Polyacrylamide Gel Electrophoresis PAGE of Proteins Ph D














- Slides: 27


Polyacrylamide Gel Electrophoresis (PAGE) of Proteins • Ph. D. Dhurata Feta • Ph. D. Artiona Laze ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Background To understand Gel electrophoresis, we should have knowledge's in Chromatography methods Chromatography is the collective term for a set of separation techniques that operate based on Chromatography the differential partitioning of mixture components between a mobile and a stationary phase. The mobile phase (a liquid or a gas) travels through the stationary phase (a liquid or a solid) in a defined direction. The distribution of components between the two phases depends on adsorption, ionic interactions, diffusion, solubility or, in the case of affinity chromatography, specific interactions. Chromatographic methods are important in the analytical and preparative separation of biological methods samples. Gel filtration chromatography (size exclusion chromatography) is often the method of choice to purify macromolecules, taking advantage of their different sizes and shapes. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

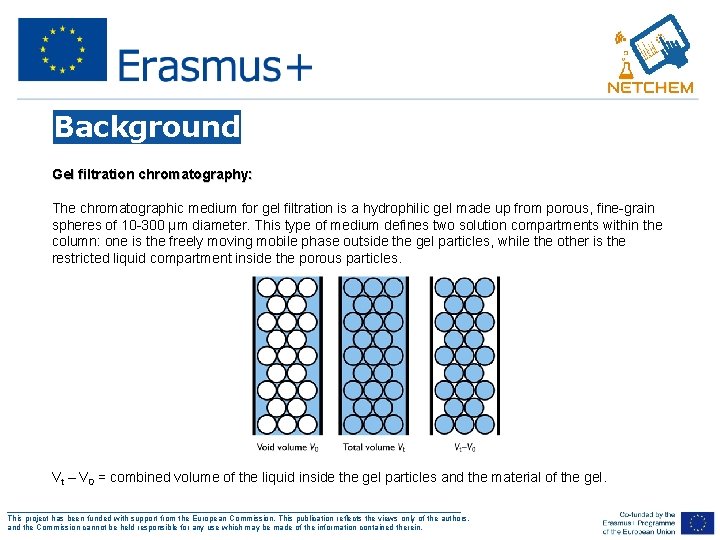
Background Gel filtration chromatography: The chromatographic medium for gel filtration is a hydrophilic gel made up from porous, fine-grain spheres of 10 -300 µm diameter. This type of medium defines two solution compartments within the column: one is the freely moving mobile phase outside the gel particles, while the other is the restricted liquid compartment inside the porous particles. Vt – V 0 = combined volume of the liquid inside the gel particles and the material of the gel. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

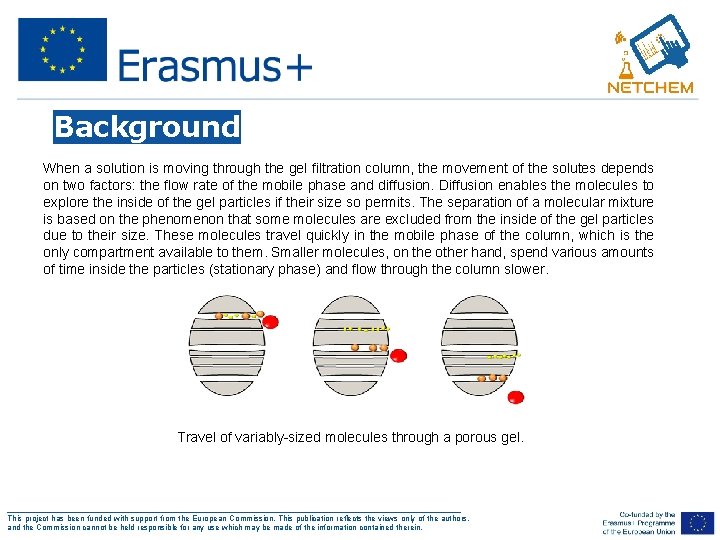
Background When a solution is moving through the gel filtration column, the movement of the solutes depends on two factors: the flow rate of the mobile phase and diffusion. Diffusion enables the molecules to explore the inside of the gel particles if their size so permits. The separation of a molecular mixture is based on the phenomenon that some molecules are excluded from the inside of the gel particles due to their size. These molecules travel quickly in the mobile phase of the column, which is the only compartment available to them. Smaller molecules, on the other hand, spend various amounts of time inside the particles (stationary phase) and flow through the column slower. Travel of variably-sized molecules through a porous gel. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Gel electrophoresis can provide information about the molecular weights and charges of proteins, the subunit structure of proteins and the purity of a particular protein preparation. It is relatively simple to use and it is highly reproducible. The most common use of gel electrophoresis is the qualitative analysis of complex mixtures of protein. Micro analytical methods and sensitive, linear image analysis system make gel electrophoresis popular for quantitative and preparative purposes as well. The technique provides the highest resolution of all methods available for separating proteins. Sodium dodecyl sulfate – polyacrylamide gel electrophoresis (SDS - PAGE) is the most commonly practiced gel electrophoresis technique used for proteins. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

