POLIOMYELITIS Poliomyelitis is a clinicopathologic syndrome characterized by

POLIOMYELITIS

Poliomyelitis is a clinicopathologic syndrome characterized by transient or permanent paresis of one or more extremities and aseptic meningitis.
Etiology Polioviruses belong to the genus Enterovirus (family Picornaviridae). Three polioviruses serotypes are recognized: type 1 -Brunhilde, type 2 -Lansing and type 3 -Leon. - single –stranded RNA genome approximately 7500 bases in length.

Epidemiology The possible routes for polioviruses transmission are: Fecal-oral transmission is the most important route Spread through upper respiratory tract secretions are favored by crowding, water quality, levels of hygiene Risk factors Recent intramuscular injections Injuries Exercise during the early stages of the major illness.

Pathogenesis After ingestion, polioviruses implant in the oropharynx and small bowel (Payer’s patches). “Minor” viremia occurs as a consequence of spread to the regional lymph nodes and to bone marrow, liver and spleen. -“major” viremia, which coincides with the onset of the clinical symptoms associated with the “minor” illness 3 to 7 days after infection. Immunity to poliovirus infection is type-specific.

Clinical manifestations Approximately 95% of poliovirus infections are inapparent. Acute clinical poliomyelitis has 2 phases (” dromedary “ pattern): Minor illness (coinciding with viremia); has a duration of 1 -5 days -incubation period is 3 -7 days -consists of nonspecific symptoms: -frequently resolves within 1 -2 days Asymptomatic period: 3 -5 days

Major illness -incubation period is 9 -12 days Nonparalytic poliomyelitis: -abrupt onset of headache, vomiting, meningeal signs -CSF pleocytosis at an early stage, ; -hypotonia Paralytic stage (within 2 -3 days): -fever -prodromal myalgias; -paresis, paralysis (asymmetric, flaccid, involves proximal muscles of the limbs), progress for 1 -7 days after onset; -deep tendon reflexes become absent; -cranial nerve involvement (the 9 -th and the 10 -th are the most commonly involved).

Evolution Recovery of patients with limb paresis takes weeks to months after acute disease. Paralysis improves slowly as follows: 60% of eventual recovery is achived by 3 months and 80% by 6 months. Minimal further improvements continue over almost 2 years.

Classification: Nonparalytic poliomyelitis: “minor” illness, meningitis (normal value of proteins in CSF in the early stage, and increased level of proteins after 2 weeks of evolution). Paralytic poliomyelitis: spinal forms, “bulbar poliomyelitis” Meningomyeloencephalitis

Complications Respiratory: Cardiovascular Gastrointestinal: paralytic ileus, gastric dilatation; Urinary: ileus of the bladder, urinary tract infection; Decubitus ulcers
Laboratory findings Virus can be grown in tissue culture from pharyngeal swab, (in the first week of the illness), from the feces (for at least 3 weeks), from blood and CSF (less frequently). Serological tests: complement fixation reaction and neutralizing reaction (neutralized antibodies are life-long persistent).

Differential diagnosis Paralytic diseases caused by nonpolio enteroviruses; Guillain-Barre syndrome: paralysis is classically ascending, symmetric, and accompanied by sensory abnormalities. Other conditions with acute paralysis: transverse myelitis, botulism, encephalitis, epidural abscess, intramedullary abscess, cord tumors, brucellosis, syphilis.

Prognosis Nonparalitic poliomyelitis has a complete recovery, and only 5055% of paralysis has the same good evolution. Overall mortality for spinal pliomyelitis is about 4%-6% and for bulbar poliomyelitis is 25 -75%.

Treatment consists of supportive therapy and reduced physical activity for 710 days in nonparalitic poliomyelitis and for 10 -14 days in paralitic forms. Mechanical ventilation is sometimes required in severe cases.

Prevention 1. Live, attenuated poliovaccine (OPV) 2. Inactivated poliovirus vaccine (IPV).

