POLARITY INTERMOLECULAR FORCES HYBRIDIZATION Molecular Polarity The uneven

POLARITY INTERMOLECULAR FORCES HYBRIDIZATION
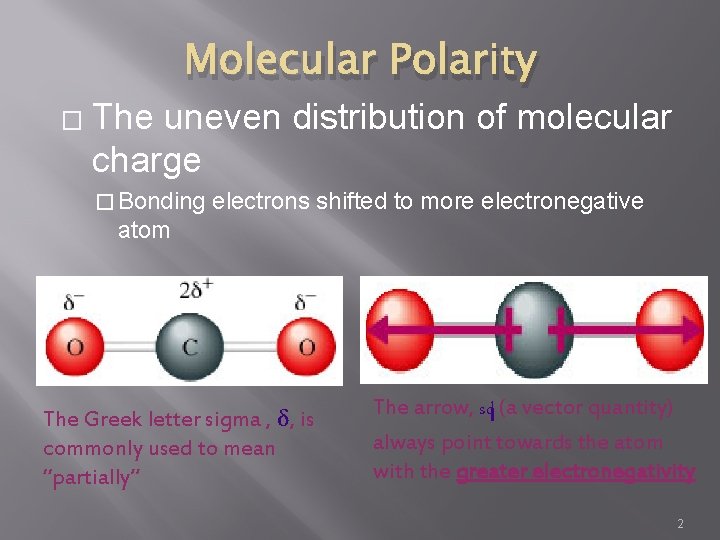
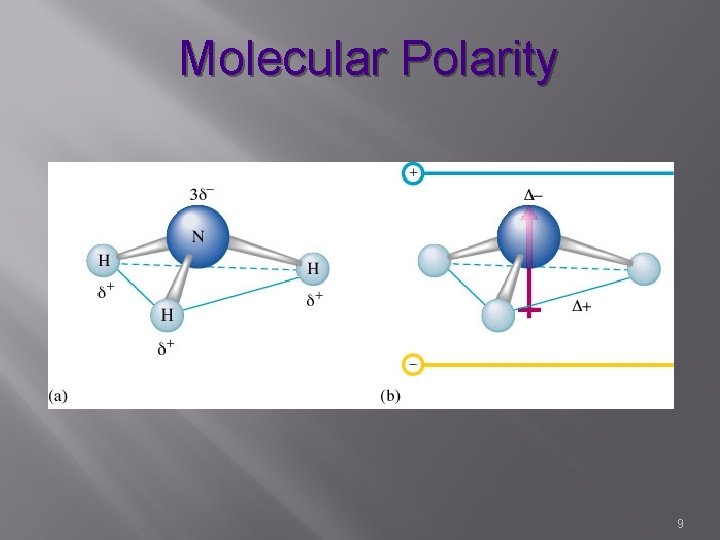
Molecular Polarity � The uneven distribution of molecular charge � Bonding electrons shifted to more electronegative atom The Greek letter sigma , δ, is commonly used to mean “partially” The arrow, sd (a vector quantity) always point towards the atom with the greater electronegativity 2

Molecular Polarity � For molecules containing more than 2 atoms, molecular polarity depends on: �Polarity of each bond EN difference �Orientation of each bond � In other words, both the magnitude and the direction must be considered 3

