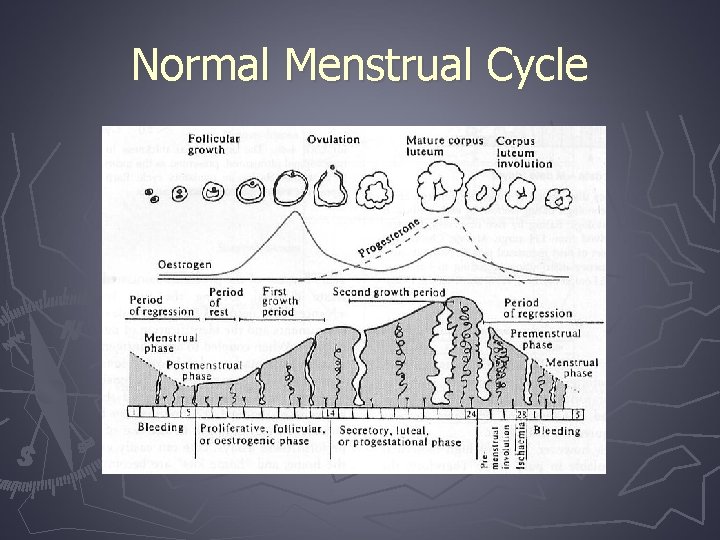
Historical Perspective ► Evolution from an anatomical disorder to an complex endocrine disorder ► 1800 s § Original description – “cystic oophoritis” § Anatomical disorder – thickened ovarian capsule and thus not able to ovulate

Historical Perspective ► 1930 s § Stein and Leventhal reported associated physical findings of irregular menstruation and infertility ► 1945 § Stein added excessive male pattern hair growth and obesity to the list ► 1960 s § Ability to measure hormones levels ► Noted increased androgen production from ovaries ► Noted abnormal gonadotropin secretion (LH)

Historical Perspective ► 1980’s § Relationship between hyperinsulinemia and hyperandrogenism § HAIR-AN syndrome § Insulin resistance and impaired glucose tolerance linked to central obesity, DM, HTN ► Metabolic Syndrome

Historical Perspective ► 1990 NIH workshop – standardize criteria § Chronic anovulation § Clinical or biochemical signs of hyperandrogenism § Exclusion of other causes (adult onset CAH, thyroid disease, hyperprolactinemia) § * * polycystic ovaries on u/s wasn’t included ► 2000 § PCOS viewed as a complex metabolic disorder with an array of cardiac and metabolic risk factors

Historical Perspective ► 2003 § Rotterdam Criteria – add u/s findings ► 2008 § Androgen Excess and PCOS Society § Requirement of hyperandrogenism ► 2012 § Institutes of Health Evidence-based Methodology Workshop on PCOS ►Concluded 2003 Rotterdam criteria should be adopted because most inclusive


Diagnostic Schemes

Prevalence ► Prevalence depends on diagnostic criteria used to define the disorder § Global population – 6. 5 to 8% (NIH 1990) § BJOG 2006 report - 827 women oligo-ovulation § 456 (55%) had PCOS by NIH 1990 criteria § 754 (91%) had PCOS by Rotterdam 2003 criteria

Prevalence ► Difficult to estimate secondary to variable expression patterns – § Hyperandrogenism (hirsutism, acne, alopecia) § Menstrual or ovulatory dysfunction § Infertility § Insulin resistance § Polycystic ovaries on ultrasound § Obesity

Prevalence ► 40% with PCOS do not express the classic signs and symptoms frustration ► Hyperandrogenism § Hirsutism – 75% have PCOS (NIH 1990) § Acne – 19 to 37% have PCOS (criteria not well defined) § Hirsutism, acne, or alopecia – 72% have PCOS (Rotterdam 2003)

Prevalence of Polycystic Appearing Ovaries on Ultrasound ► 92% with idiopathic hirsutism ► 87% with oligomenorrhea ► 82% with DM 2 ► 82% with adult-onset congenital adrenal hyperplasia ► 67% with androgenic alopecia ► 40% with history of GDM ► 26% with amenorrhea ► 20% of women WITHOUT androgen excess

Epidemiology ► High Risk Groups § Oligo-ovulatory Infertility § Obesity § Insulin Resistance § Type 1, Type 2, or Gestational DM § History of premature pubarche § First Degree relative with PCOS

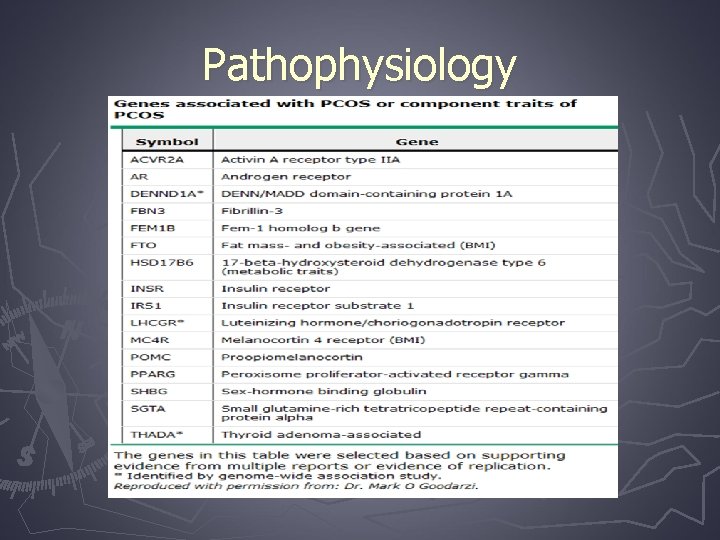
Pathophysiology ► Principal molecular defect that causes PCOS is unknown ► Interaction of multiple genetic variants and environmental factors (diet, obesity)

Pathophysiology
Pathophysiology

Pathophysiology ►Principal genetic targets § Gonadotropin secretion § Insulin secretion § Androgen biosynthesis § Weight and energy regulation
Normal Menstrual Cycle