ENTEROVIRUSES INFECTIONS

As members of the genus Enterovirus, the group A coxsackieviruses, group B coxsackieviruses, echoviruses, and newer enteroviruses

Epidemiology The enteroviruses have a worldwide distribution. The proportion of infected individuals who will develop illness varies from 2 to 100 percent

Pathogenesis and pathology After primary replication in the epithelial cells and lymphoid tissues in the upper respiratory and gastrointestinal tracts, viremic spread to other sites can occur.

Illnesses caused by Enteroviruses Acute Aseptic Meningitis Acute aseptic meningitis is a syndrome characterized by signs and symptoms of meningeal irritation and cerebrospinal fluid (CSF) pleocytosis in the absence of bacteria or fungi.

Encephalitis Frank encephalitis is an unusual manifestation of CNS infection with coxsackieviruses and echoviruses that sometimes complicate the course of aseptic meningitis. The differential diagnosis includes: encephalitis due to other viruses (arboviruses, herpes simplex virus -especially in focal encephalitis, mumps); postinfectious encephalitis after measles, rubella, varicella, or pertussis; Reye’s syndrome; Lyme disease; toxic encephalopathies.

Paralysis and other neurologic abnormalities Sporadic cases of flaccid motor paralysis have been associated with several coxsackievirus (A 7, A 9, B 1 -5) and echovirus (6, 9) serotypes, and with enterovirus 71. Guillain-Barré syndrome is caused by coxsackievirus serotypes A 2, A 5, and A 9, and with echovirus serotypes 6 and 22. Systemic coxsackievirus B 2 disease Reye syndrome.

Exanthems Coxsackieviruses and echoviruses cause a variety of exanthems: rubelliform or morbilliform, roseoliform, vesicular, and petechial. Roseoliform exanthems. Exanthem subitum (roseola infantium). Hand-foot-and-mouth (HFM) disease or vesicular stomatitis. Generalized vesicular eruptions are caused by coxsackievirus A 9 and echovirus 11. Petechial and purpuric rashes

Acute Respiratory Disease Undifferentiated febrile illnesses ("summer grippe"). Coxsackieviruses A 21 and A 24 produce illness resembling the common cold. Coxsackieviruses B have been associated with a variety of respiratory illnesses, .

Herpangina primarily affects children between the ages of 3 and 10 years. Coxsackieviruses A (types 1– 10, 16, 22) are the etiologic agents in most cases. The illness begins with fever of 37. 7– 40. 5 0 C. Sore throat and pain on swallowing precede the appearance of the enanthem that begins as punctate macules

Epidemic Pleurodynia Epidemic pleurodynia is an acute infectious disease characterized by fever and sharp, spasmodic pain in the chest or upper abdomen. Pain in the chest may mimic pneumonia, pulmonary infarction, myocardial ischemia, and the preeruptive phase of zoster.

Myopericarditis Enteroviral myocarditis occurs at all ages but has a special predilection for adolescents and active young adults. Most children and adults recover uneventfully. The complications consist in persistent electrocardiographic abnormalities, cardiomegaly, chronic congestive heart failure. Chronic constrictive pericarditis occurs after 5 weeks to 1 year. Differential diagnosis include: others viral myopericarditis

Coxsackievirus and Echovirus Disease in the newborn Infant Many enterovirus serotypes causes the same self-limited clinical syndromes in neonates as they do in older persons (e. g. , aseptic meningitis, exanthems, hand-foot-and-mouth syndrome), but some serotypes are capable of producing fulminant, frequently fatal disease (group B coxsackievirus serotypes 2– 5 and echovirus 11).

Acute hemorrhagic conjunctivitis is a contagious ocular infection characterized by pain, swelling of the eyelids, and subconjunctival hemorrhages.

Laboratory diagnosis The diagnosis is established by: virus isolation from throat, swabs, stool or rectal swabs, body fluids, and occasionally tissues; polymerase chain reaction, enhancing and speeding detection in tissue and CSF, but is not currentlly available serodiagnosis. White blood cell counts and erythrocyte sedimentation rates are usually mildly elevated. Hyperbilirubinemia and elevated transaminase and alkaline phosphatase levels.

Prophylaxis and treatment Glucocorticoids are contraindicated.