Molecular Polarity � Conditions for NONPOLAR MOLECULES �All atoms have the same electronegativity (diatomic Br. INCl. HOF) �If central atom is symmetrically surrounded by identical atoms, these molecular shapes permits cancellation of bond dipoles – tetrahedral, trigonal planar, linear �No NET Molecular dipole 4
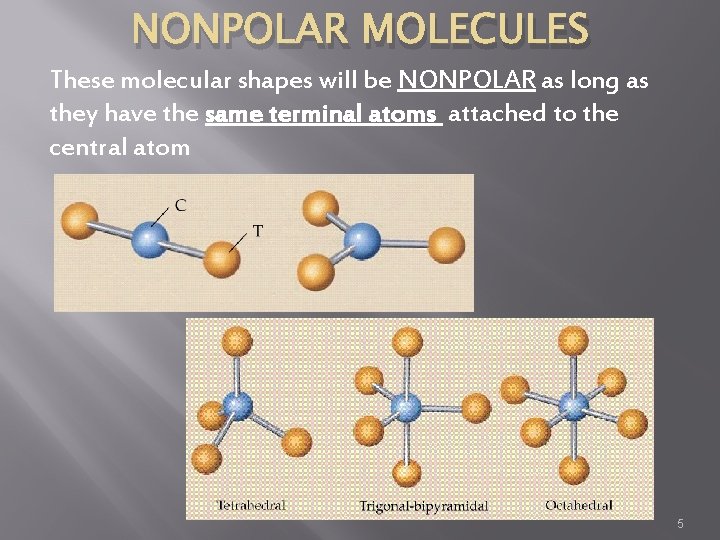
NONPOLAR MOLECULES These molecular shapes will be NONPOLAR as long as they have the same terminal atoms attached to the central atom 5
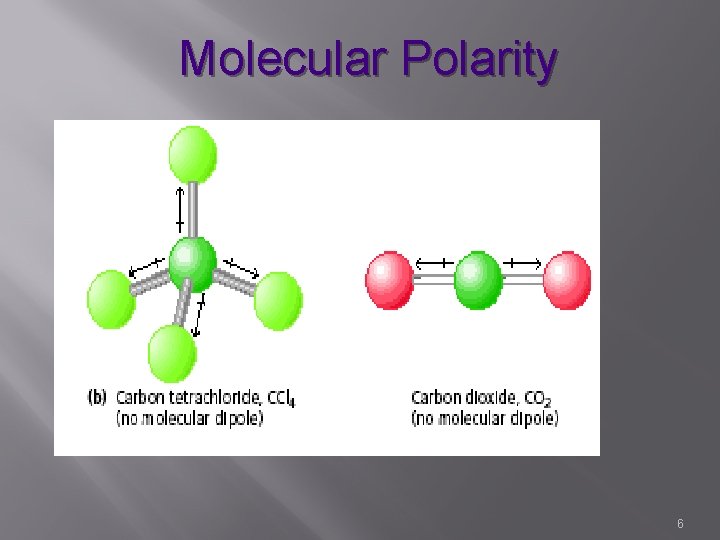
Molecular Polarity 6

Molecular Polarity � POLAR MOLECULES � Contain at least one pair of bonded atoms with different electronegativity (not all identical atoms) � Shapes that do not allow cancellation of bond dipoles – pyramidal, bent ( or other shape with NON identical terminal atoms) � Since dipoles do not directly oppose each other, they do not cancel out (NOT equal and opposite) 7
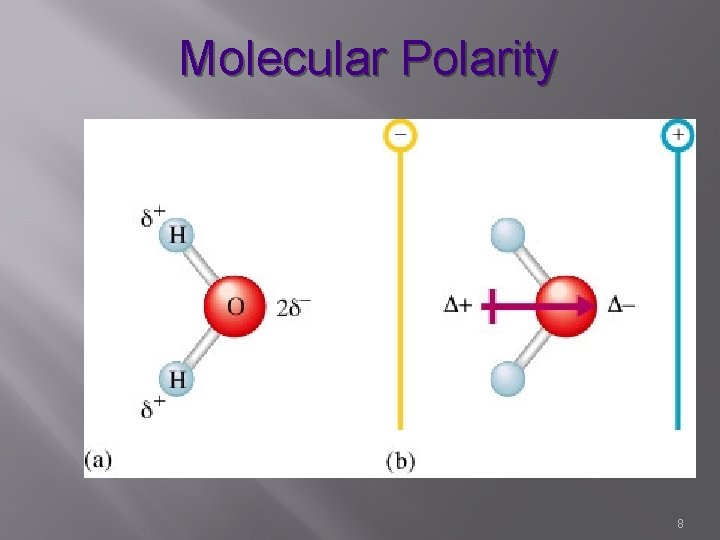
Molecular Polarity 8
Molecular Polarity 9