Anovulation
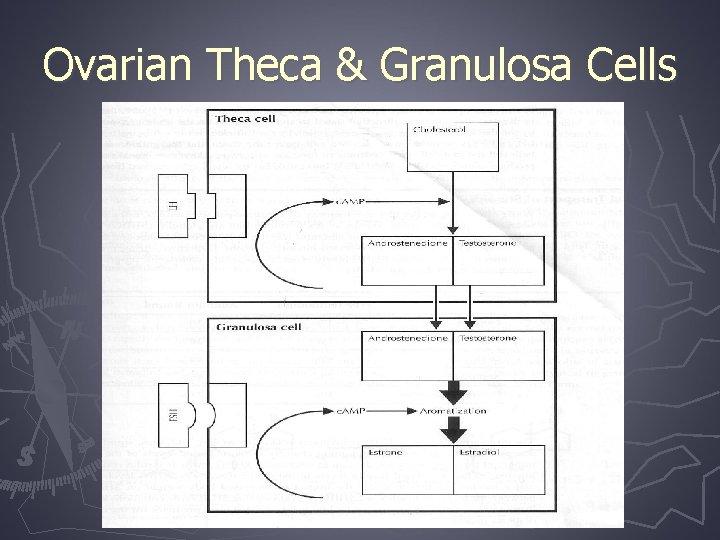
Ovarian Theca & Granulosa Cells

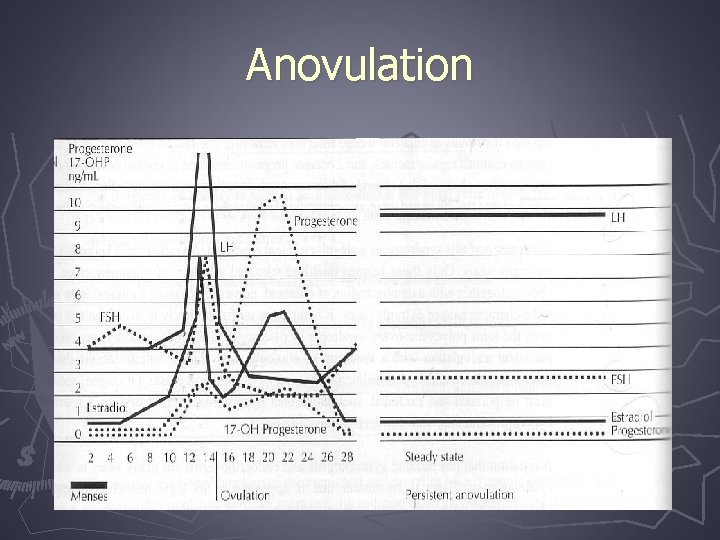
Pathophysiology Gonadotropin secretion ► Higher LH concentration compared to age matched controls ► FSH normal or low ► Elevated ► LH LH/FSH level not required for increased ovarian androgen secretion or polycystic ovarian morphology

Pathophysiology Gonadotropin secretion ► LH action enhanced at ovarian level ► LH receptor is overexpressed in theca cells ► LH increased relative to FSH levels ► Follicular arrest and increased androgen production in the ovarian theca cells ► The most likely cause of anovulation is an FSH level too low to fully mature the follicles ► FSH levels may be suppressed by negative feedback inhibition from mid-follicular estradiol level ► Anovulation
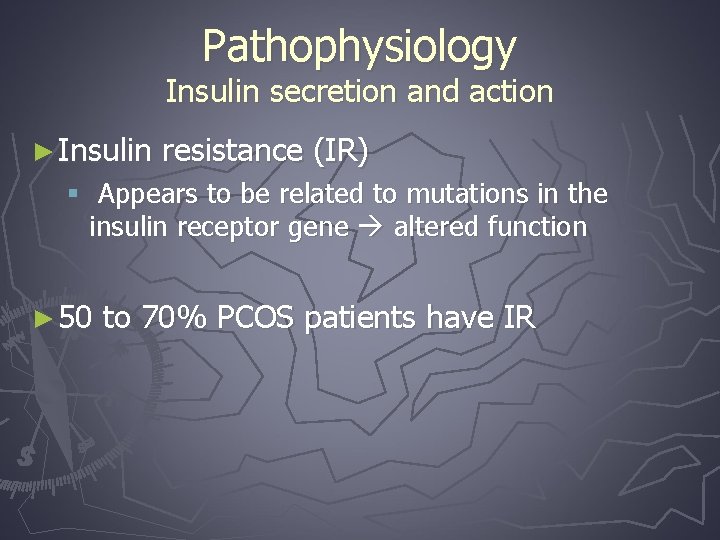
Pathophysiology Insulin secretion and action ► Insulin resistance (IR) § Appears to be related to mutations in the insulin receptor gene altered function ► 50 to 70% PCOS patients have IR

Pathophysiology Insulin secretion and action ► IR leads to hyperandrogenism § Hyperinsulinemia and LH synergistically stimulates theca cell secretion of androgens § Hyperinsulinemia inhibits hepatic sex-hormone binding globulin (SHBG) production ►Resulting in an increase in free androgens

Pathophysiology Insulin secretion and action ► Insulin resistance – no recommended screening test § Insulin level assessment has not been shown to identify women who will respond to therapy ► Instead of IR testing, there is increasing recognition of metabolic syndrome § 33% of PCOS patients

Metabolic Syndrome

Pathophysiology Androgen biosynthesis and action ► Produced by ovaries and adrenal glands ► Testosterone ► 70% bound to SHBG ► 20 -30% bound to albumin ► 1% free - - biologically active

Sources of Serum Androgens


Serum Androgens ► Testosterone (T) § Majority made in ovary § Most potent circulating androgen § Biological activity determined by the amount of binding to sex hormone binding globulin § Free testosterone is active

Serum Androgens ► Androstenedione (A) § Immediate precursor to testosterone § Ovary and adrenal production ► Dehyrdoepiandrosterone sulfate (DHEA-S) § Majority derived from adrenal glands § Small percentage from ovary

Serum Androgens ► Dihydrotestosterone (DHT) § Peripheral conversion in androgen responsive tissues § Intracellular 5 -alpha reductase converts T to DHT § DHT binds to androgen receptor with affinity 10 x greater than T

Serum Androgens ► Dihydrotestosterone (DHT) § Women with PCOS have increased 5 -alpha reductase activity (converts T to DHT) ►Resulting in increased activation of the pilosebaceous unit (hair growth, sebum production) with only modest increases in bioavailable testosterone
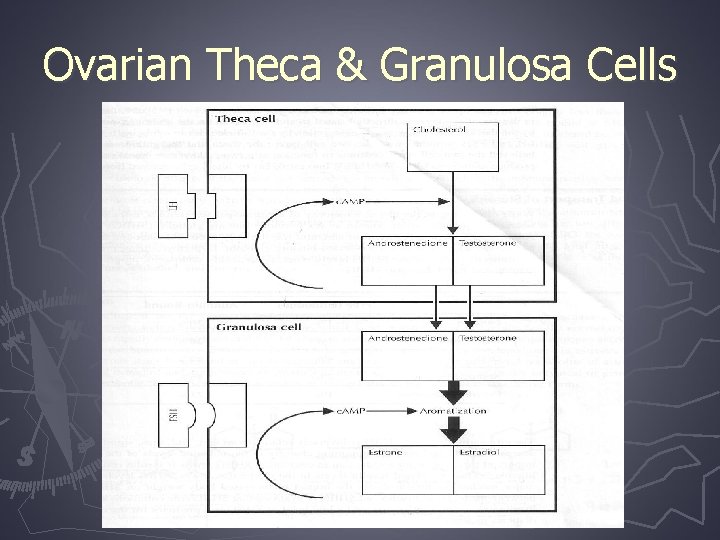
Ovarian Theca & Granulosa Cells