GASTROINTESTINAL INFECTIONS

Etiology The microorganisms that cause infectious gastroenteritis are: Bacteria Shigella E. coli (enteroinvasive, enterohemorrhagic E. coli, entero toxigen E. coli) Salmonella spp Vibrio cholerae Vibrio parahaemolyticus Clostridium difficile Clostridium perfringens Campylobacter jejuni S. aureus Bacillus cereus Yersinia enterocolitica

Viruses Rotavirus Astrovirus Caliciviruses Adenoviruses Parvoviruses (Norwalk, Hawaii, agents, etc) Coronaviruses

Parasites Entamoeba histolytica Giardia lamblia Cryptosporidium

Pathogenesis There are three types of enteric infections: Type I - is a noninflammatory (enterotoxin or adherence/superficial invasion) mechanism Etiology of this type of diarrhea: S. aureus Vibrio cholerae Bacillus cereus E. coli (ETEC-entero toxigen E. coli) Clostridium perfringens Rotavirus Norwalk-like viruses Giardia lamblia Cryptosporidium

Type II. Characterized by inflammatory destruction of the ileal or colonic mucosa (by invasion, cytotoxin), producing dysentery illness. Stool examination reveal fecal polymorphonuclear leukocytes and a high level of lactoferrin. Etiology: Shigella E. coli (enteroinvasive and enterohemorrhagic E. coli) Salmonella enteritidis Vibrio parahaemolyticus Clostridium difficile Campylobacter jejuni Entamoeba histolytica

Type III. Characterized by penetration through an intact mucosa to the reticuloendothelial system. Stool examination reveals fecal mononuclear leukocytes. Etiology: Salmonella typhi Yersinia enterocolitica

The intestinal host factors that may help prevent the acquisition of diarrheal disease are: Host species, genotype and age Personal hygiene. Gastric acidity. Intestinal motility, -Normal bacterial flora Intestinal immunity is represented by phagocytic, humoral and cell-mediated elements. . Human milk is protective.

Microbial factors involved in producing disease are: Toxins: Neurotoxins: (Clostridium botulinum, S. aureus, B. cereus) Enterotoxins (V. cholerae, E. coli, Salmonella, Shigella dysenteriae, B. cereus) Cytotoxins (Shigella, C. perfringens, V. parahemolyticus, S. aureus, EHEC, Helycobacter pylori, Campylobacter jejuni) Attachment Ivasiveness Other virulence factors: motility, chemotaxis, mucinase production

General aspects and diagnosis Viral diarhea The principal presentation of rotavirus infection is acute voluminous watery diarrhea with or without vomiting. Diagnosis can be made by electron microscopy, antigen detection or genome detection (RT-PCR)-the latter is the most sensitive and can also be adapted to genotype of the virus. The human astrovirusis are the second commonest cause of diarrhea in hospitalized children. The clinical features are similar to these of rotavirus but in general are less severe. The human Caliciviridae have the typical “star of David” morphology and small round-structured viruses. Epidemic and sporadic diarrhea and vomiting can result from calicivirus infection. Diagnosis has improved greatly with the application of antigen detection and RT-PCR.

ACUTE DISENTERY

Shigella spp. are the main cause of dysentery worldwide and have also been related to hemolytic uremic syndrome. Dysentery is defined as frequent, small bowel movements accompanied by blood and mucus with tenesmus or pain on defecation. There is a substantial risk of person-to person transmission. .

Pathogenesis Invasion of Shigella in the colon produces an inflammatory colitis.

Clinical manifestations The incubation period ranged from 6 hours to 9 days (usually <72) The onset is manifested by fever, followed by small amounts of watery and then bloody diarrhea with mucus. Patients present abdominal pain, tenesmus and pain on defecation. Extraintestinal manifestations.

Complications Hemolytic-uremic syndrome Pseudomembranous colitis Arthritis (10%) - 2 to 5 weeks after the dysenteric illness. Rectal prolapse Reiter syndrome (arthritis, urethritis and conjuntivitis) Toxic megacolon Protein-loosing enteropathy.