Intermolecular Forces (vs. Intramolecular forces) � Forces of attraction between molecules � Vary in strength but are generally weaker than bonds that join atoms in molecules, ions in ionic compounds and metal atoms in solid metals � Large numbers can create strong forces � Also known as Van der Waals forces 10

Intermolecular Forces (vs. Intramolecular forces) � Responsible for whether a molecule is gas, liquid or solid �Molecules remain intact �Changes in state/phase due to changes in forces between molecules NOT forces within molecules 11

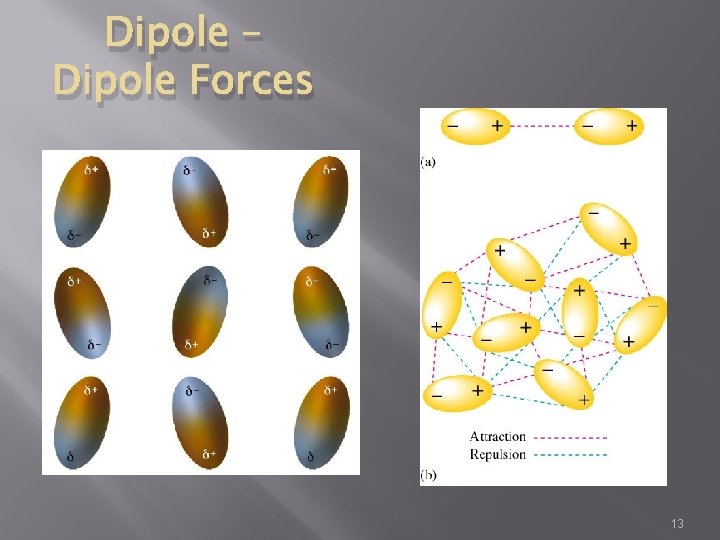
Dipole – Dipole Forces � DIPOLE �molecule that has two electrically charged regions or poles DIPOLE – DIPOLE FORCES Forces of attraction between polar molecules or dipoles Responsible for the relatively high boiling and melting points of polar molecules 12
Dipole – Dipole Forces 13

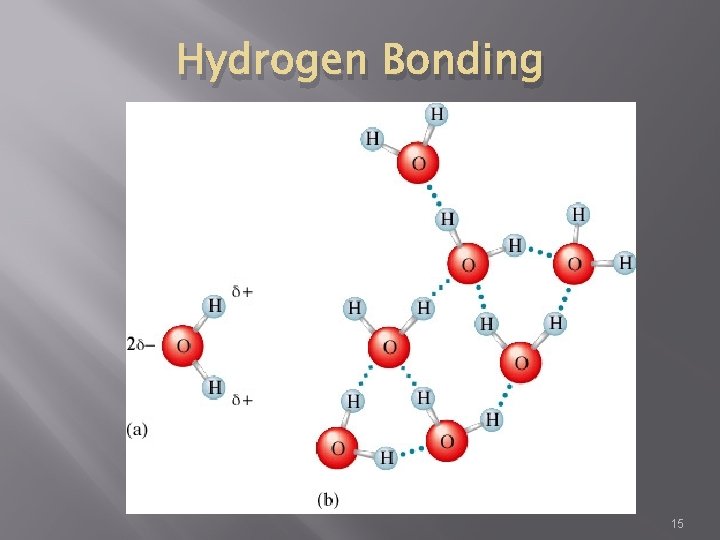
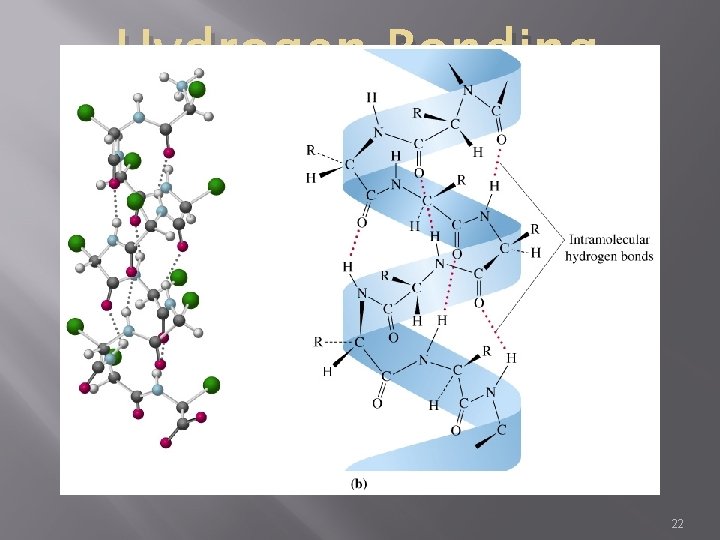
Hydrogen Bonding � Intermolecular force in which a hydrogen atom that is bonded to a highly electronegative atom is attracted to an unshared pair of electrons of an electronegative atom in a nearby molecule � A special type of dipole-dipole force 14
Hydrogen Bonding 15

