Polarity in Covalent Bonds Polar Chemical Bonds being

Polarity in Covalent Bonds

Polar Chemical Bonds • being polar refers to: • uneven distribution of electrons • one end of bond has more electrons than other end • due to large electronegativity difference between bonded atoms

Non-polar Chemical Bonds • being non-polar refers to: – even distribution of electrons – electron cloud same on one end of bond as other end – due to low or no electronegativity difference between bonded atoms

H 2 is symmetric: - same # e- at each end - football-shaped electron cloud HCl is asymmetric: - different # e- at each end - electron cloud is lopsided (Cl side has more e- than H side)

How can predict if BOND is polar or not? compare difference in electronegativity values of two atoms joined in bond

Electronegativity ability of atom to attract electrons in bond values found in Table S!

Bond Polarity • more electronegative atom has greater pull on bonding e– gets more than fair share of electron cloud (uneven distribution of e-) – leads to separation of charge (delta notation) • electron-rich side partially negative ( -) • electron-poor side partially positive ( +)

BOND Polarity • depends on electronegativity difference between two atoms A B elements A and B chemically bonded together EA - EB • - care only about SIZE of difference, not sign • - bigger the difference, the more polar the bond

Bond Polarity • What can say about polarity of bond where atom A is same as atom B? both atoms are same so electronegativity difference = 0 expect bond to be non-polar

electronegativity difference bond type 0. 0 to 0. 4 non-polar covalent >0. 4 to <1. 7 polar covalent ≥ 1. 7 ionic

calculate electronegativity difference for each of these bonds Li. H : 2. 2 – 1. 0 1. 2 FALLS IN polar covalent range but is IONIC!! H 2 : 2. 2 – 2. 2 0 non- polar covalent HF: 2. 2 – 4 1. 8 FALLS IN ionic range but is POLAR COVALENT!!


Review • Which of following bonds is most polar? A) O 2 3. 4 – 3. 4 = 0 2. 2 – 3. 2 = -1. 0 = 1. 0 B) HCl 3. 0 – 2. 2 = 0. 8 C) NH in NH 3 3. 0 – 2. 2 = 0. 8 D) HBr answer =(B) HCl

Review • Which substance contains bond with greatest ionic character? A) KCl 0. 8 – 3. 2 = -2. 4 = 2. 4 2. 2 – 3. 2 = -1. 0 = 1. 0 B) HCl 3. 2 – 3. 2 = 0 C) Cl 2 D) CCl 4 2. 6 – 3. 2 = -0. 6 = 0. 6 answer = (A) KCl

Summary • non-polar covalent bonds form between: – atoms having equal or low electronegativity values: 0. 0 – 0. 4 • polar covalent bonds form between: – atoms with greater electronegativity difference in values: >0. 4 to <1. 7
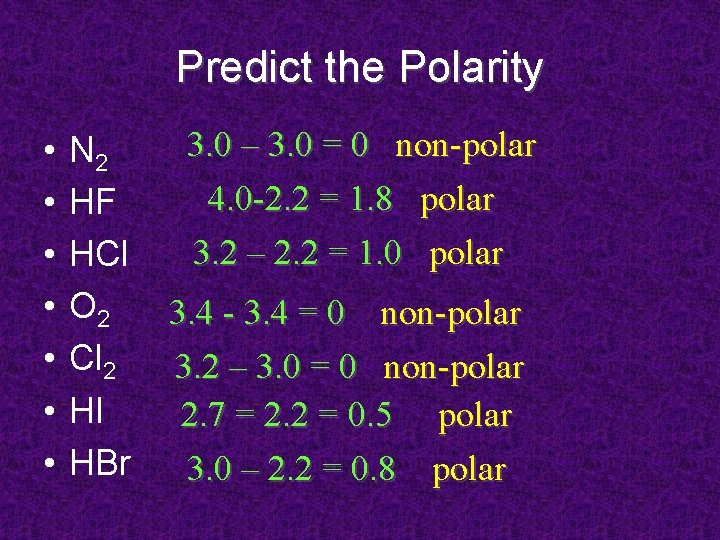
Predict the Polarity • • N 2 HF HCl O 2 Cl 2 HI HBr 3. 0 – 3. 0 = 0 non-polar 4. 0 -2. 2 = 1. 8 polar 3. 2 – 2. 2 = 1. 0 polar 3. 4 - 3. 4 = 0 non-polar 3. 2 – 3. 0 = 0 non-polar 2. 7 = 2. 2 = 0. 5 polar 3. 0 – 2. 2 = 0. 8 polar