Differential diagnosis I. Specific infectious processes II. Proctitis III. Other syndromes Necrotizing enterocolitis Enteritis necroticans Pseudomembranous enterocolitis (Clostridium difficile) Diverticulitis Typlitis

IV. Heavy metal poisoning (As, Sn, Fe, Cd, Hg, Pb) V. Syndromes without known infectious causes: Idiopathic ulcerative colitis Chron’s disease Ischemic colitis Radiation enteritis. VI. Noninfectious endocrine causes: VII. Impaired small bowel absorption: tropical sprue, enzymes deficiencies, solute loads. VIII. Noninfectious chronic noninflammatory diarrhea IX Tumors, polyposis

Principles of treatment in infectious diarrhea Treatment consists of: Correcting and maintaining hydration Eliminating the etiological agent if possible Maintaining adequate nutrition
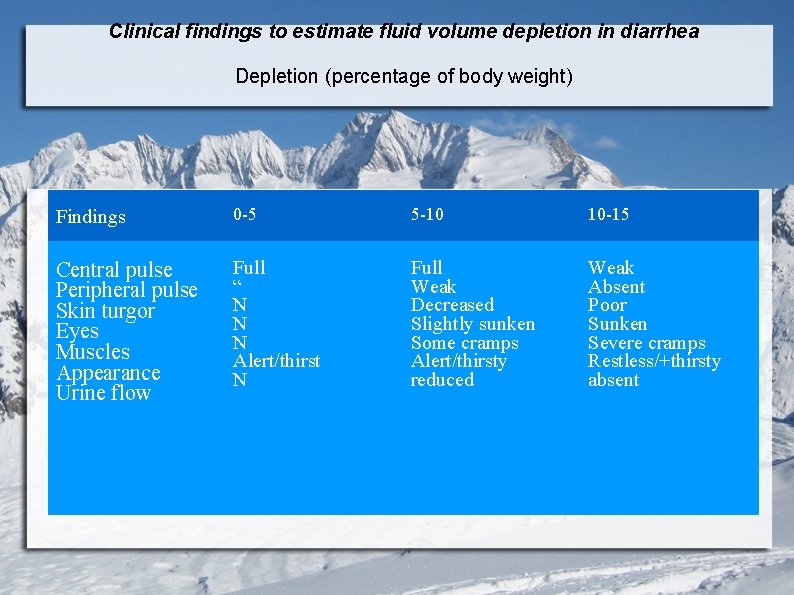
Clinical findings to estimate fluid volume depletion in diarrhea Depletion (percentage of body weight) Findings 0 -5 5 -10 10 -15 Central pulse Peripheral pulse Skin turgor Eyes Muscles Appearance Urine flow Full “ N N N Alert/thirst N Full Weak Decreased Slightly sunken Some cramps Alert/thirsty reduced Weak Absent Poor Sunken Severe cramps Restless/+thirsty absent

A. Replacement of fluid loss Deficit of body weight Volume of fluids <5% 5 -10% 10 -15% 50 ml/kg 50 -100 ml/kg 100 -150 ml/kg

B. Supply of metabolic necessities Body weight Volume of fluids 1 -10 kg 11 -20 kg 21 -80 kg 100 ml/kg 1000 ml+(50 ml/kg x nr of kg>10 kg) 1500 ml+(20 ml/kg x nr of kg > 20 kg)

Composition of oral electrolyte solutions in Who solution (1 packet) is as follows: 3, 5 g Na. Cl, 1, 5 g K, 2, 9 G trisodium citrate dihydrate and 20 g of glucose. The solution is made by addign 1 packet of electrolyte to one liter of water.

FOOD BORNE DISEASES

Food borne disease is defined as an acute illness with gastrointestinal or neurologic manifestations that results from ingestion of contaminated foods (with microorganisms or microbial toxins).