Properties of H-Bonded Substances Accounts for higher than expected melting and boiling points (more H bonds = higher pt) H bonds occur where H is bonded to a N, O, or F) H 2 O is liquid at room temp (bp: 100°C) while H 2 S with more electrons is a gas at room temp (bp: -62°C) C 2 H 5 OH (bp: 78. 5°C) while CH 3 OCH 3 (bp: 23°C) Increased solubility between substances mutually involving hydrogen bonding Expansion of water upon freezing When water changes to ice, H-bonding between molecules directed by the shape of the water molecules leaves hexagonal holes 16

Hydrogen Bonding � Accounts for the high boiling points of substances containing hydrogen, like water � The strongest of the intermolecular forces due to: �Great polarity of bond �Small size of H atom allows close contact 17

Hydrogen Bonding � About 5% stronger than the average covalent bond � Extremely important in determining the properties of water and biological molecules such as proteins 18

Properties of H-Bonded Substances Shape and stability of certain chemical structures Biochemical processes Allows blood (mostly H 2 O) to absorb and transport a large amount of nutrients DNA’s alpha helix shape is due to hydrogen bonds between strands Behavior and stability of protein molecules are related to their shape which is related to H-bonding present 19

Hydrogen Bonding � 20
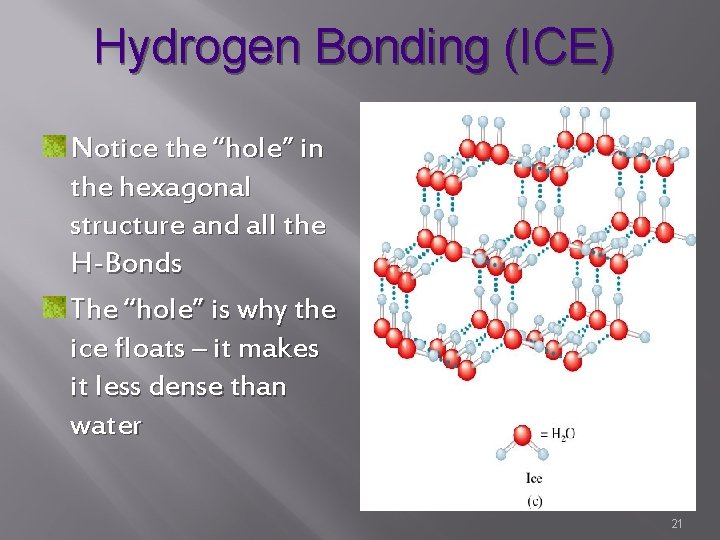
Hydrogen Bonding (ICE) Notice the “hole” in the hexagonal structure and all the H-Bonds The “hole” is why the ice floats – it makes it less dense than water 21
Hydrogen Bonding 22

Hydrogen Bonding Surface Tension 23

London Dispersion Forces � Results from the constant motion of electrons and the creation of instantaneous/temporary dipoles � Strength increases with increasing atomic or molar mass �More electrons, stronger LDF �Increases chance of temp. dipole formation 24

