POLARIZATION USING TRITYLRADICAL WHAT IS TRITYLRADICAL Trityl radical

![CHARACTERISTICS Radical ⊿g/g[10 -3] TEMPO 3. 85± 0. 2 Porphyrexide 3. 0± 0. 3 CHARACTERISTICS Radical ⊿g/g[10 -3] TEMPO 3. 85± 0. 2 Porphyrexide 3. 0± 0. 3](https://slidetodoc.com/presentation_image_h2/fc85bac54a31d0e2a1f6614d3dc255a3/image-5.jpg)
![CHARACTERISTICS Radical ⊿g/g[10 -3] TEMPO 3. 85± 0. 2 Porphyrexide 3. 0± 0. 3 CHARACTERISTICS Radical ⊿g/g[10 -3] TEMPO 3. 85± 0. 2 Porphyrexide 3. 0± 0. 3](https://slidetodoc.com/presentation_image_h2/fc85bac54a31d0e2a1f6614d3dc255a3/image-7.jpg)

- Slides: 9

POLARIZATION USING TRITYLRADICAL

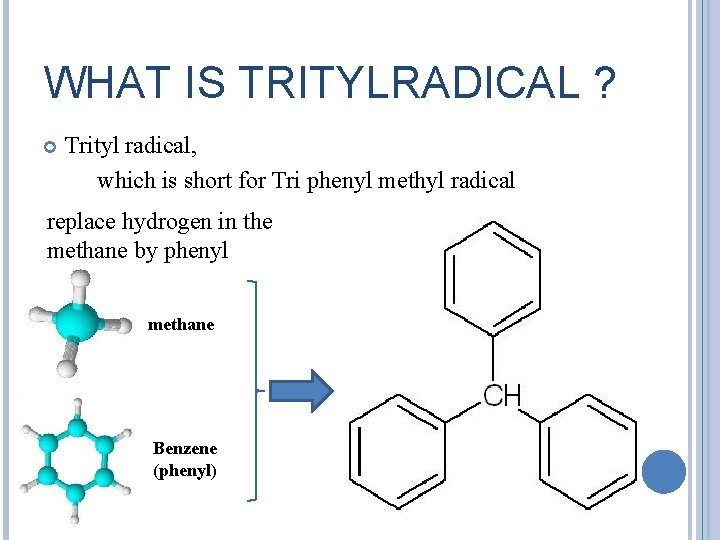
WHAT IS TRITYLRADICAL ? Trityl radical, which is short for Tri phenyl methyl radical replace hydrogen in the methane by phenyl methane Benzene (phenyl)

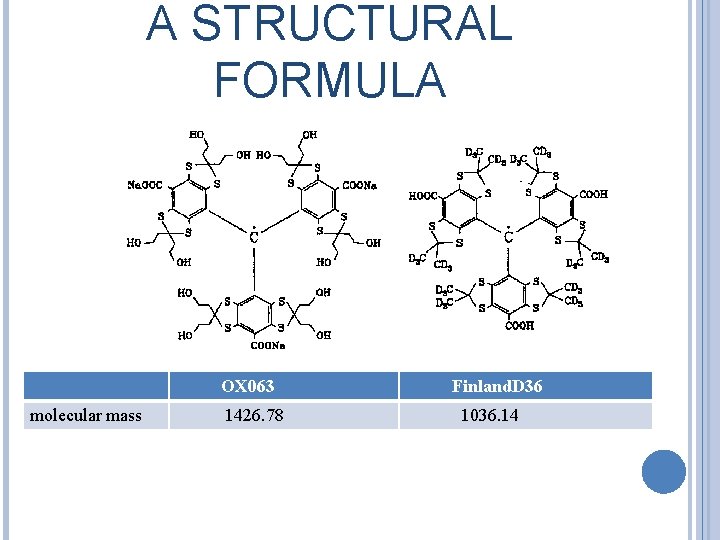
A STRUCTURAL FORMULA OX 063 molecular mass 1426. 78 Finland. D 36 1036. 14

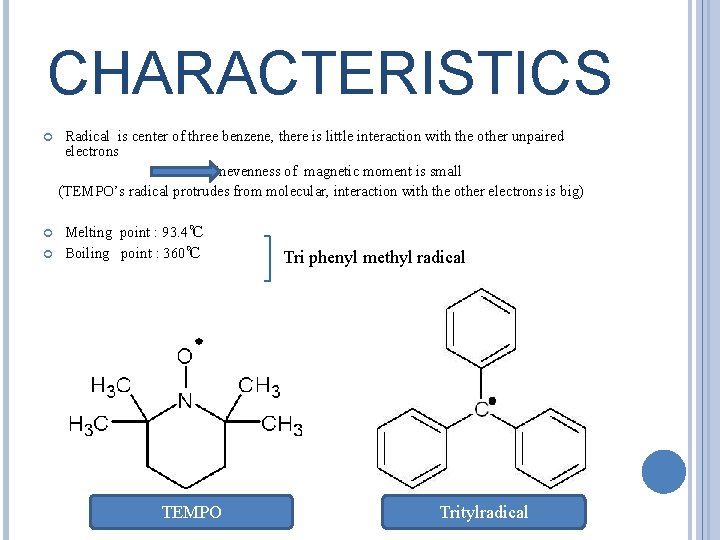
CHARACTERISTICS Radical is center of three benzene, there is little interaction with the other unpaired electrons Unevenness of magnetic moment is small (TEMPO’s radical protrudes from molecular, interaction with the other electrons is big) Melting point : 93. 4℃ Boiling point : 360℃ TEMPO Tri phenyl methyl radical Tritylradical
![CHARACTERISTICS Radical gg10 3 TEMPO 3 85 0 2 Porphyrexide 3 0 0 3 CHARACTERISTICS Radical ⊿g/g[10 -3] TEMPO 3. 85± 0. 2 Porphyrexide 3. 0± 0. 3](https://slidetodoc.com/presentation_image_h2/fc85bac54a31d0e2a1f6614d3dc255a3/image-5.jpg)
CHARACTERISTICS Radical ⊿g/g[10 -3] TEMPO 3. 85± 0. 2 Porphyrexide 3. 0± 0. 3 EHBA/EDBA 6. 0± 0. 2 g=μβH/hν