Etiology and pathogenic mechanism of food borne diseases Preformed toxin S. aureus Bacillus cereus C. botulinum Toxin produced in vivo C. perfringens Bacillus cereus • ETEC • V. cholerae Shiga-toxinproducing E. coli C. botulinum (in infants) Tissue invasion • EIEC • Salmonella Shigella Campylobacter jejunii Toxin production and/or tissue invasion V. parahemoliticus Yersinia enterocolitica

Staphylococcal food poisoning – occurs after consumption of ham, milk, eggs, salads, poultry. Characteristics: Short incubation period (1 -6 hours) Manifested by nausea and vomiting (rarely diarrhea) Fever is uncommon Enterotoxins produced by S. aureus are heat-stable protein (A-E)

B. cereus Occurs after ingestion of fried rice, meats, vegetables C. perfringens Food borne disease occurs after ingestion of meat (beef and poultry), gravies. E. coli Enteroaggregative E. coli (EAEC), Enterohemorrhagic (EHEC), Enteropathogenic(EPEC), Enterotoxigenic(ETEC). E. coli remains the most common bacterial enteric pathogen reported worldwide. Enterohemorragic E. coli and especially strain O 157: H 7 has emerged as a cause of serious gastrointestinal disease since its recognition 20 years ago. Clinical manifestations: fever, abdominal cramps and diarrhea bloody diarrhea without fever-when Shiga toxin-producing strains of E. coli are involved. complications are: hemolytic –uremic syndrome, thrombotic thrombocytopenic purpura.

Salmonella Food poisoning occurs after comsumption of: poultry, eggs, beef, dairy products, fresh produce (tomatoes, melons). Clinical manifestations (frequently within 16 -48 hours): fever, abdominal cramps, watery diarrhea. Yersinia enterocolitica serogroup O: 3 is the predominant strain in acute enterocolitis, primarily affecting children younger than 5 years of age. In young children the disease is a febrile diarrhea. In older children and in adults Y. enterocolitica mimics acute appendicitis. Yersinia enterocolitica septicemia is less common and is most often reported in patients with diabetes mellitus, severe anemia, cirrhosis, malignancy and in elderly patients. Erythema nodosum and reactive polyarthritis

Vibrio parahaemolyticus V. parahaemolyticus is a gram-negative, halophilic, marine organism that causes a selflimited gastroenteritis. Diarrhea, cramps, weakness, chills, headache, fever and vomiting Campylobacter jejuni Campylobacter spp. -the cytolethal distending toxin of Diagnosis: fever, abdominal cramps, diarrhea within 16 -72 hours after the ingestion; fecal blood, and polymorphonuclear leukocytes. Duration of illness is < 1 week with relapse in untreated patients. Complications: Guillain-Barre syndrome occurs 1 -3 weeks after diarrheal illness. Treatment: erythromycin.

Differential diagnosis Heavy metal food borne disease (Copper, zinc, Cadmiu) Fish poisoning evolves with symptoms resembling those of a histamine reaction: Mushroom poisoning (onset within 2 hours).

Laboratory diagnosis Culture of different specimens: feces, vomitus, serum, blood and food handlers. Isolation and serotyping of organism from the feces: Rectal swab Toxin testing of food Serology for Y. enterocolitica, V cholerae, Shiga toxin producing E. coli.

Therapy Antibiotics should be avoided in uncomplicated gastrointestinal infections caused by non-typhoidal Salmonella, C. perfringens, B. cereus or staphylococcal food poisoning. Doxycycline and TMP-SMZ have been shown to reduce the incidence of traveler’s diarrhea-is not recommended universal use of doxycycline.

TYPHOID FEVER

Typhoid fever is caused by Salmonella typhi, but, other Salmonellae (S. paratyphi, S. schottmulleri, S. hirschfeldii, and S. choleraesuis) may cause similar clinical syndrome.

Etiology S. typhi, a gram-negative rod, possesses two O antigens (surface polysaccharide) and one H (flagellar) antigen; the envelope antigen, Vi (virulence antigen) is not constant.

Pathogenesis S. typhi are ingested with food/water pass from the stomach into the duodenum. In the distal small bowel they penetrate the intestinal epithelium, over Payer’s patches. They multiply in the intestinal lymphoid tissue and then disseminate via lymphatic circulation or hematogenous route. Local concentration of endotoxin are responsible for cytotoxic and ischemic damage, and circulating endotoxin is responsible of septic shock. Inflammation of the epithelial cells and the subsequent release of chemical mediators, which can stimulate the secretion of fluid into the gut lumen may explain the diarrhea.