London Dispersion Forces 25

London Dispersion Forces � Halogens have major attraction for one another due to dispersion forces �Chlorine is a gas –fewest total e�Bromine is a liquid since it has greater total # of electrons which generate larger dispersion forces �Iodine has bigger dispersion force due to largest # of e- making it solid at room temperature 26
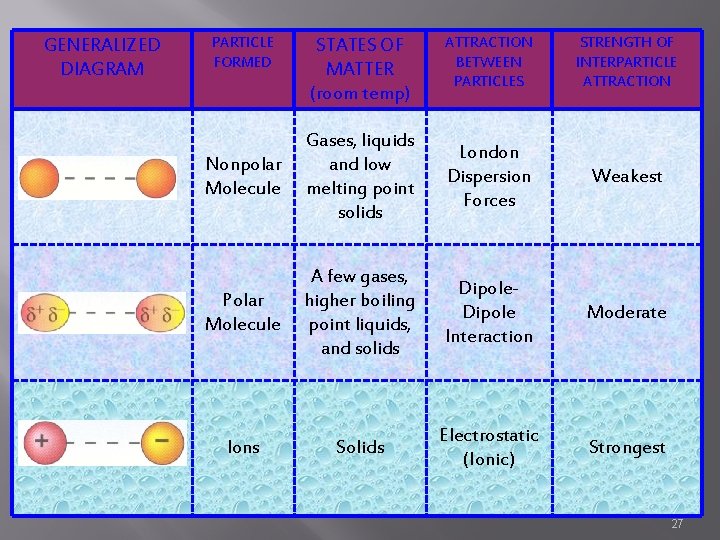
GENERALIZED DIAGRAM PARTICLE FORMED STATES OF MATTER (room temp) ATTRACTION BETWEEN PARTICLES STRENGTH OF INTERPARTICLE ATTRACTION Nonpolar Molecule Gases, liquids and low melting point solids London Dispersion Forces Weakest Polar Molecule A few gases, higher boiling point liquids, and solids Dipole Interaction Moderate Ions Solids Electrostatic (Ionic) Strongest 27

Strength of Bonds & Forces � � The stronger the oppositely charged forces, the stronger the bond Strong to Weak � Ionic bond>H bonding>covalent bond>dipole-dipole force>London dispersion force � Remember: More charge attraction = stronger bond!! 28

Hybridization � � � VSEPR theory explains the shapes of molecules but does not reveal the relationship between a molecule’s geometry and the occupied orbitals. The hybridization model does. IMPORTANT TO UNDERSTAND: Hybridization happens as an atom is bonding & the type depends on what other atoms the atom is bonding with. Relationship between electrons in orbitals and molecular shape. 29

Hybridization � � Whenever we mix a certain number of atomic orbitals, we get the same number of hybrid orbitals. Each of these hybrid orbitals is equivalent to the others but points in a different direction. Several atomic orbitals mix to form the same # of equivalent hybrid orbitals on the same atom Results in greater stability and stronger bond (THIS IS THE GOAL) 30