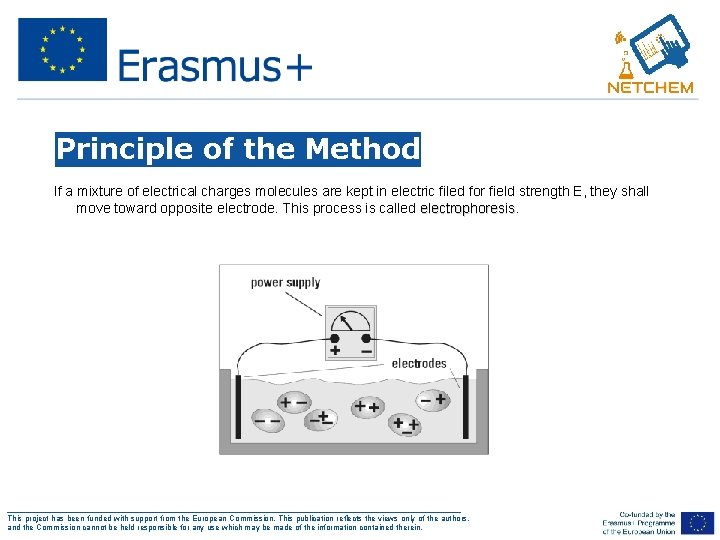
Principle of the Method If a mixture of electrical charges molecules are kept in electric filed for field strength E, they shall move toward opposite electrode. This process is called electrophoresis ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

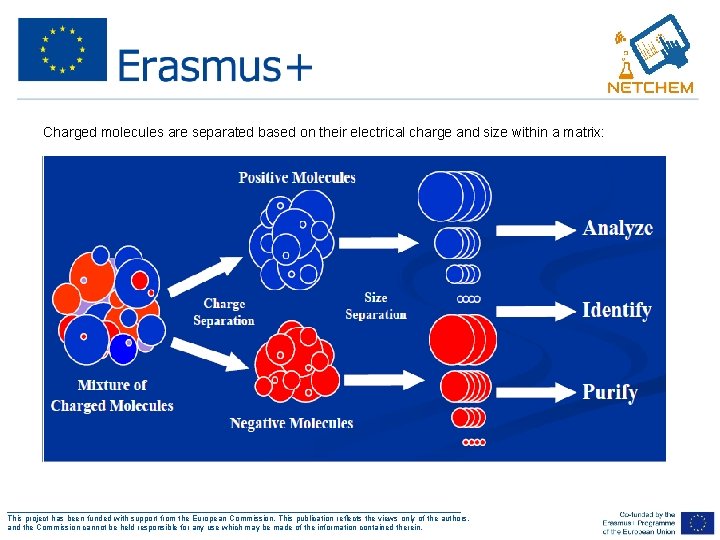
Charged molecules are separated based on their electrical charge and size within a matrix: ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