Clinical manifestations Incubation period is 7 -14 days. Clinical features are: Fever: begins as a remittent feverduring the first 72 hours, rising in a stepwise fashion after this period associated with chills. Diarrhea is present in 40 -50% of cases, and, sometimes constipation. Headache Diffuse or localized abdominal pain (often in the right upper quadrant) Relative bradicardia Rose spots: maculopapular lesions on the abdominal wall and chest (in the first few days of illness). Splenomegaly Pharingitis is infrequent.

Laboratory findings For definitive diagnosis Isolation of S. typhi/another Salmonella spp from: Blood (positive for about 2 weeks in untreated patients) Bone marrow Stool (positive for several months in untreated patients) Urine Duodenal contents Serology Widal test-detect anti-S. typhi antibodies ELISA using a cell envelope antigen or lipopolysaccharide of S. typhi or purified Vi antigen. PCR.

Additional laboratory tests: • leukopenia • absence of eosinophils on peripheral smears • mildly elevated bilirubin/transaminase level • urinalysis: proteinuria, piuria, casts. • Chest radiographic films reveale infiltrate • Mononuclear cells in fresh stool specimens.

Differential diagnosis Enteric fever-like syndrome causing by other bacteria : Yersinia enterocolitica, Y. pseudotuberculosis, Campylobacter (manifested by fever, headache, abdominal pain). Systemic infection that may mimic typhoid fever: Bacterial infections: Parasitic infections: Viral infections: . Mycotic infections: disseminated histoplasmosis. Rickettsial infections: epidemic/endemic typhus, Q fever, erlichiosis, Rocky Mountain spotted fever.

Complications Major complications are: • hemorrhage from ulcerating lesions –late in the course of disease (2% of untreated patients); • perforations of the terminal ileum or proximal colon; Other complications are: psychosis, cholecystitis, hepatitis, meningitis, pericarditis, pneumonitis, nephritis.

Treatment

Laboratory findings For definitive diagnosis Isolation of S. typhi/another Salmonella spp from: Blood (positive for about 2 weeks in untreated patients) Bone marrow Stool (positive for several months in untreated patients) Urine Duodenal contents Serology Widal test-detect anti-S. typhi antibodies ELISA using a cell envelope antigen or lipopolysaccharide of S. typhi or purified Vi antigen. PCR. Additional laboratory tests: leukopenia absence of eosinophils on peripheral smears mildly elevated bilirubin/transaminase level urinalysis: proteinuria, piuria, casts. Chest radiographic films reveale infiltrate Mononuclear cells in fresh stool specimens.

Differential diagnosis Enteric fever-like syndrome causing by other bacteria : Yersinia enterocolitica, Y. pseudotuberculosis, Campylobacter (manifested by fever, headache, abdominal pain). Systemic infection that may mimic typhoid fever: Bacterial infections: Parasitic infections: Viral infections: . Mycotic infections: disseminated histoplasmosis. Rickettsial infections: epidemic/endemic typhus, Q fever, erlichiosis, Rocky Mountain spotted fever. Complications Major complications are: hemorrhage from ulcerating lesions –late in the course of disease (2% of untreated patients) perforations of the terminal ileum or proximal colon. Other complications are: psychosis, cholecystitis, hepatitis, meningitis, pericarditis, pneumonitis, nephritis.

Treatment Antibiotic therapy can eliminate the fever over 3 to 5 days. Ampicillin: Chloramphenicol. Trimethoprim-sulfamethoxazole; Fluoroquinolones: ciprofloxacin and ofloxacine Third generation cephalosporins; Monobactam.

Evolution Untreated patients maintain a constant temperature pattern for 3 to 4 weeks. Relapses occur in about 10 -15% of treated patients, usually about 2 weeks after therapy was stopped. These patients need another short course of therapy.
Prevention: Typhoid vaccine is recommended for travelers in endemic areas where the water supply is suspect.