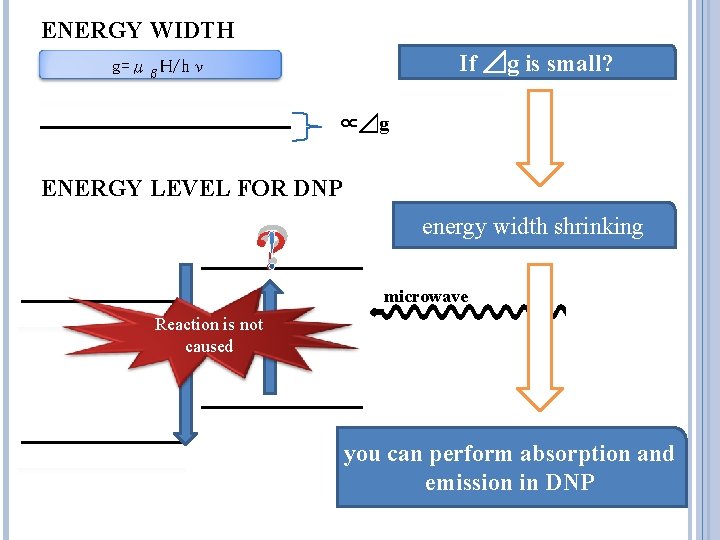
ENERGY WIDTH If ⊿g is small? g=μβH/hν ∝⊿g ENERGY LEVEL FOR DNP ? energy width shrinking microwave Reaction is not caused you can perform absorption and emission in DNP
![CHARACTERISTICS Radical gg10 3 TEMPO 3 85 0 2 Porphyrexide 3 0 0 3 CHARACTERISTICS Radical ⊿g/g[10 -3] TEMPO 3. 85± 0. 2 Porphyrexide 3. 0± 0. 3](https://slidetodoc.com/presentation_image_h2/fc85bac54a31d0e2a1f6614d3dc255a3/image-7.jpg)
CHARACTERISTICS Radical ⊿g/g[10 -3] TEMPO 3. 85± 0. 2 Porphyrexide 3. 0± 0. 3 EHBA/EDBA 6. 0± 0. 2 OX 063 0. 3 g=μβH/hν ⊿g/g of Trityl radical is small in comparison with Nitroxylradical In energy width shrinking, you can perform absorption and emission in DNP

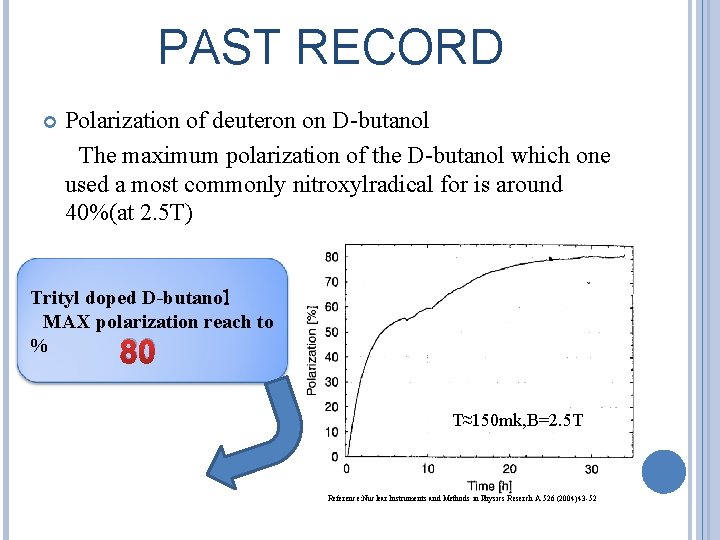
PAST RECORD Polarization of deuteron on D-butanol The maximum polarization of the D-butanol which one used a most commonly nitroxylradical for is around 40%(at 2. 5 T) Trityl doped D-butanol MAX polarization reach to % 80 T≈150 mk, B=2. 5 T Reference: Nuclear Instruments and Methods in Physics Reserch A 526 (2004)43 -52

FROM NOW Trityl radical get mixed in with CD 2 polarization of CD 2 doped TEMPO reach to % CD 2 melts in Xylene(dimethylbenzene) at 100 ℃ Does Tritylradical dissolve in Xylene? Optimize the spin density When doped TEMPO in CD 2, the most suitable value was 2. 0~3. 0× 1019 spin/cm 3 (When doped TEMPO in D-butanol, the most suitable value was 2. 0× 10 , doped OX 063 , value was 1. 5× 10 spin/cm ) 19 19 spin/cm 3. On 3 Try to the world record of the deuteron polarization the other hand