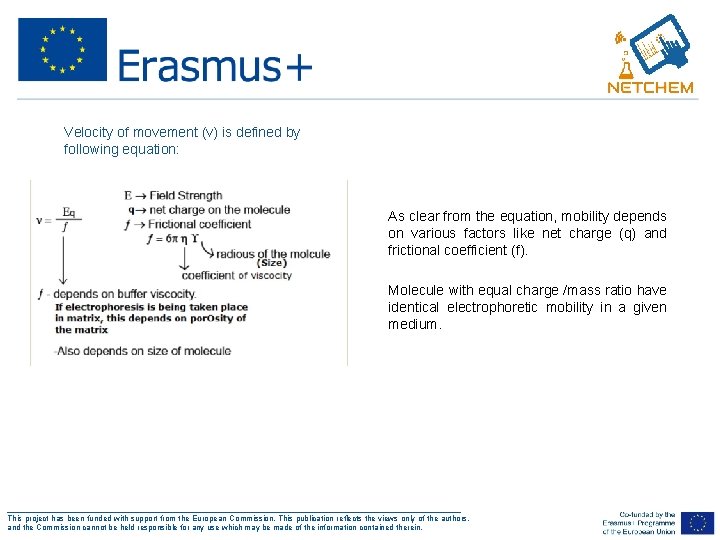
• Velocity of movement (v) is defined by following equation: • As clear from the equation, mobility depends on various factors like net charge (q) and frictional coefficient (f). • Molecule with equal charge /mass ratio have identical electrophoretic mobility in a given medium. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Now let us assume the electrophoresis is being taken place in liquid medium. What happens when voltage is removed? All molecules are diffused and mixed up again and electrophorectic mobility is not detectable soon after voltage was removed. If we want to see the position of molecule after electrophoresis, we need to minimize the diffusion. For this purpose, solid matrix is required in the process of electrophoresis and polyacrylamide is commonly used matrix in protein electrophoresis. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

I Electrophoresis at constant current or constant voltage? Ohm’s law relates V to current, I by electrical resistance, R as follow: We might think that increasing V would result in increase in current which in turn give faster electrophoretic mobility and doing experiment at fixed high voltage is better (? ? ). However, large current results in sufficient power generation (watts law). The power generated during electrophoresis is given by: Heat generation is not desirable in electrophoresis as this result in bad resolution of separation due to convection of buffer causing mixing of separated molecule. Moreover, temperature (heat) decreases viscosity of buffer. Decrease in viscosity results in easier movement of ions resulting in decrease in resistance. According to Ohm’s law decrease in resistance at fixed voltage result in further increase in current and head generation. Additionally, at high temperature thermolabile molecule like protein may breakdown during electrophoresis and may not be separated in native form. Thus, except specify applications, constant current may be a better option. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

SDS -PAGE - SDS-PAGE ( sodium dodecylsulphate-polyacrylamide gel electrophoresis) - The purpose of this method is to separate proteins according to their size, and no other physical feature - In order to understand how this works, we have to understand the two halves of the name: SDS and PAGE ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

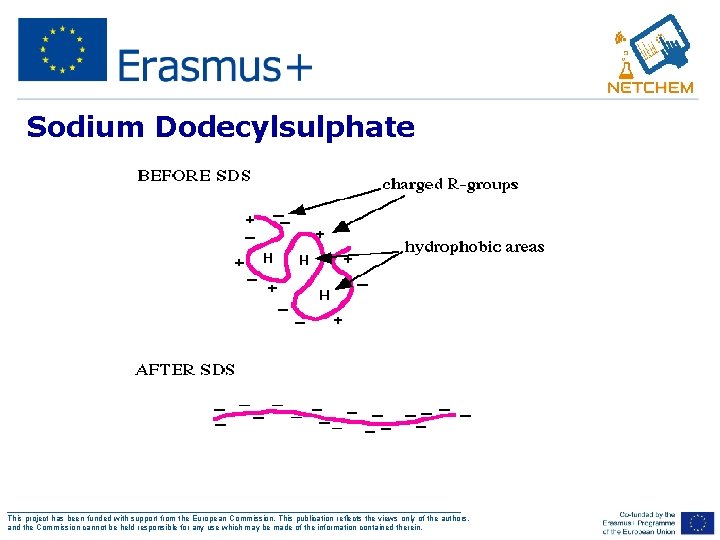
Sodium Dodecylsulphate Since we are trying to separate many different protein molecules of a variety of shapes and sizes, we first want to get them to be linear no longer have any secondary, tertiary or quaternary structure (i. e. we want them to have the same linear shape). Not only the mass but also the shape of an object will determine how well it can move through and environment. So we need a way to convert all proteins to the same shape - we use SDS ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Sodium Dodecylsulphate Ø SDS (sodium dodecyl sulfate) is a detergent that can dissolve hydrophobic molecules but also has a negative charge (sulfate) attached to it. Ø If SDS is added to proteins, they will be soluablized by the detergent, plus all the proteins will be covered with many negative charges. Ø A sample of protein, often freshly isolated and unpurified, is boiled in the detergent sodium dodecyl sulfate and beta-mercaptoethanol The mercaptoethanol reduces disulfide bonds The detergent disrupts secondary and tertiary structure Ø The end result has two important features: 1. all proteins contain only primary structure and 2. all proteins have a large negative charge which means they will all migrate towards the positive pole when placed in an electric field. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Sodium Dodecylsulphate ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