MOLECULE Polarity • diatomic molecules: – only ONE BOND so molecule polarity is same as bond polarity

MOLECULE Polarity (3+ elements) • depends on 2 factors 1. type bonds in molecule 2. arrangement of bonds or shape of molecule Think SYMMETRY!!
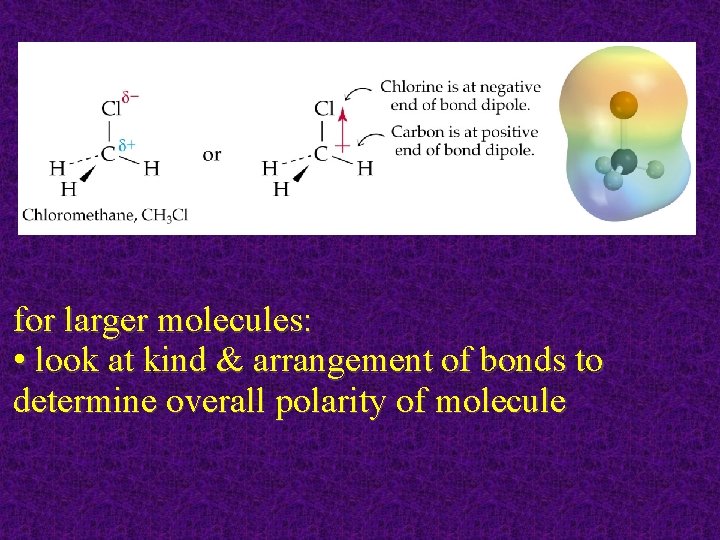
for larger molecules: • look at kind & arrangement of bonds to determine overall polarity of molecule