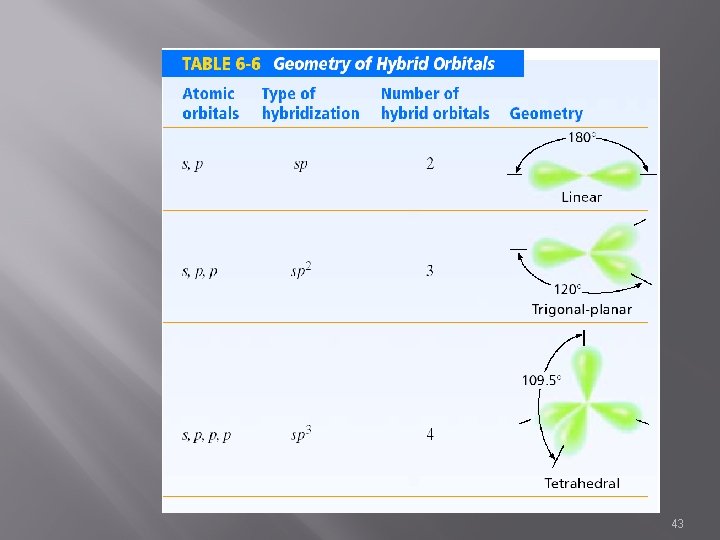
Hybridization � � � Hybrid orbitals are orbitals of equal energy, length, strength, etc to each other Explains the bonding and geometry of many molecules including those formed by Group 15 and 16 elements (atoms with multiple valences & charges) sp 3, sp 2, sp 31
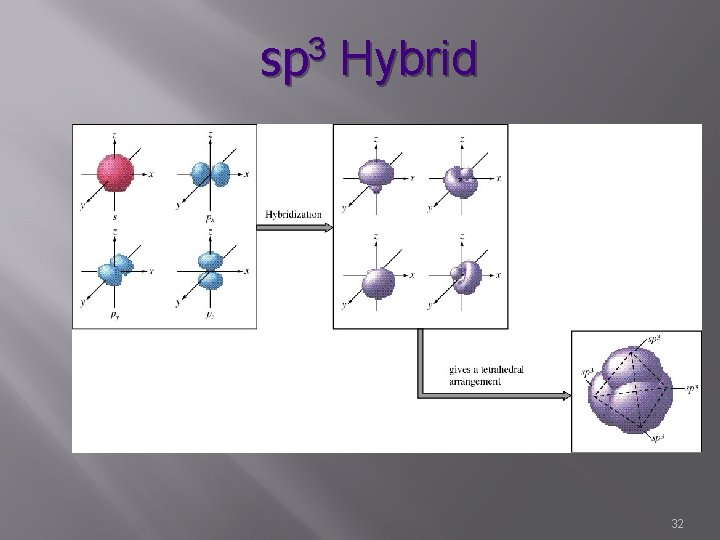
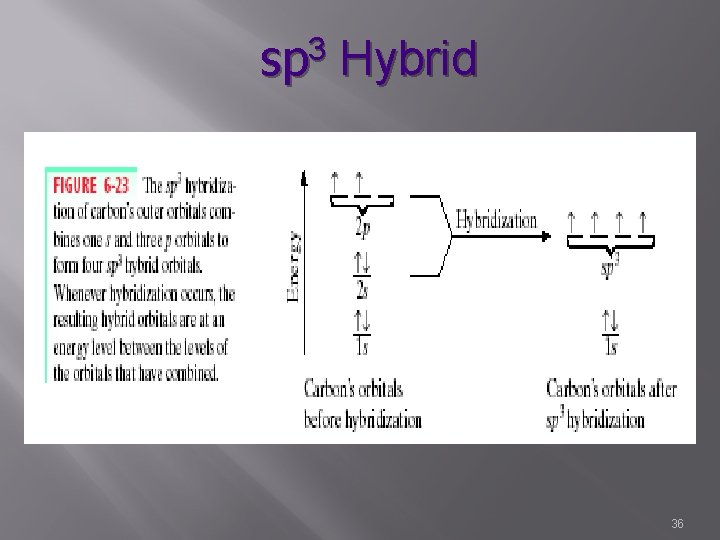
sp 3 Hybrid 32
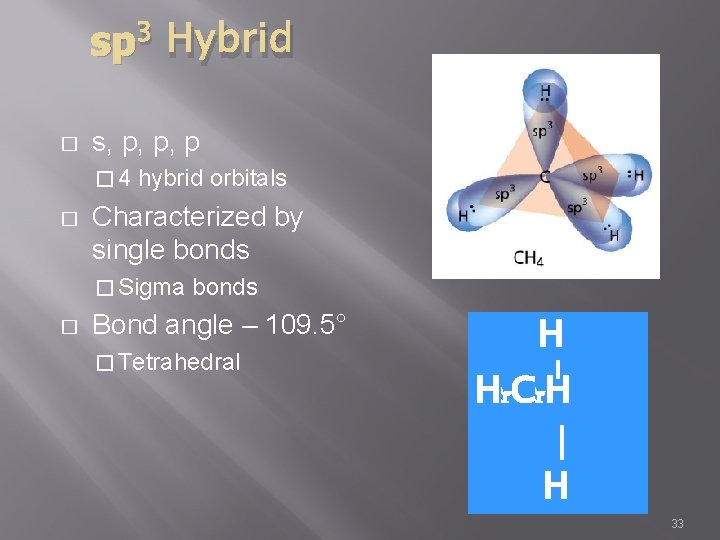
sp 3 Hybrid � s, p, p, p � 4 � hybrid orbitals Characterized by single bonds � Sigma � bonds Bond angle – 109. 5° � Tetrahedral H Hr. Cr. H | H 33