Ovarian Androgen Secretion ► Androgens produced in theca cells which respond to LH ► Role of insulin § Synergistic effect of LH and insulin to increase androgen secretion ► Theca cells synthesize mostly androstenedione and some testosterone ► They diffuse across the basement membrane to the granulosa cells

Ovarian Androgen Secretion (continued) ► The granulosa cells, in response to stimulation by FSH, produce aromatase which converts androgen precursors to estrone and estradiol (negative feedback to FSH) ► Impeded normal follicular growth, resulting in follicular arrest at the 4 -8 mm diameter size ► A dominant follicle (18 -25 mm) does not develop therefore ovulation does not occur

Ovarian Androgen Secretion (continued) ► Women with PCOS have increase androgen production because of one or more of the following: ►Increased volume of theca cells ►Increased LH stimulation of theca cells ►Increased theca cell sensitivity to LH ►Potentiation of the action of LH by hyperinsulinemia ►Polymorphism of the gene for the beta subunit of LH and subsequent different biological activity (even in the presence of normal LH level) ►Increased expression of the LH receptor

Adrenal Androgen Secretion ► Adrenal androgen secretion § Under control of ACTH § Over 50% of women with PCOS have evidence of increased adrenal androgen secretion

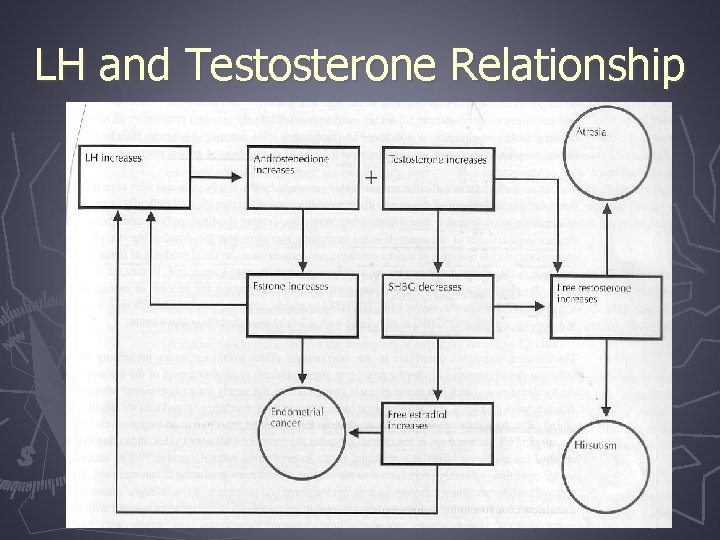
LH and Testosterone Relationship

Pathophysiology Weight and energy regulation ► Obesity § Truncal obesity ► 40 -80% of PCOS are overweight or obese § Presence of obesity results in: ►Insulin resistance hyperinsulinemia § Hyperinsulinemia synergy with LH and drop in SHBG ► Hyperandrogenism hirsuitism/acne ► Arrest of follicular development chronic anovulation § PCOS patients ► 31 -35% have IGT (1. 6% in non-PCOS) ► 7. 5 -10% have DM (2. 2% in non-PCOS)

Clinical Features Adolescence ► No formal diagnostic criteria § Obesity § Irregular cycles ► 50% of cycles are anovulatory in first 2 years after menarche ► More Concerning… § Hyperandrogenism § Peripubertal girls with pubarche before age of 8

Clinical Features Reproductive age ► Anovulatory Symptoms ► Hirsutism ► Acne ► Male pattern hair loss § 2/3 patients § Erratic cycles with breakthrough bleeding § Primary amenorrhea § Oligomenorrhea <9 per year § Intervals < 22 days ► Polycystic Ovary § 70% of women with hirsutism and oligomenorrhea § Multiple small preantral and antral follicles § Periperhal location § Increased volume of stroma § Thickened and sclerotic cortex

Clinical Features Reproductive age ► Obesity ► Insulin Resistance ► § 40 -80% have BMI > 30 § majority of PCOS patients regardless of obesity § (30% lean and 70% obese) Diabetes (7. 5 to 10% of PCOS patients) § 31 -35 % of PCOS patients have glucose intolerance § 2 -5 fold increase in developing DM ► Infertility ► Pregnancy § 75% of infertility causes § Poor FSH stimulation and elevated LH levels impair follicle maturation and ovulation § Spontaneous abortion rate 20 -40% higher § Pregnancy complication rate (GDM 3. 4 x, GHTN 3. 4 x, Pre. E 2. 2 x, PTB 1. 9 x)

Clinical Features Reproductive age ► Endometrial Hyperplasia/Cancer § Chronic exposure to unopposed estrogens ► Dyslipidemia – low HDL, high triglycerides ► Metabolic syndrome – 30 to 40% ► Nonalcoholic fatty liver disease – 30% in PCOS compared to 2% all women and 5% women with DM 2 ► Coronary heart disease ► Sleep apnea ► Depression/anxiety ► Eating disorders (binge eating)

Differential Diagnosis


History ► Anovulatory Bleeding ► * No moliminal symptoms * ► Oligomenorrhea § > 35 day cycle, <9 per year ► Hair growth or loss ► Acne ► Weight changes ► FH – PCOS, DM, CV disease ► Age (less common diagnosis >30 yrs)

History ► Exclusion of other etiologies § Rapid onset – think androgen secreting tumor § Onset at puberty – think CAH § Glactorrhea with oligomenorrhea – think hyperprolactinemia § Hypothyroid findings

Physical Exam ► BP, BMI, waist circumference ► Classical findings § Hirsutism § Central obesity ► Seborrhea, acne, and alopecia ► Acanthosis nigricans

Physical Exam ► Palpable ovarian mass § Androgen-secreting tumor? ► Virilization (rare) § Clitoromegaly § Deepening of voice § Temporal balding § Androgen-secreting tumor?