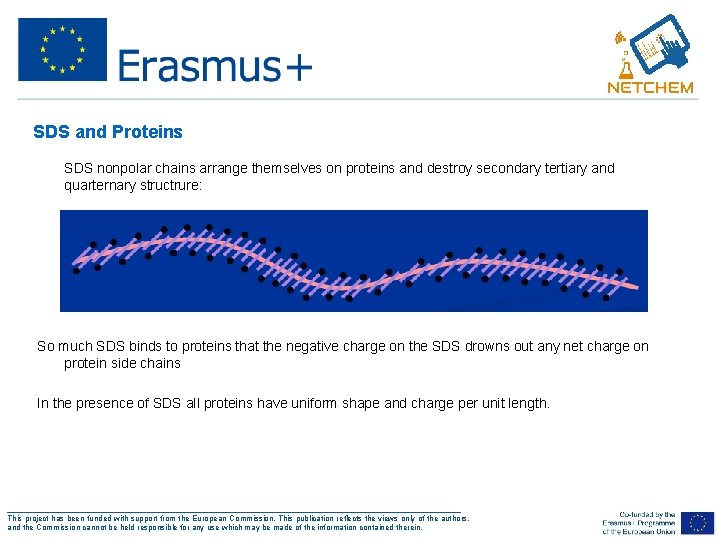
SDS and Proteins - SDS nonpolar chains arrange themselves on proteins and destroy secondary tertiary and quarternary structrure: So much SDS binds to proteins that the negative charge on the SDS drowns out any net charge on protein side chains In the presence of SDS all proteins have uniform shape and charge per unit length. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Polyacrylamide Gel Electrophoresis (PAGE) Ø PAGE is the preferred method for separation of proteins Ø Gel prepared immediately before use by polymerization of acrylamide and N, N'-methylene bis acrylamide. Ø Porosity controlled by proportions of the two components. Catalyst of polymerization ØPolymerization of acrylamide is initiated by the addition of ammonium persulphate and the base N, N, N’-tetrametyhlenediamine (TEMED). ØTEMED catalyses the decomposition of the persulphate ion to give a free radical. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

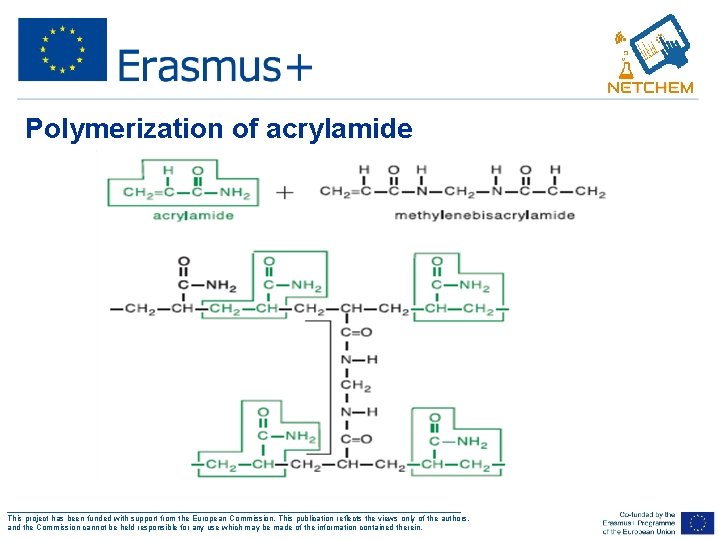
Polymerization of acrylamide ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