Polarity of Molecules • molecule may contain polar bonds, and not be polar! – depends on geometry of molecule • if molecule is symmetric: – “pull” of one polar bond is offset by “pull” of another polar bond – tug-of-war that no one can win!
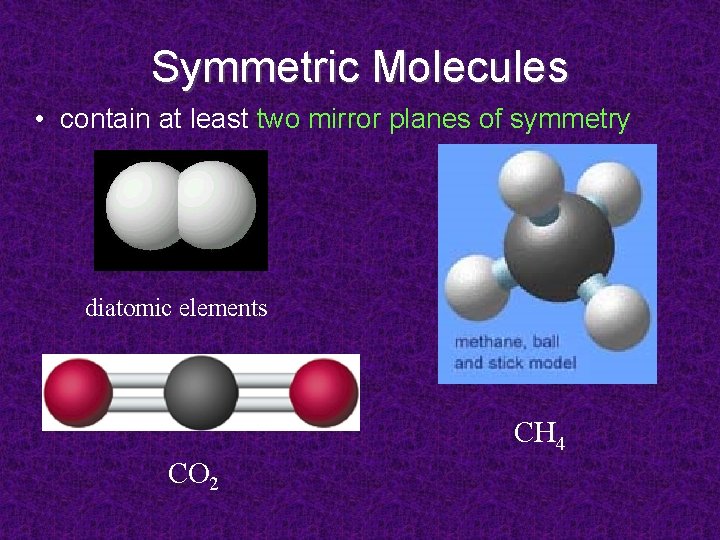
Symmetric Molecules • contain at least two mirror planes of symmetry diatomic elements CH 4 CO 2
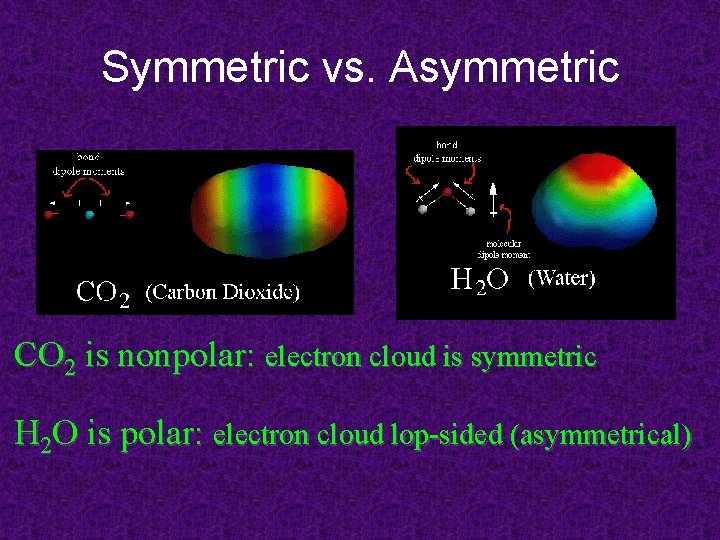
Symmetric vs. Asymmetric CO 2 is nonpolar: electron cloud is symmetric H 2 O is polar: electron cloud lop-sided (asymmetrical)
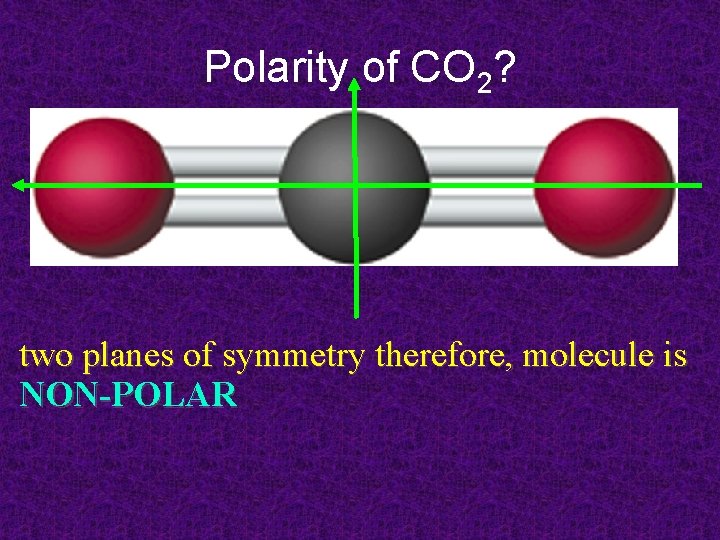
Polarity of CO 2? two planes of symmetry therefore, molecule is NON-POLAR
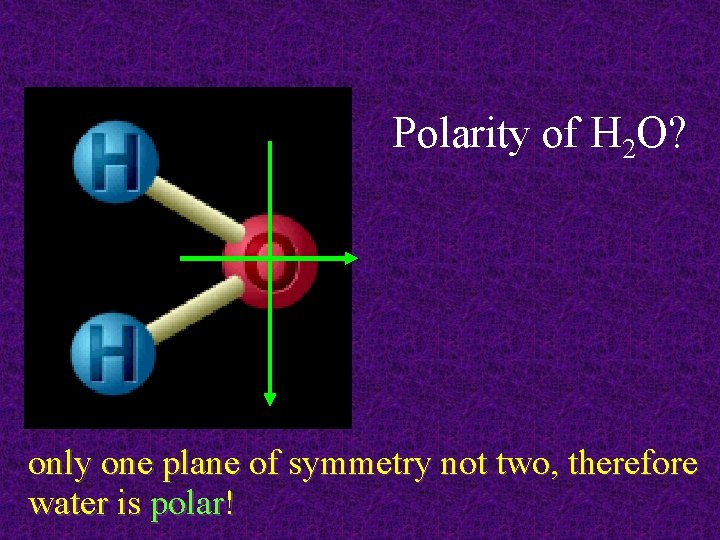
Polarity of H 2 O? only one plane of symmetry not two, therefore water is polar!