Physical Exam ► Galactorrhea § Hyperprolactinemia ► Central obesity, moon face, hirsutism, purple striae, plethora § Cushings

Physical Exam ► Hirsutism § Excessive sexual hair in male pattern distribution ► Ferriman-Gallwey score § Helpful in determining response to treatment § 5 grades based on densities and areas involved for each of nine hormonal sites ► <8 is normal ► 8 -15 mild hirsutism ► >15 moderate to severe hirsutism

Ferriman-Gallwey Score

Labs ► Goal is to exclude other ► Androgens § Total Testosterone etiologies ► widely available ► mildly elevated in PCOS ► If >150 ng/d. L (normal<70) § Free Testosterone ► more sensitive test ► Less sensitive for women’s § DHEA-S consider androgen secreting tumor levels ► marker for adrenal hyperandrogenemia ► If >800 mcg/d. L (normal <270) consider tumor § SHBG ? ? androgen secreting
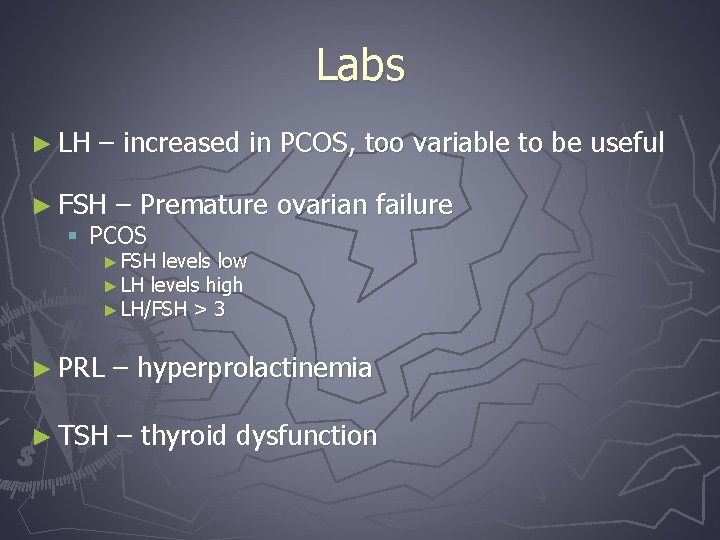
Labs ► LH – increased in PCOS, too variable to be useful ► FSH – Premature § PCOS ovarian failure ► FSH levels low ► LH levels high ► LH/FSH > 3 ► PRL – hyperprolactinemia ► TSH – thyroid dysfunction

Labs ► Fasting glucose § Fasting plasma glucose ►<100 mg/d. L normal ► 100 -125 mg/d. L impaired fasting glucose/prediabetes ►>126 mg/d. L DM ► 2 § § § hour glucose level after 75 gm oral glucose load 140 -199 mg/d. L indicates impaired glucose tolerance Above 199 mg/d. L is diagnostic for type diabetes Recheck every 2 years if IGT

Labs ► Fasting insulin – simple and inexpensive ►>25 -30 μU/m. L is abnormal ► 40% PCOS patients who have IGT may have normal fasting insulin level… § Some suggest >20 μU/m. L or by averaging 2 or 3 readings to account for day-to-day variability ► Insulin level assessment has not been shown to identify women who will respond to therapy ► Lipid panel

Who Should be Screened for Nonclassical Congenital Adrenal Hyperplasia ► Present in adult women with anovulation and hirsutism § Due to genetic defects in the steroidogenic enzyme, 21 hydroxylase § Highest prevalence among Ashkenazi Jews, followed by Hispanics, Yugoslavs, Native American Inuits in Alaska, and Italians § Women in groups at higher risk for nonclassical congenital adrenal hyperplasia and a suspected diagnosis of PCOS should be screened with a 17 hydroxyprogesterone value
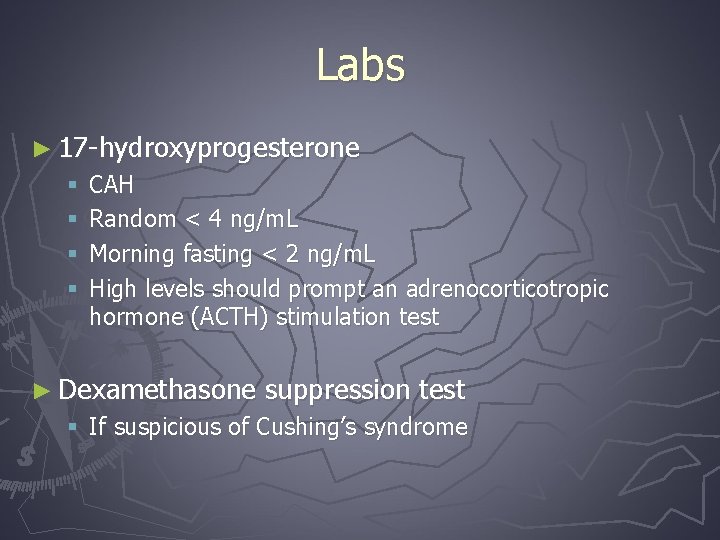
Labs ► 17 -hydroxyprogesterone § § CAH Random < 4 ng/m. L Morning fasting < 2 ng/m. L High levels should prompt an adrenocorticotropic hormone (ACTH) stimulation test ► Dexamethasone suppression test § If suspicious of Cushing’s syndrome

Additional Testing ► Non-alcoholic fatty liver – no screening tests ► Depression/anxiety – validated screening tools (PHQ-9 for depression and GAD-7 for anxiety) ► Sleep – inquire for apnea and daytime somnolence § sleep study PRN