BOTULISM

Botulism is a paralytic illness caused by the neurotoxin produced by the bacterium C. botulinum.

Etiology Members of genus Clostridium are gram-positive, anaerobic bacilli. C. botulinum and closely related organisms produce toxins designated as types A, B, C, D, E, F, and G. The toxins causing this disease are among the most potent bioactive substances known (the oral lethal dose for humans is 0. 05 -0, 5 microg).

Pathogenesis There are four clinical type of botulism Foodborne botulism. Wound botulism Infant. “Infectious“ botulism-

Clinical manifestations The latent period is typically 12 -36 hours, but can range from 6 hours to 10 days. Patients are afebrile in the absence of complications. The symptoms are: gastrointestinal-nausea, vomiting, abdominal cramps, diarrhea/constipation, dryness of mucous membranes; neurologic, which are manifestations of cranial nerve dysfunction: muscle weakness; manifestations of parasympathetic nervous system dysfunction: pupils may be dilated or nonreactive.

Complications include : • otitis media; • aspiration pneumonia; • respiratory distress syndrome.

Laboratory diagnosis Detection and identification of botulinum toxin in blood, feces and in the food consumed before onset of illness. Isolation of C. botulinum from the stool. CSF is normal

Differential diagnosis The diseases that may be confused with botulism are: tick-borne encephalitis, diphtheria, poliomyelitis, Guillain-Barre syndrome, myastenia gravis, food poisoning of other etiology. For infant botulism differential diagnosis includes: sepsis, dehydration, pneumonia, Epstein-Barr infection, diphtheria, congenital myastenia gravis, muscular dystrophy, hypothyroidism, metabolic and toxic conditions which may produce weakness and hypotonia. Mortality is 25 -30%.

Therapy Supportive care, nutritional support. Prevention of nosocomial pulmonary and urinary tract infection. Trivalent botulinum antitoxin (neutralize only circulating toxin)-must be administrated as early as possible, with a test for the hypersensitivity. Antitoxin must be given intravenously, in one vial (if there are no signs of hypersensitivity). Hypersensitivity reactions must be averted through the use of human botulism immune globulin. Botulism antitoxin desensitization: with serial subcutaneous injections of antitoxin in 20 -minutes intervals

. Guanidine hydrochloride High-dose penicillin therapy is generally given Antibiotics if there are complications Debridement of the wound C. botulinum spores are not killed by boiling at 100 C. The toxin is heat-labile, so it may be inactivated by boiling of foods before consumption. Mortality is 25 -30%.
CHOLERA

An acute infection involving the entire small bowel, characterized by profuse watery diarrhea, vomiting, muscular crumps, dehydration, oliguria and collapse.

Etiology V. cholerae is not invasive, short (1. 5 -3 mm by 0. 5 mm), curved, motile, gram negative, aerobic rod, oxidase-positive, grows luxuriantly in alkaline media in the presence of bile salts, and produces a neuraminidase that has the property of degrading gangliosides to the monosialosyl form, which is the specific receptor for cholera toxin. Antigenic structure include: - flagellar (H) antigens; antisera prepared against them do not distinguish the vibrio causing human epidemic disease from water vibrios. - somatic (O) antigens do distinguish V. cholerae Ogawa, Inaba,

Pathophysiology V. cholerae is swallowed either with water or food. It must survive passage through the stomach to colonize the small intestine even though it is extremely acid sensitive.

Epidemiology Cholera is spread by ingestion of water, sea foods and other foods contaminated by the excrement of persons with symptomatic or asymptomatic infection.

Clinical manifestations Cholera may be present: in an asymptomatic state, mild diarrhea, or the typical "full-blown" syndrome. The first symptoms of cholera are an increase in peristalsis. There is little abdominal pain in cholera, most of the anxiety, muscle cramps, thirst, and faintness being related in their prominence to the rate of fluid loss. Rarely, ileus may occur at the onset of illness with profound -"Cholera Sicca". Altered consciousness. Electrolyte Uncomplicated cholera is self- limited; recovery occurs within 3 to 6 days. The fatality rate can be > 50% in untreated severe cases but is < 1% with prompt and adequate fluid and electrolyte therapy.