Example: Promotion � Carbon � Example: know basic concept not exact example � Has 4 valence e� 2 s 2 2 p 2 � 2 bonds expected � Example: � 4 actual bonds due to hybridization of one s and three p orbitals to form four sp 3 hybrid orbitals 34

Promotion Process � Each p orbital gives some energy to the s orbital �p comes down in energy � s comes up in energy � Hybridized sp 3 molecular orbital is formed � All equivalent to each other in bond stability, bond length, bond energy, and bond strength (but between that of the parent orbitals) 35
sp 3 Hybrid 36
sp 2 Hybrid 37
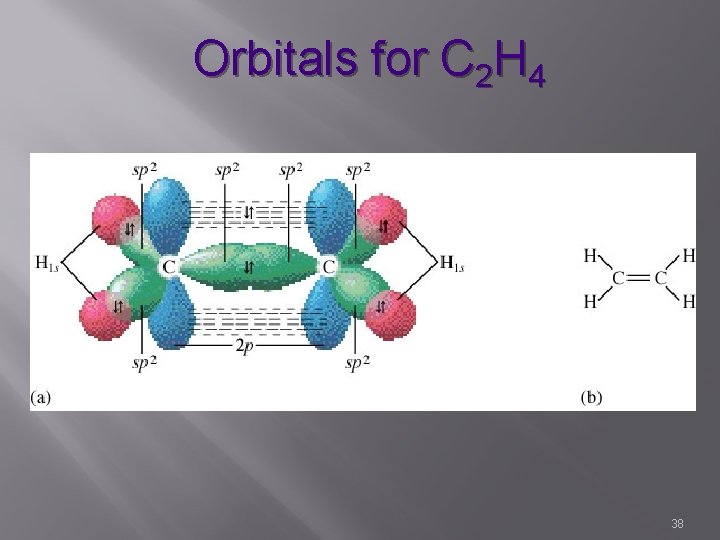
Orbitals for C 2 H 4 38