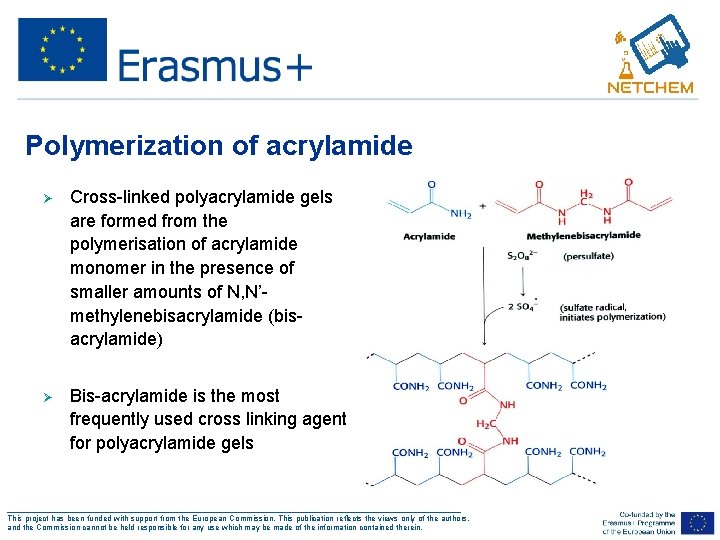
Polymerization of acrylamide Ø Cross-linked polyacrylamide gels are formed from the polymerisation of acrylamide monomer in the presence of smaller amounts of N, N’methylenebisacrylamide (bisacrylamide) Ø Bis-acrylamide is the most frequently used cross linking agent for polyacrylamide gels ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Polyacrylamide Gel • Bis-Acrylamide polymerizes along with acrylamide forming cross-links between acrylamide chains ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

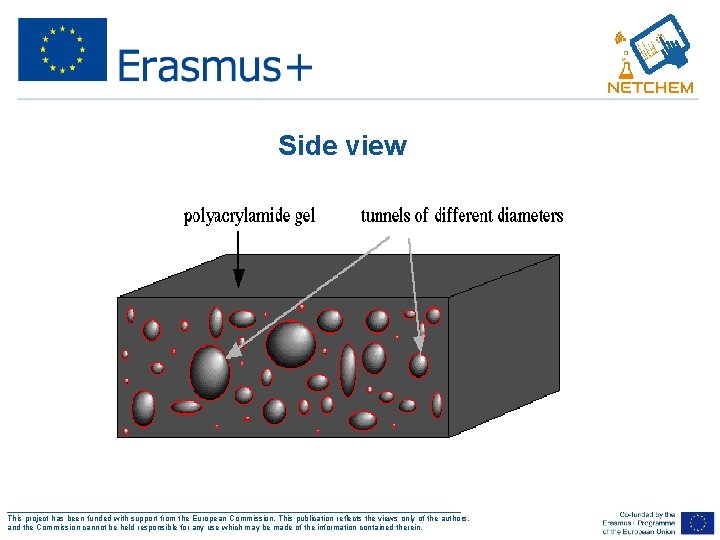
Side view ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

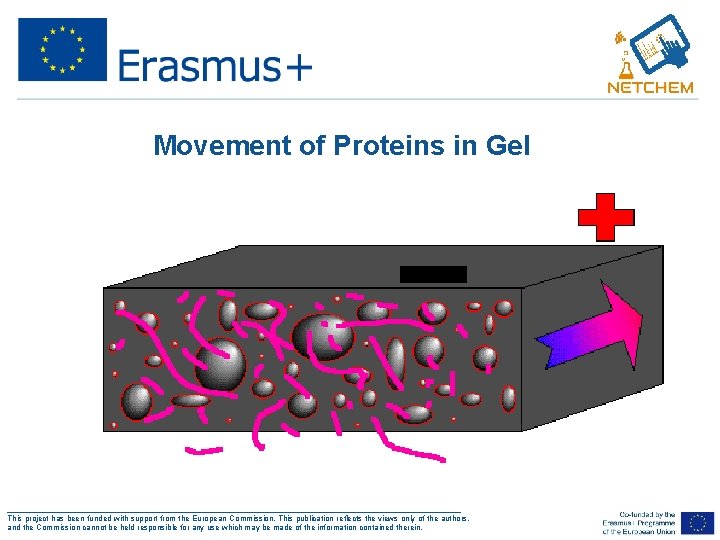
Movement of Proteins in Gel ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