Testing on the horizon… ► Anti-Müllerian hormone (AMH) § Exclusively of ovarian origin – marker of ovarian reserve § It reduces preantral and antral follicle responsiveness to FSH § Levels decrease with age § Levels elevated in PCOS likely secondary to aberrant activity of the granulosa cells § Treatment with insulin sensitizers are associated with a reduction in both serum AMH levels and antral follicles – assess treatment efficacy
Imaging ► Assessment of endometrial abnormalities ► Pelvic U/S to rule out ovarian mass § Follow up MRI or CT if needed ► PCOS ovaries are enlarged (>5 cm) § > 12 subcapsular follicles (2 -9 mm) in one or both ovaries § Ovarian volume >10 m. L § Dense hyperechoic stroma
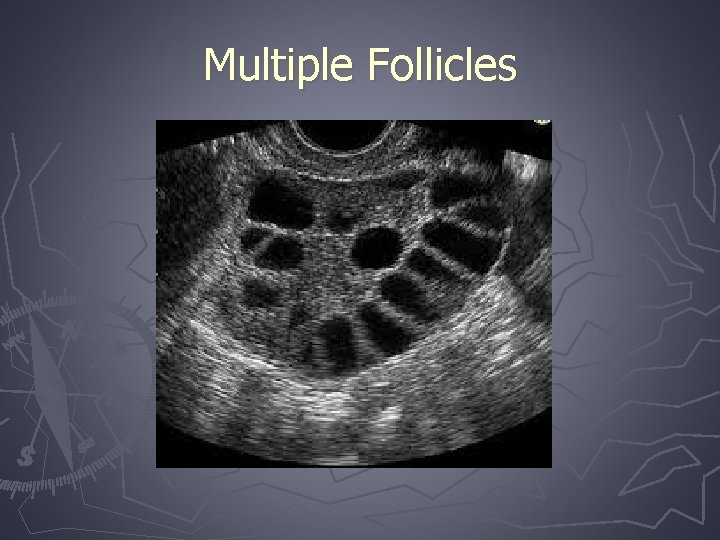
Multiple Follicles

Goals of Treatment ► Lifestyle ► Lower ► Avoid changes risk for DM and CV disease effects of hyperinsulinemia ► Reduce production and circulating levels of androgens

Goals of Treatment ► Protect the endometrium against effects of unopposed estrogen ► Induce ovulation to achieve pregnancy ► Contraception treatment – return of ovulation with

Treatment for those NOT pursuing pregnancy ► Menstrual protection dysfunction and endometrial § OCPs – first line ►Cycle regulation – predictable/regular withdrawal bleed ►Contraception ►Progestin antagonizes the proliferative effect of estrogen and prevents endometrial hyperplasia

Treatment for those NOT pursuing pregnancy ► Menstrual dysfunction and endometrial protection (continued) § Progestin only ►Cyclical or continuous oral dosing ►Progestin IUD ►Progestin rod implant § Metformin – second line ►Restoration of ovulatory cycles in 50% of women

Treatment for those NOT pursuing pregnancy ► Androgen excess § OCPs – first line ►Decreases LH secretion decrease ovarian androgen production ►Increases hepatic production of SHBG decrease in bioavailable testosterone ►Decrease in adrenal androgen secretion

Treatment for those NOT pursuing pregnancy ► Androgen excess (continued) § OCPs – first line ►Chose OC with desogestrel or drosperinone (both with higher VTE risk) ►Also consider norethindrone (not as low andorgenic acitivity but less VTE risk) ►May need 30 -35 mcg ethinyl estradiol to suppress androgens


Treatment for those NOT pursuing pregnancy ► Anti-androgen – added if suboptimal effects after 6 months * * MUST use contraception § Spironolactone – 50 -100 mg BID ► Aldosterone antagonist diuretic ► Competitive androgen receptor antagonist § Finasteride, flutamide, Gn. RH agonist ► Eflornithin HCl (Vaniqa) 13. 9% cream BID ► Concomitant therapy (OC and anti-androgen) § Cosmetic – mechanical (shaving, waxing, depilatories, electrolysis, laser

Treatment for those NOT pursuing pregnancy ► Metabolic abnormalities § Obesity – weight loss 5 -10% to restart ovulatory patterns (diet/exercise, pharmacotherapy, bariatric surgery) ►Caloric restriction is main factor ►No data supporting one diet over the other § Low glycemic makes sense!

Treatment for those NOT pursuing pregnancy ► Metabolic abnormalities (continued) § IR/risk of DM 2 – metformin (first line) ►thiazolidinediones (wt gain, less studied in PCOS) § Metabolic effects of OCs in PCOS – no increased risk of VTE § Dyslipidemia – exercise/weight loss, pharmacotherapy if needed § OSA – CPAP

Metformin ► Major effect is to decrease hepatic glucose production thus less need for insulin secretion ► Target dose 1500 -2000 mg/day (can use short acting or extended dosing)

Metformin ► Side effects – diarrhea, nausea/vomiting, flatulence, indigestion, abdominal discomfort ► Avoid if risk for lactic acidosis (renal insufficiency) ► “Off label” use – oligomenorrhea, hirsuitism, obesity, prevention of DM 2

Metformin Use In… ► Oligomenorrhea § OCPs (first line) or progestin only option § Metformin (second line) – 50% will restore ovulation – may take 6 months or longer ► Metabolic effects § Improvement in plasma insulin and insulin sensitivity (glucose clamp studies), reduction in serum free testosterone, and increase in mean HDL concentration

Metformin Use In… ► Endometrial effects § 50% become ovulatory, however unless REGULAR and ovulatory, cannot assume global sloughing of endometrium ►Addition of OCPs or progestin only option ► Hirsutism § OCPs – first line § Anti-androgen – second line (after 6 months) § Metformin – although may reduce serum androgens, limited benefit in treating hirsutism

Metformin Use In… ► Anovulatory infertility § Lifestyle – caloric restriction and exercise for BMI > 27 § Metformin effective for restoring ovulation, it appears to be less effective for fertility (live birth rates) compared to clomiphene ►Experts recommending reserving use of Metformin in those with glucose intolerance ► Obesity – metformin plus calorie restriction

Metformin Use In… ► Diabetes prevention § Delay conversion from IGT to DM ► Pregnancy § SAB rate in PCOS 20 -40% - no studies addressing with metformin § GDM – 7 x higher in women with PCOS, inconsistent prevention results § Preeclampsia, PTB – no significant differences § Neonatal outcomes – no malformations § Breast feeding – compatible

Metformin and OCPs ► Metformin + OCPs § Inadequate evidence to recommend routine addition of metformin as unclear whether this combination has important cosmetic or metabolic advantages over OCP monotherapy ► Metformin vs OCPs § OCPs first line for oligomenorrhea and hyperandrogenism. OCPs less beneficial for insulin sensitivity while metformin better at reducing fasting insulin

Metformin and Patient Goals ► The use of metformin for the management of women with PCOS depends on the individual patient’s goals: § Oligomenorrhea who need endometrial protection – recommend OCP first line, progestin only option second line. Metformin has not be proven to be endometrial protective § Hyperandrogenic symptoms alone – OCP first line, anti-androgen second line

Metformin and Patient Goals ► The use of metformin for the management of women with PCOS depends on the individual patient’s goals: § Desire pregnancy – weight loss is first line, ovulation induction with clomiphene, add metformin? § Obesity – caloric restriction/exercise, metformin as an adjunct § DM 2/IGT – management same as non-PCOS § Suggest against use of metformin to prevent pregnancy loss in PCOS patients