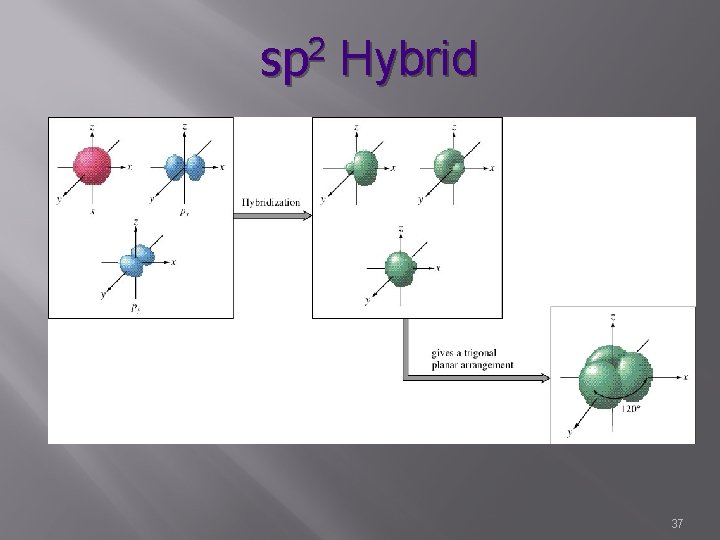
sp 2 Hybrid s, p, p 3 hybrid orbitals & 1 unhybridized Characterized by double bonds 1 sigma bond 1 pi bond H H / C=C / H H Bond angle – 120° Trigonal Planar 39
sp Hybrid 40
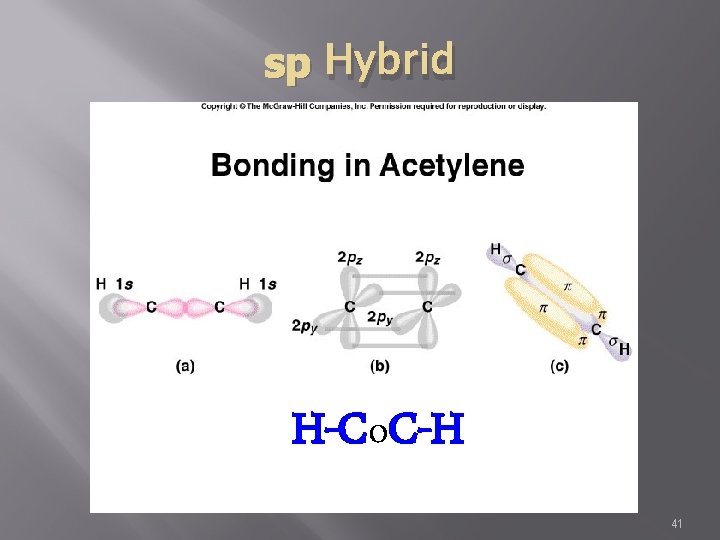
sp Hybrid H-Co. C-H 41
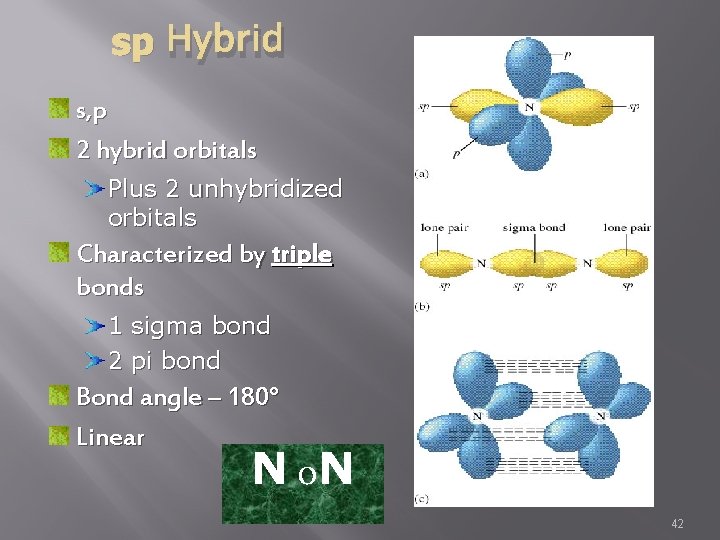
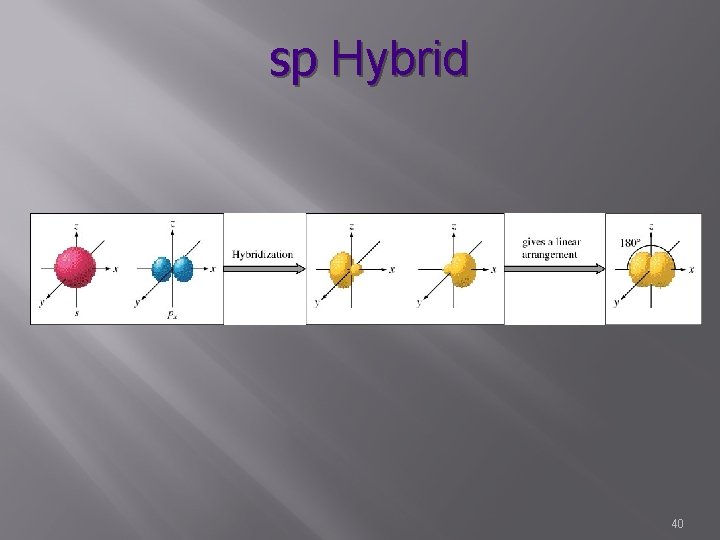
sp Hybrid s, p 2 hybrid orbitals Plus 2 unhybridized orbitals Characterized by triple bonds 1 sigma bond 2 pi bond Bond angle – 180° Linear N o. N 42
43

� Practice Problems Determine the type of hybrid orbitals involved in the bonding of carbon atoms for the following molecules: �H 3 C-NCO �CH 4 �H 2 Ct. CH 2 �HCo. CH �No. C-Co. N 44
- Slides: 44