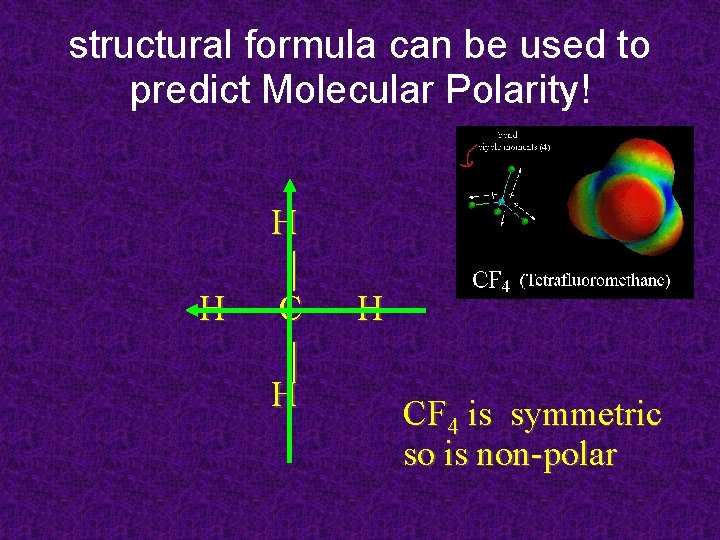
structural formula can be used to predict Molecular Polarity! H H CF 4 is symmetric so is non-polar
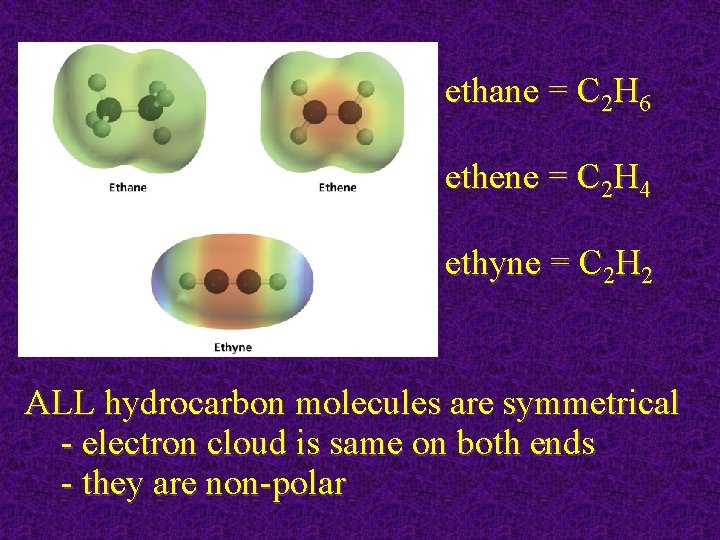
ethane = C 2 H 6 ethene = C 2 H 4 ethyne = C 2 H 2 ALL hydrocarbon molecules are symmetrical - electron cloud is same on both ends - they are non-polar

Compare & Contrast BOND POLARITY MOLECULE POLARITY • All about the ELECTRONEGATIVITY difference between the TWO atoms in a bond • All about the SYMMETRY of the molecule • NON-POLAR BOND: electronegativity difference is 0. 4 or less • POLAR BOND: electronegativity difference is > 0. 4 & <1. 7 • SYMMETRICAL molecules are NONPOLAR • ASYMMETRICAL molecules are POLAR
- Slides: 27