Treatment for those pursuing pregancy ► Weight loss – 5 -10% loss yields resumption of ovulatory cycles ► Ovulation induction – be sure to do a semen analysis and HSG to complete infertility § Clomiphene § Letrozole § Metformin § Gonadotropins

Treatment for those pursuing pregancy ► Ovulation induction agents § Clomiphene – first line ►Selective estrogen receptor modulator (SERM) – competitive inhibitor of estrogen binding to receptors in hypothalamus (blocks the negative feedback loop of estrogen) and results in increase in Gn. RH, FSH, and LH and influence follicular development. It is an estrogen agonist enhancing FSH stimulation of LH receptors in the granulosa cells ► 80% will ovulate and 50% will conceive

Treatment for those pursuing pregancy ► Ovulation induction agents § Letrozole – aromatase inhibitor (off label use) ►Aromatase catalyzes the rate limiting step in production of estrogen thus suppresses ovarian estradiol secretion and rise in FSH and follicle production ►Also used as adjuvant endocrine therapy in postmenopausal breast cancer § Metformin – with or without clomiphene § Gn. RH – higher risk for ovarian hyperstimulation syndrome

Treatment for those pursuing pregancy ► Laparoscopic surgery § Wedge resection – abandoned secondary to adhesion formation, better results with clomiphene § Ovarian drilling/diathermy ► In vitro fertilization (IVF) ► Intracytoplasmic sperm injection (ICSI)

Laparoscopic Surgery for Ovulation Induction in PCOS ► Majority with anovulatory infertility will ovulate in response to clomiphene, however up to 30% remain anovulatory § Of 70% who do ovulate, only 50% will conceive § Addition of metformin can help ovulation % § Those that are still unresponsive/resistant move to gonadotropin therapy ►Issues: difficult to titrate the dose to achieve monofollicular ovulation, 30% risk of multiples, risk of ovarain hyperstimulation syndrome, cost, SAB risk is higher

Laparoscopic Surgery for Ovulation Induction in PCOS ► Dates to 1930’s – bilateral ovarian wedge resection resulted in restoration of regular menses and pregnancy § fell out of favor secondary to post-op adhesion formation and the introduction of clomiphene ► Ovarian drilling/electrocautery – less adhesions, similar pregnancy rates to gonadotropin with less multiple risk
Laparoscopic Surgery for Ovulation Induction in PCOS ► Outcomes § Drop in LH, testosterone, inhibin § Uncertain mechanism promoting ovulation but suspect that involves a sudden drop in intraovarian androgens that results in increased FSH secretioin and intrafollicular environment more conducive to normal follicular maturation and ovulation

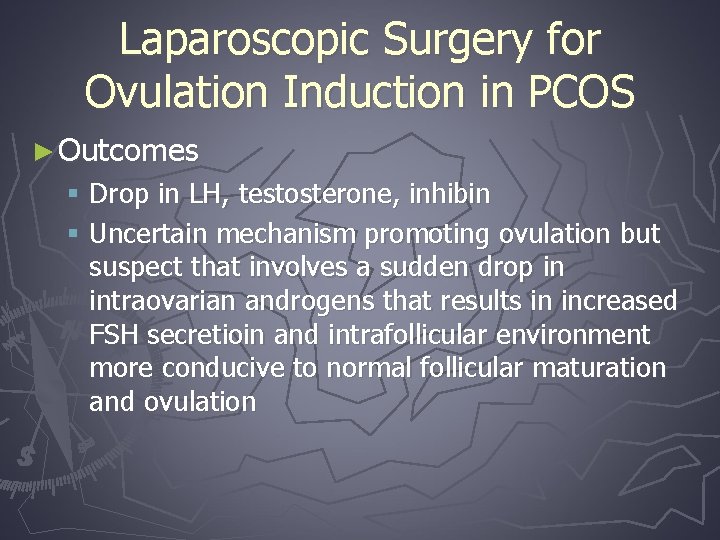
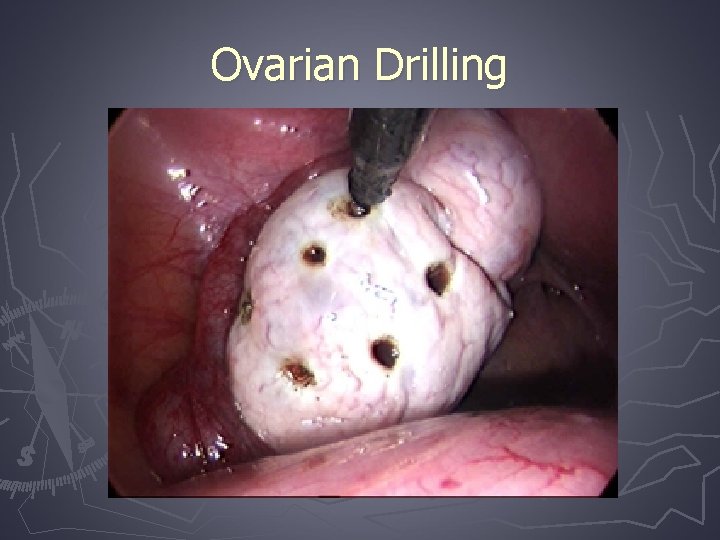
Ovarian Drilling ► Create focal areas of damage to the ovarian cortex and stroma ► Unipolar needle electrode insulated down to 2 cm of exposed probe. 4 -6 punctures of each ovary
Ovarian Drilling

Ovarian Drilling ► Efficacy – similar conception rates to gonadotropin therapy § Advantages – no cyclical monitoring, more cost effective, no increase risk of multiple gestations or OHSS § Disadvantages – anesthesia, surgical risk (bleeding, infection, damage to surrounding tissues, adhesive disease)

Ovarian Drilling ► Long term effects of drilling are not-known thus limit to those trying to achieve pregnancy ► Other considerations – often unsuccessful in obese women, patients should have no other infertility factors (tubal, endometriosis, male factor), IVF success


Treatment for those pursuing pregancy ► Anovulatory PCOS patients – weight loss (if overweight/obese), ovulation induction (clomiphene and metformin) § If still anovulatory, then exogenous gonadotropins or ovarian drilling (same success rate, less chance of multiples) ► Gonadotropin concerns § Higher cost, frequent monitoring § L/S anesthesia risk, surgical risk, adhesion formation, theoretical risk of premature ovarian

Treatment for those pursuing pregancy ► Laparoscopic candidates – PCOS patients who have failed clomiphene and metformin, non-obese BMI <30, and no other fertility factors ► Balance of risks – most will suggest gonadotropins for first given the invasiveness, however some women prefer less monitoring and want the decreased risk of multiple gestation