Diagnosis Clinical diagnosis rests on the history of acute onset and the watery stool in the absence of high fever or much abdominal pain.

Laboratory diagnosis The stool, microscopically, will show limited numbers of white cells and rarely red cells. darkfield or phase microscopy. Cultures Antibodies to the specific somatic O antigens: direct agglutination of heated V. cholerae, the agglutination of chicken red blood cells that have been coated with antigens, a vibriocidal test

Treatment The water and salts lost in the cholera stool must be replaced in comparable amounts and concentrations. The replacement can, except in severe cases, be accomplished by mouth (see chapter “Diarrheal diseases”). Intravenous replacement therapy is needed when: the volume of stool output exceeds 100 ml/kg/24 hour, appropriate oral rehydration has not been given

Antimicrobials will shorten the duration of diarrhea and, thereby, reduce fluid losses in cholera. These includes: Tetracycline: Ampicillin, Trimethoprim-sulfamethoxazole Furazolidone: , doxycycline: ) fluoroquinolones: The most important reversible life-threatening complication of cholera after fluid and electrolyte loss is hypoglycemia. .

Prevention Prompt prophylaxis with tetracycline 500 mg orally q 6 h in adults (12 mg/kg q 6 h in children < 8 years of age

TRICHINOSIS

Trichinella spiralis was first seen in human tissue at autopsy in the early 1800 s. By the 1900 s, trichinosis was definitely recognized as a public health problem.

Trichinella spiralis: life cycle stages Time after infection Stage in life cycle first 2 -4 hours 30 h Day 6 Excysted larvae enter intestinal mucosa Worms mature and mate Females deposit larvae/muscle, invasion begins Heaviest muscle invasion Decrease in larval deposition Encapsulation of larvae Intestine free of adult worms Encapsulation almost complete Adult worms die Cyst calcification begins Cyst calcification usually complete Most larvae still viable within calcified cyst Day 10 Day 14 Day 21 Day 23 Month 1 Month 3 Month 6 Year 1 Year 6

Clinical disease Incubation has a 1 -30 days duration Intestinal effects. Muscle penetration and larva encapsulation (invasion phase) – fever, facial (periorbital) edema, muscle pain, swelling, and weakness; conjunctivitis, subconjunctival hemorrhages, pruritus. Skin rashes include: Less frequent manifestations are: Myocarditis. Central nervous system involvement (10 -20%): Pneumonitis: pulmonary infiltrates, pleural reaction Convalescent phase (after 2 months): fever subsides, improvement in muscular symptoms.

Laboratory abnormalities Peripheral eosinophilia (of at least 20%, and possibly up to 90%) is present during the muscle invasion phase of the infection. Leukocytosis Hypoproteinemia Hypokalemia Elevated cretinphosphokinase Mild elevation of alaninaminotransferase level (ALAT)

Diagnosis The history may suggest possible trichinosis (consumption of rare or raw infected meat). Eosinophilia Serologic tests: Muscle biopsy (gastrocnemius, deltoid, and biceps)- does not provide positive results until 2 to 3 weeks after the onset of the illness. Larvae or adult worms are rarely recovered during the intestinal phase (diarrhea). Examination of suspect meat may reveal larvae (artificial digestion procedure).

Differential diagnosis During the intestinal phase: Viral gastroenteritis, Food poisoning During the visceral phase: Typhoid fever Dermatomiositis, Serum sickness Polyarteritis nodosa, Periorbital cellulitis, angioneurotic edema Rheumatic fever Trypanosomiasis Neurologic involvement may mimic: polyneuritis, acute anterior poliomyelitis, myastenia gravis, meningitis, viral encephalitis Cardiac involvement may mimic: viral or bacterial myocarditis, endocarditis, ischemic cardiomyopathy.

Treatment Albendazole: 400 mg po bid, 14 days. Mebendazole: 5 mg /kg po bid, 10 -13 days. Concomitant prednisone: 40 -60 mg po qd; it is indicated in severe disease
- Slides: 107