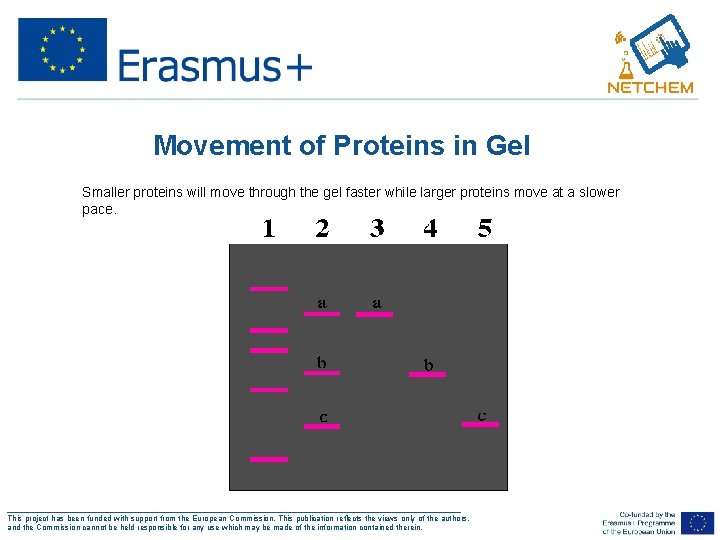
Movement of Proteins in Gel • Smaller proteins will move through the gel faster while larger proteins move at a slower pace. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

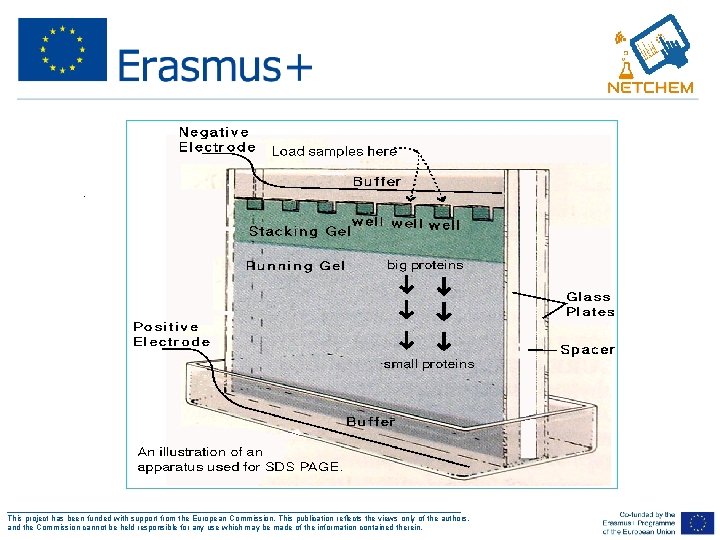
Vertical Gel Format: Polyacrylamide Gel Electrophoresis • • Reservoir/Tank • Power Supply Glass Plates, Spacers, and Combs ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

• . ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Author, Editor and Referee References This remote access laboratory was created thanks to work done primarily at University of Niš. Contributors to this material were: Ph. D. Dhurata Feta, Ph. D. Artiona Laze, full professors Refereeing of this material was done by: ___________ Editing into NETCHEM Format and onto NETCHEM platform was completed by: __________________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

References and Supplemental Material The NETCHEM platform was established at the University of Nis in 2016 -2019 through the Erasmus Programme. Please contact a NETCHEM representatives at your institution or visit our website for an expanded contact list. The work included had been led by the NETCHEM staff at your institution. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.