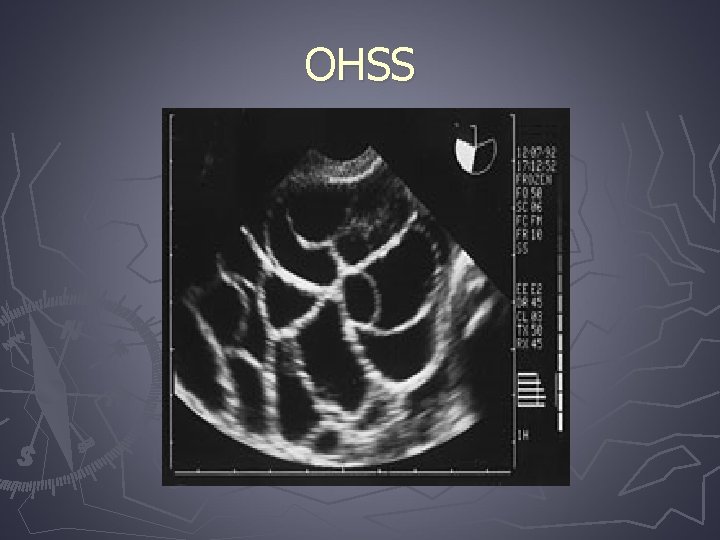
Ovarian hyperstimulation syndrome (OHSS) ► Most serious complication of controlled ovarian hyperstimulation for assisted reproduction technologies ► It is a broad spectrum of signs and symptoms that include abdominal distention and discomfort, enlarged ovaries, ascites, and other complications of enhanced vascular permeability

Ovarian hyperstimulation syndrome (OHSS) ► The pathophysiology of OHSS is not fully understood, but increased capillary permeability with the resulting loss of fluid into the third space is its main feature ► In the susceptible patient, h. CG administration for final follicular maturation and triggering of ovulation is the pivotal stimulus for OHSS

Ovarian hyperstimulation syndrome (OHSS) ► After the HCG administration, this leading to overexpression of vascular endothelial growth factor (VEGF) in the ovary, release of vasoactive-angiogenic substances, increased vascular permeability, loss of fluid to the third space, and full-blown OHSS ► OHSS is a life-threatening condition because it can cause venous or arterial thromboembolic events, including stroke and loss of perfusion of an extremity
OHSS
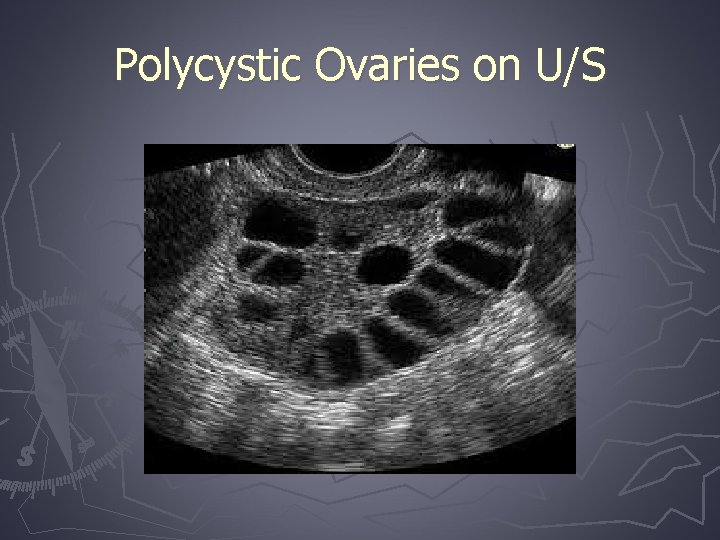
Polycystic Ovaries on U/S
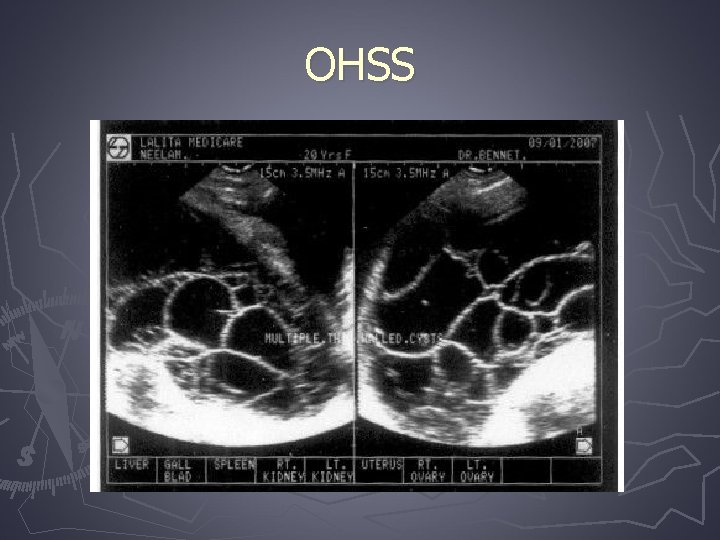
OHSS
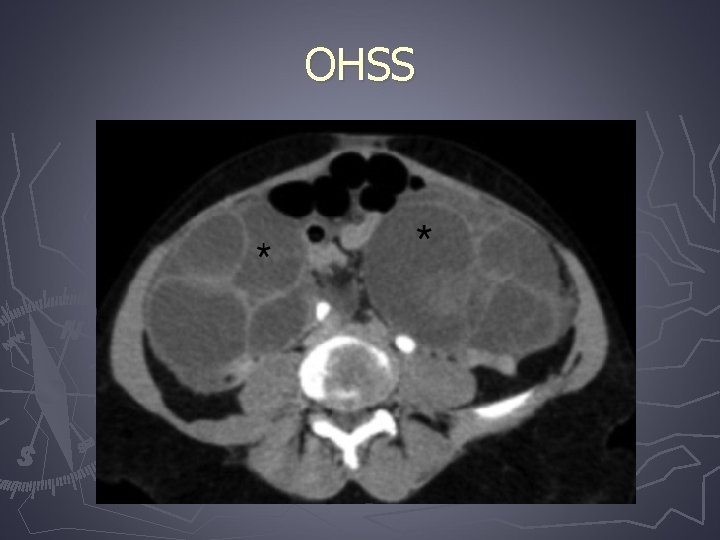
OHSS
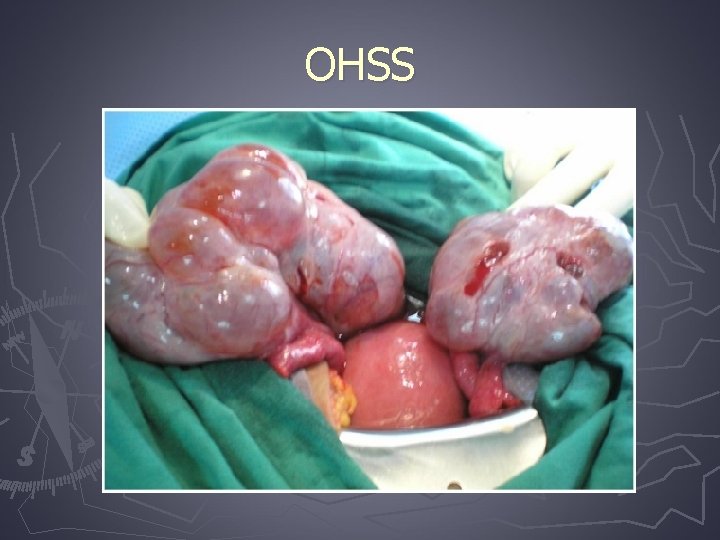
OHSS

Summary ► PCOS is an heterogenous disorder with the phenotype of oligoovulation and hyperandrogenism § Caused by combination of… ► Elevated LH ► Hyperinsulinemia ► Ovarian androgen overproduction ► Disruption of follicle growth § Manifests in reproductive and metabolic abnormalities ► Future molecular studies will help delineate the pathophysiology and help direct new treatment strategies

Summary ► Manifests in reproductive and metabolic abnormalities ► Future molecular studies will help delineate the pathophysiology and help direct new treatment strategies

The End

Bibliography ► ► ► ► Clinical Gynecologic Endocrinology and Infertility. 7 th Edition. Leon Speroff and Marc Fritz. Comprehensive Gynecology. 4 th Edition. Stenchever, Droegemueller, Herbst, Mishell. Clinical Gynecology. 1 st Edition. Bieber, Sanfilippo, Horowitz. Polycystic Ovary Syndrome. ACOG Practice Bulletin. Number 108. October 2009. Reaffirmed 2013 A practical approach to the diagnosis of polycystic ovary syndrome. American Journal of Obstetrics and Gynecology. Volume 191, Issue 3, Pages 713 -717 (September 2004). Polycystic Ovarian Syndrome: 3 Key Challenges. Dale W. Stovall, MD OBG Management · June 2003 · Vol. 15, No. 6 Polycystic ovary syndrome: How are obesity and insulin resistance involved? . OBG Management. October 2012 · Vol. 24, No. 10

Bibliography ► ► ► Polycystic ovary syndrome: Where we stand with diagnosis and treatment and where we’re going. OBG Management. September 2012 · Vol. 24, No. 9 Polycystic ovary syndrome: The long-term metabolic risks. OBG Management. November 2012 · Vol. 24, No. 11 Polycystic ovary syndrome: Cosmetic and dietary approaches. OBG Management. December 2012 · Vol. 24, No. 12 Epidemiology and pathogenesis of the polycystic ovary syndrome in adults. Uptodate. com. Literature review current through: Jul 2015. | This topic last updated: May 01, 2013. Clinical manifestations of polycystic ovary syndrome in adults. Uptodate. com. Literature review current through: Jul 2015. | This topic last updated: Mar 24, 2015. Diagnosis of polycystic ovary syndrome in adults. Uptodate. com. Literature review current through: Jul 2015. | This topic last updated: Mar 24, 2015.

Bibliography ► ► ► Diagnosis of polycystic ovary syndrome in adults. Uptodate. com. Literature review current through: Jul 2015. | This topic last updated: Mar 24, 2015. Treatment of polycystic ovary syndrome in adults. Uptodate. com. Literature review current through: Jul 2015. | This topic last updated: Sep 22, 2014. Steroid hormone metabolism in polycystic ovary syndrome. Uptodate. com. Literature review current through: Jul 2015. | This topic last updated: May 18, 2015. Metformin for treatment of polycystic ovary syndrome. Uptodate. com. Literature review current through: Jul 2015. | This topic last updated: Sep 17, 2014. Laparoscopic surgery for ovulation induction in polycystic ovary syndrome. Uptodate. com. Literature review current through: Jul 2015. | This topic last updated: Nov 08, 2013.

Summary of Recommendations Level A ► The following recommendations and conclusions are based on good and consistent scientific evidence (Level A):

Summary of Recommendations Level A ► An increase in exercise combined with dietary change has consistently been shown to reduce diabetes risk comparable to or better than medication. ► Improving insulin sensitivity with insulinsensitizing agents is associated with a decrease in circulating androgen levels, improved ovulation rate, and improved glucose tolerance.

Summary of Recommendations Level A ► The recommended first-line treatment for ovulation induction remains the antiestrogen clomiphene citrate. ► The addition of eflornithine to laser treatment is superior in the treatment of hirsutism than laser alone.

Summary of Recommendations Level B ► The following recommendations and conclusions are based on limited and inconsistent scientific evidence (Level B):

Summary of Recommendations Level B ► Women with a diagnosis of PCOS should be screened for type 2 diabetes and impaired glucose tolerance with a fasting glucose level followed by a 2 -hour glucose level after a 75 -g glucose load. ► Women with PCOS should be screened for cardiovascular risk by determination of BMI, fasting lipid and lipoprotein levels, and metabolic syndrome risk factors.

Summary of Recommendations Level B ► Reduction in body weight has been associated with improved pregnancy rates and decreased hirsutism, as well as improvements in glucose tolerance and lipid levels. ► There may be an increase in pregnancy rates by adding clomiphene to metformin, particularly in obese women with PCOS.

Summary of Recommendations Level B ► If clomiphene citrate use fails to result in pregnancy, the recommended second-line intervention is either exogenous gonadotropins or laparoscopic ovarian surgery.

Summary of Recommendations Level C ► The following recommendations and conclusions are based primarily on consensus and expert opinion (Level C):

Summary of Recommendations Level C ► Combination low-dose hormonal contraceptives are most frequently used for long-term management and are recommended as the primary treatment of menstrual disorders.

Summary of Recommendations Level C ► Women in groups at higher risk for nonclassical congenital adrenal hyperplasia and a suspected diagnosis of PCOS should be screened to assess the 17 hydroxyprogesterone value. ► A low-dose regimen is recommended when using gonadotropins in women with PCOS. ► There is no clear primary treatment for hirsutism in PCOS.


- Slides: 128